#NephMadness 2026: Animal House Region
Submit your picks! | @NephMadness | @nephmadness.bsky.social | NephMadness 2026
Selection Committee Member: Autumn Harris
At NC State University, Dr. Autumn Harris investigates how kidneys regulate acid-base balance and what drives kidney disease progression. Her clinical trials seek earlier diagnosis and better treatments for chronic kidney disease in dogs and cats—advancing both veterinary care and comparative nephrology.
Selection Committee Member: Kelly Hyndman @DrKeeksPhD
Kelly Hyndman is an Associate Professor of Medicine in the Section of Cardio-Renal Physiology and Medicine, Division of Nephrology, at the University of Alabama at Birmingham. She has trained both in comparative physiology and kidney physiology labs and is currently a principal investigator of a basic science lab with research interests in novel mechanisms of fluid-electrolyte balance.
Writer: Sarah Street
Sarah Street started with a PhD in Physiology at the University of North Carolina at Chapel Hill. Directing an MS1 Physiology course for 5 years inspired her to attend UNC medical school herself. For residency and Nephrology fellowship, she went north to Massachusetts General Hospital. She is interested in medical education, mentorship, and clinical reasoning. She is an Assistant Professor of Medicine at Virginia Commonwealth University and a co-director of MS2 Renal Course and MS3 Internal Medicine Clerkship. Importantly, she is mom to the most precious Goldendoodle Mabel.
Writer: Susan DiGiovanni
Susan DiGiovanni is a Professor Emeritus of Medicine and Nephrology at Virginia Commonwealth University. She recently retired from a career focused on medical education, including Assistant Dean for Preclinical Education and Senior Associate Dean for Medical Education. Now she is seeing patients part time and enjoying traveling, reading and taking care of her four cats Cleopatra, Remus, Molly Weasley and O’Malley.
Competitors for the Animal House Region
This year’s Animal House invites us to discuss nephrology as it applies to our canine companions and feline friends, appreciate the interconnectedness of humans, animals, and our shared environment, and join efforts to advance One Health.
Team 1: Dogs
versus
Team 2: Cats

Image generated by Matthew Sparks using ChatGPT at http://chat.openai.com, February 2026. After using the tool to generate the image, Sparks and the NephMadness Executive Team reviewed and take full responsibility for the final graphic image.
Team 1: Dogs
“And the Woman said, ‘His name is not Wild Dog any more, but the First Friend, because he will be our friend for always and always and always.’” – Rudyard Kipling
Humans and dogs have co-evolved to form one of the most successful symbiotic relationships in biological history, so it’s no surprise that our understanding of human and canine kidneys is closely enmeshed. Let’s learn how canine Glomerular Gladiators moved on from disease models to full participants in large scale clinical trials.

Copyright: @Dogs4NephMadness, photo courtesy of the owner
The most remarkable evolutionary story of our canine companions likely started some 23,000 years ago, in the bitter cold of the last Ice Age. Those curious wolves that forged the earliest semblance of a canine-human connection by prowling campsites for food scraps would hardly recognize their modern progeny – complete with personal social media followings and luxe, bone-shaped-pool-equipped doggie daycares. Humans and dogs have co-evolved to form one of the most successful symbiotic relationships in biological history, so it’s no surprise that our understanding of human and canine kidneys is similarly closely enmeshed.
In fact, how humans and dogs have benefitted one another in our quest to understand and treat kidney disease, parallels the overarching themes of our co-evolution in a shared environment. Similar to how our ancestors might have pragmatically viewed their dogs as mere hunting partners, our interest in canine kidney physiology was initially driven by their role as experimental models. For instance, beagles have been widely used for investigating both diabetic and hypertensive disorders, with type I diabetes chemically induced by alloxan and streptozocin followed by unilateral nephrectomy to accelerate kidney failure. Without a doubt, canine animal models have furthered our understanding of renal disease; this knowledge is now being reciprocally applied back to our beloved pets in order to diagnose and treat their kidney problems. After all, they have become close members of our families, and we would do anything to keep them healthy and thriving, since the biggest flaw in our co-evolved history is that they just don’t live as long as we want them to (which honestly, would be forever!).
