#NephMadness 2026: POCUS Region
Submit your picks! | @NephMadness | @nephmadness.bsky.social | NephMadness 2026
Selection Committee Member: Mohamed Hassan Kamel
Mohamed Hassan Kamel is an Assistant Professor, nephrologist and intensivist at Stony Brook University. His clinical interests include hemodynamic assessment, resuscitation and de-resuscitation in shock. He is passionate about furthering the use of point-of-care ultrasound in the field of nephrology and ensuring trainees have adequate experience and confidence.
Selection Committee Member: Vandana Niyyar @vandyniyyar
Vandana Dua Niyyar is Professor of Medicine at Emory Nephrology, designated Master Physician at Emory DOM, ASDIN Immediate Past-President and chairs ASN’s TDAT Initiative and KHI’s Device Committee. Dr. Niyyar has expertise in hemodialysis vascular access and ultrasonography and is passionate about promoting multidisciplinary collaborations to optimize access care and processes for dialysis patients.
Writer: Priyal Sakhuja
Priyal Sakhuja is currently a second-year nephrology fellow at Mount Sinai Hospital in New York City. She attended NYITCOM for medical school and completed her internal medicine training at Stony Brook University in Long Island. Her professional interests include renal physiology, dialysis, acid-base medicine, continuous replacement therapies, and critical care medicine. She will be pursuing an additional year of training in critical care at Mount Sinai Hospital.
Competitors for the POCUS Region
Nephrology has always been about interpreting imperfect clues—history, exam, and labs—while the kidneys quietly do their thing. POCUS flips the script by giving us a real-time window into anatomy and physiology at the bedside, letting us “see” what we used to infer. In this bracket, it’s AKI vs ESKD: two heavyweights fighting for the same prize—better decisions, faster.
Team 1: POCUS in AKI
versus
Team 2: POCUS in ESKD

Image generated by Matthew Sparks using ChatGPT at http://chat.openai.com, February 2026. After using the tool to generate the image, Sparks and the NephMadness Executive Team reviewed and take full responsibility for the final graphic image.
While it is safe to assume Henry David Thoreau was not thinking about modern medicine when he wrote “it’s not what you look at that matters, it’s what you see” the idea of seeing (the active process of interpreting) rather than looking (a passive act) is still relevant today. What if there was a cheap, non-invasive tool that would allow one to look beyond the superficial, and to see within a patient, to assess an individual’s physiology in real time? We’re of course talking about none other than the rapidly progressing (and ever shrinking) technology that is Point of Care Ultrasound (POCUS). Although it should be viewed more as an extension than a substitute for physical examination skills and clinical acumen, if one knows what they are looking for and how to interpret the information, it has proven to be a valuable adjunct to traditional practice.
Initially developed based on the principles of sonar in World War 1, the roles of ultrasonography have significantly expanded since its use was adopted in the 1950s as a diagnostic tool primarily for radiologists and obstetricians. Since the late 1980s, POCUS has been increasingly recognized for its utility across a broad range of specialities including emergency medicine, critical care medicine, cardiology, surgery, and obstetrics to name a few, aiding in improved outcomes of procedures, and a decreased time to diagnosis.
While its use may not be as prevalent in nephrology as in other fields, nephrology encompasses a wide range of diseases with a diverse and clinically complex patient population which could make POCUS a valuable player in the game if utilized to its full capabilities. Though it is increasingly recognized as a necessary skill to learn, it is not widely incorporated in medical training, with only 35% of internal medicine residency programs and only 23% of nephrology fellowships having formal training.
So come join us, as we explore how getting a direct window into a patient can help guide our care in two of the specialties most common diseases, acute kidney injury (AKI) versus end stage kidney disease (ESKD).
In this episode, Shreya P. Trivedi, Noah Markewitz, and Jeffrey Kott cover detecting post-renal obstruction in a patient who reported normal urination, using VEXUS to uncover hidden venous congestion and guide further diuresis, distinguishing COPD from volume overload in a dialysis patient using lung ultrasound, and assessing AV fistula maturity at the bedside to potentially avoid placing a temporary dialysis line
POCUS for AKI & Dialysis | Real Cases That Changed Management
COMMENTARY BY ABHILASH KORATALA:
Stop Guessing, Start Scanning – POCUS in AKI
Team 1: POCUS in AKI
AKI workups are bread-and-butter, but POCUS turns guesswork into game film—quickly sorting structure and hemodynamics before the labs even catch up. And when volume status is the plot twist, tools like IVC and VExUS bring the “pumps-pipes-leak” physiology to life, because congestion doesn’t just feel bad, it looks bad.

Copyright: Amirkhans world/ Shutterstock
AKI diagnosis and work-up remains the bread and butter of nephrology, yet in its workup, we rely on imperfect tools such as history, physical exam, and labs. POCUS adds speed and precision, offering bedside insight into structure, obstruction, and volume status to help delineate pre-renal, intrinsic, and post-renal causes.
Obstructive Nephropathy and Beyond
Obstructive Nephropathy accounts for approximately 5-10% of AKIs and in certain cases timely diagnosis can make a difference in a patient’s medical decision making. When evaluating for an obstruction with ultrasound, bladder scanning for distension or post-void suggests lower tract involvement while anechoic dilatation of the renal pelvis and calyces signals back-up of urine proximally, resulting in the classic picture of hydronephrosis. A formal renal ultrasound, which is commonly ordered as part of the workup for acute kidney injury to rule out obstruction, can be both unnecessary, except in specific populations, and costly.
POCUS provides a quick, easy and cost effective means to detect obstruction. One study evaluating use of POCUS in patients with AKI admitted to an acute medical unit found POCUS to be nearly as accurate as a radiology ultrasound (90% sensitive/100% specific) at identifying obstruction, and about 20 hours faster! Another single-center study, evaluating use of POCUS compared to CT or radiology ultrasound in patients with AKI, found a high sensitivity, but slightly lower specificity compared to the prior study; this decreased specificity was attributed to urinary tract decompression with foley catheter prior to formal imaging. However, when risk stratified to moderate to high risk for obstruction, POCUS again became almost as accurate as CT or radiology ultrasound.
POCUS can potentially go one step further and shed light on the cause of obstruction, such as the presence of kidney/bladder stones. Calcified stones appear highly echogenic on sonography, resulting in an “acoustic shadow” behind the stone. Visualization of ureteral stones remains more challenging and CT remains the gold standard. However, when ureteral stones cause hydronephrosis, POCUS would still provide valuable insight.
Now, as with every test, POCUS for detection of obstructive AKI is not perfect. False negatives may occur in early obstruction, volume depletion, or retroperitoneal fibrosis/tumors due to lack of collecting system dilation. Additionally, the absence of hydronephrosis does not eliminate obstructive uropathy; patients with acute bladder outlet obstruction can have AKI without significant hydronephrosis. In this scenario, POCUS can be helpful to assess bladder wall thickness and trabeculations, pointing to signs of chronic obstruction.
Hydronephrosis is one of the more easily identifiable manifestations of renal ultrasound; however, there is much more information one can obtain from POCUS, especially if trying to differentiate between acute vs chronic disease. Kidney size, cortical thickness, and echogenicity offer additional data points that could help clue into various etiologies of kidney disease within a clinical context. Furthermore, smaller kidneys certainly indicate a sign of chronicity, and larger kidneys, especially if unilateral, may identify acute issues such as pyelonephritis. While increased echogenicity is typically thought of as chronic, it can be seen in AKI when caused by pathologies such as acute interstitial nephritis or glomerulonephritis.
While POCUS assessment gives us a host of information on the anatomical features of the kidney, it can also be taken one step further in its utility for kidney biopsies, which are often performed in the outpatient setting. Patients with AKI, hematuria, and proteinuria are referred for biopsies to obtain tissue diagnosis. Data have shown that real-time ultrasound guided kidney biopsies have similar success and complication rates to CT-guided biopsies. As with everything, it is integral to have an experienced operator performing the ultrasound to ensure proper angling to obtain optimal specimen (>20 glomeruli) with minimal bleeding.
POCUS for Volume Assessment in AKI
Determination of volume status, a core part of a nephrologist’s expertise, remains incredibly variable, with physical exam and tracked fluid balance not being reliable. Two of the most frequent reasons for hospital admission, sepsis and congestive heart failure, are associated with hemodynamic shifts and it is critical to assess volume status correctly to optimize management of these patients. These patients also receive some form of IV fluids (including as diluents for medications) during their hospital stay. So let’s dive further to determine how POCUS may provide some insights into a direct evaluation of volume status.