Dogs tend to have a higher GFR than other small domesticated animals like cats. While this is their normal physiological state, it may render the glomerular filtration barrier more vulnerable to damage from systemic stressors. In a dog, a disease that raises blood pressure or induces inflammation could exacerbate pre-existing intraglomerular hypertension, contributing to “leakiness” of the glomeruli. As in human glomerular disease, proteinuria is the hallmark and often the earliest clinical finding. Differences in the canine immune system (for instance, a hypothetically more active Th2 immune response compared to cats, favoring antibody generation ) can also make dogs more susceptible to glomerular conditions, often mirroring human ones. Persistent antigenic stimulation from multitudes of infections they can get, as well as autoimmune and neoplastic causes, tends to overwhelm dog glomerular clearance mechanisms and result in immune complex glomerulonephritis (ICGN). ICGN is, in fact, responsible for half of canine glomerular disease cases. Fascinating examples are Lyme nephritis, an ICGN originally described and quite prevalent in dogs but rare in humans, and heartworm nephritis, which thankfully does not have human correlates.
As our understanding of human glomerular disease has advanced, interest in applying it to canines has followed. A mere twenty years ago, canine glomerular disorders were simplistically classified as either amyloidosis or glomerulonephritis. Further, when biopsies were taken, they were only examined with light microscopy. Fortunately, evaluation has now become much more sophisticated. Acknowledging that the decision to subject an animal to immunosuppressive therapies requires a reliable diagnosis, the World Small Animal Veterinary Association Renal Pathology Initiative released an updated pathological classification of canine glomerular disease that recommends not only light microscopic evaluation, but immunofluorescence and transmission electron microscopy as well. The classification schema divides glomerular diseases into immune-complex and non-immune complex and from there into 8 subdivisions mirroring human pathological descriptions such as FSGS, membranous and membranoproliferative nephropathies, etc. This kidney disease schema for dogs is a great example of how our understanding of human glomerular disease enabled greater clarity for the diagnosis of our canine counterparts. Management is also heavily influenced by studies in humans. Some treatments, like ACE inhibitors and angiotensin receptor blockers (ACEi/ARBs) are backed by multicenter randomized controlled trials showing improvement in proteinuria, whereas others, such as glucocorticoids, mycophenolate, and cyclophosphamide for biopsy-proven ICGN, are based on consensus guidelines.
The overlap of genotypic/phenotypic pathologies, isolated populations, and shared environments has made dogs exceptional models of inherited kidney diseases. Believe it or not, dogs have more naturally occurring genetic diseases than any species besides humans. Most are found in specific breeds, a direct consequence of a founder effect in animals selected for desired traits. Remarkably, more than half have a human equivalent, including hereditary disorders like type IV collagen defects (seen in Samoyeds, English Cocker Spaniels, Bull Terriers, and Dalmatians) and podocytopathies (in Wheaten and Airedale Terriers, or Miniature Schnauzers). Newer technologies are being employed to understand these diseases in dogs and, by proxy, in humans. Take, for instance, a recent study where RNA sequencing was done on biopsies from dogs with X-linked Hereditary Nephropathy, a canine analog of Alport Syndrome. The dogs were grouped according to speed of progression, and the study identified 70 differentially expressed genes and upstream regulators associated with fibrosis/adhesion and inflammatory pathways that are likely driving progression of kidney disease. These transcripts and regulators could eventually be used as an alternative way to stage kidney disease or potentially as targets for treatments in both dogs and humans.
Acute kidney injury (AKI) in dogs presents with remarkably similar symptoms and pathophysiology to what we experience as humans, including anorexia, lethargy, vomiting and polyuria. Interestingly, etiologic distribution is different from human AKI which is frequently nephrotoxic – in a study of 249 dogs, AKI was driven by ischemic/inflammatory conditions (58%) followed by infections (8%, pyelonephritis and leptospirosis being most common), 24% by unknown causes, and only 6% by nephrotoxicity. It’s not that dogs don’t encounter nephrotoxins – they do, simply by sharing environment with humans, where substances like ethylene glycol, NSAIDs (especially for-human-use COX-1 inhibitors), vitamin D (used as rodenticide), and of course grapes can be within reach. The toxicity of grapes is suspected to be caused by tartaric acid. The fact that nephrotoxic AKI is less common might be a credit to hypervigilant dog owners. The veterinary world has adopted IRIS AKI staging, which predicts mortality just as well as KDIGO staging in humans, with short-term death rates of 35-60%. Interestingly, fractional clearance of sodium (FCNa) is considered an accurate prognostic marker in dogs, unlike in humans where exactly the same metric, fractional excretion of sodium (FeNa), has been largely abandoned due to its limitations. In dogs, seeing FCNa normalize from >20% down to <1% is a reliable sign of tubular recovery and a predictor of survival. Oliguria is a poor prognostic sign, while improving eGFR over time is, not surprisingly, associated with better outcomes.