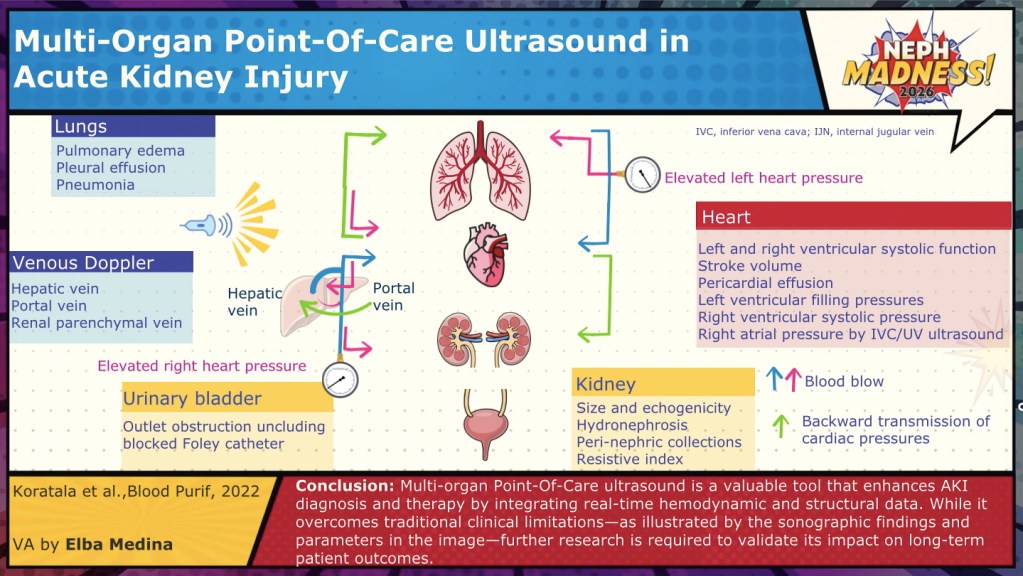
There are many ways to extend our traditional means of volume status assessment with POCUS. A comprehensive POCUS volume assessment will encompass some if not all of the following to assess for the pumps-pipes-leak theory:
- Vascular Ultrasound – Allows for assessment of central venous pressure and venous congestion.
- Cardiovascular Ultrasound– Allows for assessment of both left- and right-sided cardiac function and pressures, as well as valvular pathology and pericardial effusions. Can help identify volume responsiveness, low cardiac output, and elevated right– or left-sided filling pressures. Identification of abnormal cardiac function/pressures can help further explain findings in other systems evaluated.
- Lung Ultrasound (LUS) – Allows for assessment of intraparenchymal and intrapleural fluid.
- Abdominal Ultrasound– Allows for assessment of presence of ascites.
However, most discussions regarding POCUS and volume assessment deal specifically with evaluation of the vascular response to volume and pressure, beginning with the Inferior Vena Cava (IVC) ultrasound. Though IVC US is commonly and incorrectly used as a marker of fluid responsiveness, IVC US tends to correlate better with central venous pressure, which has long been noted as a poor surrogate for fluid responsiveness. After all, a small and variable IVC (i.e. low CVP) is normal physiology. There are some data for use of IVC ultrasound to guide volume resuscitation in critically ill patients with AKI, however its limitations in interpretation should be noted when performing. There are other POCUS parameters of volume responsiveness, but these are yet to be technically validated in critically ill patients, and may require more operator comfort level, extending beyond the point of care. Still, the underlying physiology can lend itself to extrapolation as to which patients could benefit from additional volume resuscitation.
So how about the other end of the spectrum? We’ve talked briefly about POCUS as a means of volume assessment with respect to cardiorenal syndrome previously, however we felt like a deeper dive was worth it. AKI is common in the ICU in the context of complex shock states, heart failure, and volume overload. Right at the crux of AKI in critically ill patients is the correlation of CVP and AKI, as an increase in CVP leads to both a decrease in renal perfusion pressure independent of mean arterial pressure (MAP) and an increase in renal interstitial hydrostatic pressures. IVC US can be useful at determining presence of an elevated CVP, and has some data for its use in differentiating between volume overload and hepatorenal syndrome.
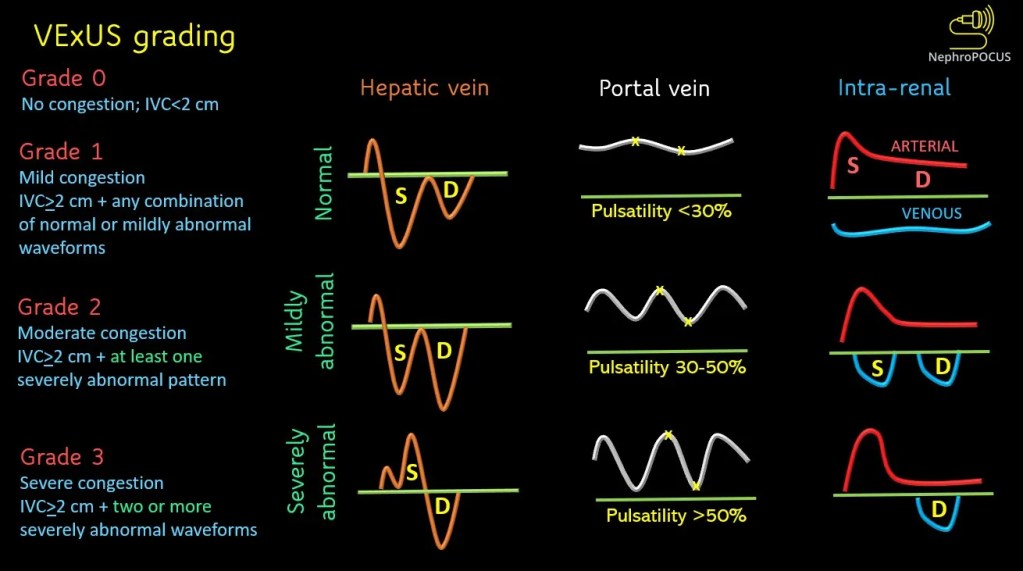
But if IVC US is the appetizer, look no further than the VExUS (Venous Excess Ultrasound) score as the main course. VExUS builds off of IVC US by assessing doppler flow in hepatic, portal, and renal veins. It stems from the idea that we can use US to determine when venous congestion becomes so severe it alters venous flow within organs such as the liver and kidney. A good primer on performing and interpreting VEXUS can be found here.
VExUS caught fire in the critical care world after its initial study, which demonstrated that a score > 2 retrospectively better quantified systemic venous congestion after cardiac surgery and had a higher likelihood ratio for detecting severe congestion leading to AKI than CVP, or the individual components of VExUS. Since its introduction, meta-analyses have further solidified the strong association between high VExUS scores and AKI in the cardiac surgery population and to a lesser degree, the non-cardiac surgery population. Another recent study demonstrated that VExUS >3 was associated with worsening renal function, decreased natriuresis/diuretic resistance, and mortality. In some ways these malassociations are unsurprising–sicker patients will tend to develop volume overload, AKI, and are naturally going to be at a higher risk for mortality.
So does identifying a risk factor for acute kidney injury change outcomes? A single-center prospective study in a medical ICU evaluated VExUS in patients with presumed cardiorenal syndrome. Improvement in daily VExUS scoring tracked well with both AKI staging and fluid balance but not with traditional markers of volume overload such as CVP, left ventricular, or right ventricular function. Another single-center study performed VExUS at 0 and 48 hours on critically ill patients with AKI. Those with reduced VExUS at 48 hours received more loop diuretics (for an overall negative fluid balance), more kidney replacement therapy (KRT) free days, and improvement in KDIGO AKI staging. Though this was relatively small, not protocolized, and uncontrolled, it certainly demonstrates that serial VExUS can be a beneficial tool which affects practice patterns. Furthermore, a recent study evaluated serial use of a modified VExUS (mVExUS, excluding renal doppler) on those admitted to a cardiovascular ICU to evaluate mortality outcomes. An early decrease in mVExUS score was associated with reduced in-hospital mortality, alongside favorable laboratory and clinical markers of decongestion (such as weight, urine output, creatinine) and outperformed other markers of congestion (lung ultrasound and diastology) in early detection of decongestion.
So while in some ways these studies tell us what we already know (congestion is bad), they also give us a validated tool that can be used in AKI serially to prognosticate and evaluate how well we are actually decongesting individuals. Where does VExUS fall short? Well, from an evidence-based standpoint, it is largely validated in a critically ill/cardiac population with AKI, so generalizability to general AKI remains limited (though the principle of congestion leading to AKI is well documented). From a technical standpoint, as with all US, operator variability and comfort level certainly remains important. It also does not differentiate between volume and pressure overload, meaning that addressing volume alone may not be definitive. Finally the three veins evaluated have their own limitations:
- Hepatic Venous Doppler relies on correlation with systole and diastole, making the need for simultaneous EKG waveforms important. It is also influenced by arrhythmias.