Chronic kidney disease (CKD) occurs in dogs with a prevalence between 0.3-3%. Canine CKD mirrors human disease, and is staged using IRIS CKD classification based on serum creatinine cutoffs, with sub-stages for both proteinuria (urine protein to creatinine ratio >0.5) and hypertension (>150 mmHg). The survival curves are uncannily similar to humans, showing stage-dependent progression with median survival of 15, 11, and 2 months for stages 2, 3, and 4, respectively. Prognostic indicators in humans also seem to show similar relevance and predictive power in dogs. Elevated FGF-23 predicts mortality with hazard ratio (HR) 2.6, as does hyperphosphatemia (HR 3.2), proteinuria (HR 3.2), and high calcium-phosphorus product (HR 4.0). In fact, the entire CKD-MBD paradigm we’ve developed in humans applies seamlessly to dogs, which means the pathophysiology is deeply conserved across species.
Given how much we love our dogs and that many dog owners want the best of human healthcare to be available to their pets, it’s not surprising that dialysis for dogs has become more widely available in recent years. Survival rates for dogs with AKI on intermittent HD are similar to humans at around 50%, and dogs with leptospirosis fare better. Hemodialysis can be administered with either intermittent or continuous modalities through a semi-permanent venous catheter similar to humans. Peritoneal dialysis has lower success rates. Systemic or regional anticoagulation is required for hemodialysis. Dogs generally are not sedated unless they are on continuous renal replacement therapy. Given the cost and small number of centers that offer dog dialysis, attempts have been made to predict which animals might benefit the most, to conserve limited resources for dogs with a better prognosis. For the most part, dialysis is reserved for AKI, though some centers do offer it for end-stage kidney disease (ESKD). Despite paving the experimental path for human transplants, kidney transplantation in dogs remains largely unsuccessful and is not a routine clinical offering. The obstacles are rapid rejection – a biological barrier that has proven difficult to overcome – and the challenge of balancing immunosuppression with dogs’ high exposure to bacterial, fungal, and protozoal infections as they dig and explore their environment.
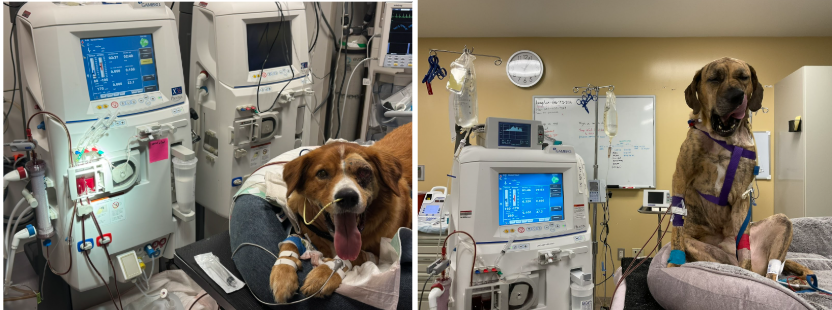
Dogs undergoing hemodialysis, pictures courtesy Dr. Autumn Harris
The close relationship between dogs and humans has come a long way, as has our understanding of human and canine kidney disease. Without a doubt the love for our canine companions as well as our determination to cure kidney disease will continue to drive us to do more until better diagnostic and therapeutic interventions have been realized for both of our species.
COMMENTARY BY AUTUMN HARRIS:
Cats, Kidneys, and the Art of Surviving on Almost No Water
Team 2: Cats
“Cat said, ‘I am not a friend, and I am not a servant. I am the Cat who walks by himself, and I wish to come into your cave.’” – Rudyard Kipling
Humans have alternately worshipped cats as domestic goddesses in ancient Egypt and persecuted them as witches’ familiars in medieval Europe, and it is still debatable whether cats are truly domesticated pets. What we can agree on is that cats are undeniable CKD Champions. Let’s explore how our beloved desert carnivores must be enticed with water fountains and fish-flavored drinks to manage their kidney disease, and how remarkably well they fare after kidney transplantation!

Copyright: @NephCatness, photo courtesy of the owner
For centuries, the taming of the cat was credited to ancient Egyptians. This was challenged by a discovery of an almost 10,000-year-old grave in Cyprus with a human and a pet cat buried together. Since cats were not native to the island, this one must have been brought in by boat from the Middle East. In fact, DNA studies show that the majority of the world’s domestic cats owe their origins to Felis sylvestris lybica from Mesopotamia. The cat-human relationship probably formed due to the cat’s superior skills in hunting mice and rats. Egyptian art and writings from around 3600 years ago portrayed cats as treasured companions. By the first millennium BCE, the goddess Basted had been transformed from lioness into a domestic cat embodying fertility and protection of the home. Fast forward through the dark period in European history when cats were persecuted as witches’ familiars, and today they’ve reclaimed an elevated status in our homes – adored if not worshipped. Some one-third of American families own at least one cat and, contrary to a popular belief, men and women are equally likely to be cat owners. The domestic cat is a single species, Felis catus. Unlike dogs, bred for specific tasks over thousands of years, cat breeding became popular only in the late 19th century, following the success of the first Cat Show in London in 1871. Cats remain of approximately the same size and are mostly bred for looks (color and texture of their coats) and perhaps personality. Their main role nowadays is being pets.
Cats share some diseases in common with their human owners. They are particularly susceptible to kidney disease, with an overall prevalence of 20%. It was estimated that a whopping 80% of geriatric cats have CKD. There are physiologic reasons for this. By origin, cats are desert carnivores who get most of their water from prey. Their urine is very concentrated with a specific gravity usually greater than 1.035, and urine osmolality varying from 500 to 3000 mOsm/kg. Cats have a high percentage of nephrons with long loops of Henle. In addition, their collecting ducts boast a large number of transporters for urea cycling, responsible for the elevated urine osmolality. They have an exquisite water conservation system, the best of any domesticated animal. This, however, can lead to long-term problems for our feline friends. Unlike dogs who are prone to proteinuric diseases, cats are susceptible to interstitial diseases as well as kidney stones, calcium oxalate in particular. Associated with crystalluria and stones, urinary tract infections are also common in cats.
For kidney stones, prevention is paramount. New cat owners need to be instructed in ways to encourage their cats to drink, as cats have a natural dislike for water. This may include water fountains (running water is more enticing to cats), wet rather than dry food, and flavored products such as Hydra Care, proven to increase hydration parameters. A recent study demonstrated that higher viscosity liquids (like 1% methylcellulose solution) can increase cats’ fluid intake by 25%, lowering urine osmolarity, calcium excretion, and calcium to oxalate ratio. Treatment of obstructive stones involves parenteral fluids (intravenous or subcutaneous), ureteral muscle relaxants, pain relief, antibiotics and, in dire circumstances, surgery. Long-term management includes special diets and monitoring for CKD and hypertension.
CKD can be seen in cats of all ages and, as mentioned previously, the risk increases with age. An inciting event is rarely identified. If biopsies are performed at all, the most common finding is interstitial nephritis with mononuclear cell infiltration, tubulointerstitial fibrosis and glomerular obsolescence. Approximately 20% of cats will have hypertension at the time of CKD diagnosis, and another 20% will become hypertensive thereafter. In studies of prognostic factors in cats with CKD – age, elevated serum creatinine, and proteinuria all reduce survival. Just like in humans, CKD affects cats’ health-related quality of life. Measured with a validated veterinary tool, quality of life in cats is decreased by the presence of anemia, low appetite, and constipation.
Besides interstitial disease, there are other forms of CKD in cats. For example, cats can have a hereditary polycystic kidney disease with a mutation in the PKD1 gene just like in humans. With a hefty 38% prevalence, Persians are the most affected breed in the US. Kidney disease is also seen in leukemia, lymphoma, and Feline Immunodeficiency Virus infection. Abyssinians and Siamese can have a form of familial AA amyloidosis affecting kidneys and other organs.
Reported prevalence of diabetes in cats is 0.58%. Like in humans, it can develop either from immune destruction of insulin-producing cells or from insulin resistance. Unlike in humans, overt diabetic nephropathy is not typically seen in cats. In a retrospective study of post-mortem tissue from diabetic cats, there were ultrastructural changes of diabetic nephropathy at the time of death in the absence of clinically overt nephropathy. The authors postulated that cats must not survive to clinical nephropathy.
Treatment of CKD in cats consists primarily of a low protein and low phosphate diet as well as hydration. In cases of severe anorexia or refusal to drink, an esophagostomy tube (e-tube) can be placed for fluid and nutritional supplementation. Several medications are FDA-approved for nausea, vomiting and anorexia in cats, from familiar-to-human doctors names like ondansetron and mirtazapine to veterinary use-only antiemetic maropitant (a selective neurokinin-1 receptor antagonist) and appetite stimulant capromorelin (a ghrelin receptor agonist). The latter is currently being studied as a way to promote lean weight gain in the setting of human frailty. Treatment of hypertension is with ACE inhibitors/ARBs or calcium channel blockers. Anemia occurs in 30-65% of cats with CKD. Although costly, darbepoetin is an effective treatment. Molidustat, a hypoxia-inducible factor propyl hydroxylase inhibitor, is conditionally FDA-approved for cats. Phosphate binders are used in cats with phosphate levels greater than 6.5 mg/dL.
Hemodialysis can be done at some specialized centers, but is in general limited to treatment of acute kidney injury and intoxications. Renal transplantation is quite successful in cats, and several centers in the US are performing feline kidney transplants. Transplanted cats are maintained on prednisone and cyclosporine with good survival rates. The expense to owners is significant. Donor kidneys are obtained from rescue cats; the donors must be adopted by the recipient’s owners, and they have a normal life expectancy.

Cats participating in CKD clinical trials, pictures courtesy Dr. Autumn Harris
An exciting treatment for CKD in cats is an oral agent that binds uremic precursors in the gut. Over 100 uremic toxins have been identified to date. Two in particular – indoxyl sulfate (IDS) and p-cresol sulfate (pCS) – are gut-derived toxins that not only induce uremic symptoms but also promote interstitial fibrosis, accelerating the progression of CKD. Renaltec is an oral carbon-based adsorbent that is claimed to trap precursors in the gut before they are converted to IDS and pCS in the liver. Renaltec was studied in 12 cats with experimental CKD and was shown to significantly reduce levels of these toxins. Only one cat progressed from CKD stage 2 to stage 3 during the study and another cat had an increase in creatinine within the same stage. Weight loss was curbed during the treatment period. The results are preliminary and are based on very few cats without a parallel control group, but could this therapy one day become applicable to humans?
Acute kidney injury in cats is less common than CKD. Notable causes are trauma, obstruction by stones, volume depletion from GI disorders, or toxins like ethylene glycol, human-use NSAIDs like ibuprofen (COX-2 inhibitors are much safer for cats), antibiotics, vitamin D and all parts of true lilies from genera Lilium and Hemerocallis. Lily ingestion causes severe renal tubular toxicity in cats, often with an early polyuric phase of AKI. It has been proposed that the toxin is a unique product of feline metabolism, because direct application of lily extract to cultured feline renal tubular cells does not reproduce damage. In all cases of AKI associated with volume depletion, judicious fluid resuscitation is paramount. Subcutaneous fluid administration is more convenient than intravenous. In the cases of toxin exposure, time to treatment counts. Admission to a critical care unit for fluids and (rarely) hemodialysis may be necessary. Prognosis for survival depends on creatinine and urine output at presentation.
In summary, there are many parallels and many differences in kidney disease between cats and humans. There are things we have learned from studying our feline friends that may impact human CKD. Likewise, our cats are beneficiaries of modern nephrology knowledge. There’s plenty of room for collaboration between veterinarians and human nephrologists to discover better ways to treat kidney disease.
Conclusion
When we think about nephrology in our canine companions and feline friends, what logically comes to mind is the One Health paradigm – a concept that recognizes the interconnectedness of humans, animals, and our shared environment. Rather than working in isolation, disciplines like veterinary and human medicine, public health, and environmental science can join efforts to achieve optimal health for all. One powerful way to advance this goal is through collaborative clinical trials. Veterinary patients with naturally occurring kidney diseases can provide insights that differ significantly from laboratory-developed rodent models, and human trials have, in turn, benefitted veterinary medicine. Pet owners are often motivated to participate in studies in order to get access to state-of-the-art diagnostics and treatments, financial coverage for tests and medications, and for altruistic benefits. For the list of ongoing trials, check out the American Veterinary Medical Association (AVMA) clinical trials registry at https://veterinaryclinicaltrials.org/. Beyond clinical research, dogs and cats serve as environmental sentinels for infections such as Lyme borreliosis and Leishmaniasis – conditions that impact both animals and humans. By joining forces across disciplines, we can unlock novel perspectives and solutions.
Let’s work together for One Health Nephrology!
– Executive Team Members for this region: Anna Vinnikova @KidneyWars – @kidneywars.bsky.social and Matthew Sparks @Nephro_Sparks – @nephrosparks.bsky.social | Meet the Gamemakers
How to Claim CME and MOC
US-based physicians can earn 1.0 CME credit and 1.0 MOC per region through NKF PERC (detailed instructions here). The CME and MOC activity will expire on June 1, 2026.













Leave a Reply