- Portal Venous Doppler can be misinterpreted in portal hypertension or individuals with low body mass index.
- Intrarenal Venous Dopplers can be difficult to obtain and data are limited in Chronic Kidney Disease/Kidney Transplant.
Taken together, POCUS enables nephrologists to integrate their traditional assessments with real-time structural findings and physiologic information that proves a more complete and actionable picture of AKI at the bedside.
Team 2: POCUS in ESKD
In ESKD, the daily grind is volume control and vascular access—two arenas where “close enough” can come at a high price. Can POCUS tip the scales by bringing physiology to the forefront of fluid assessment and sharpening our approach to access maturation, guided cannulation, and early complication detection? In a population where we need better outcomes, maybe seeing more clearly is the ultimate competitive advantage.

Copyright: Rudmer Zwerver/ Shutterstock
Let us turn our attention to the other end of the nephrology spectrum: end-stage kidney disease. More than 815,000 individuals in the United States live with ESKD, with many more worldwide. Mortality in individuals with ESKD remains high, driven by cardiovascular mortality thought to be in part due to difficulty managing volume overload. Vascular access is another area of complexity within ESKD, from temporary dialysis access placement, to arteriovenous fistulas (AVFs) failing to mature, to difficult cannulation, to complications of vascular access. So let’s break down how POCUS can help us tackle these issues in a patient population where we certainly can provide better care.
POCUS for Volume in ESKD
Establishing dry weight is central to optimizing dialysis therapy in hemodialysis (HD) patients yet remains difficult without universally accepted measures of volume status. In a survey of 173 UK dialysis centers, 78% had no policy for fluid assessment and 44% did not routinely assess it. Blood pressure and body weight are the most common surrogates, but are both influenced by numerous variables. Additional metrics such as bioimpedance, biochemical markers (BNP), and relative plasma volume monitors have shown promise, but have failed to demonstrate a significant clinical benefit. So how has POCUS and the real time data it provides fared in furthering our ability to conquer this complex situation? Let’s do a deeper dive.
It is important to note that all VEXUS studies discussed above did not include individuals with ESKD, and excluded those with AKI on KRT. Still there is some literature on VEXUS in the dialysis population. The ACUVEX study evaluated hemodialysis patients above their target dry weight, and performed VEXUS pre-, during, and post-dialysis to explore the relationship between fluid status and ultrafiltration.Though it was a relatively small sample size (only 15% of enrolled had a VExUS score >1), removal of fluid in these individuals did improve their VExUS score. However, all patients had echocardiographic evidence of right and left ventricular systolic impairment, which calls into question whether the VExUS findings were related to general volume overload or baseline cardiac impairment. Another case report did demonstrate how VExUS can be used to guide ultrafiltration in a hemodialysis patient with abnormal liver function tests with improvement in congestion. Still, dialysis patients represent a different physiology compared to the general population given changes in venous compliance and depending on dialysis modality and changes in cardiac function in the setting of arteriovenous fistula or graft. While VExUS could certainly be a key to furthering our ability to keep our patients euvolemic, further research is currently needed.
So what about other ultrasound parameters? As discussed above, IVC US correlates well with CVP. Although insightful for intravascular volume, IVC assessment is less sensitive for overall fluid overload, especially given that it is affected by tricuspid regurgitation, pulmonary hypertension, and atrial fibrillation. Because of some of these pitfalls, its utility in dialysis patients is scarce, with studies largely being performed in the critically ill dialysis population, which is not generalizable to the outpatient dialysis population. Lung congestion as evaluated by LUS is a risk factor for mortality in HD. After this association, studies showed that LUS-guided ultrafiltration can lower both dry weight and ambulatory blood pressure, as well as improve intracardiac pressures and dimension. These findings culminated in the LUST Trial, a multi-center randomized trial, which used a LUS-guided ultrafiltration protocol versus standard of care to assess for a change in major cardiovascular events (MACE). Relief of lung congestion was achieved in 78% of patients in the intervention arm compared with 56% in the control arm, but this did not translate into fewer MACE or reduced mortality compared to standard care. An observational extended analysis of the LUST trial was published in 2023, which did show a statistically significant decrease in MACE in males, but no similar effect in females which was attributed to males having a higher fluid volume and cardiovascular risk. Although valuable for volume status by detecting extravascular lung water, B-lines seen on LUS can also represent other lung pathologies such as emphysema or fibrosis, which could confound findings.
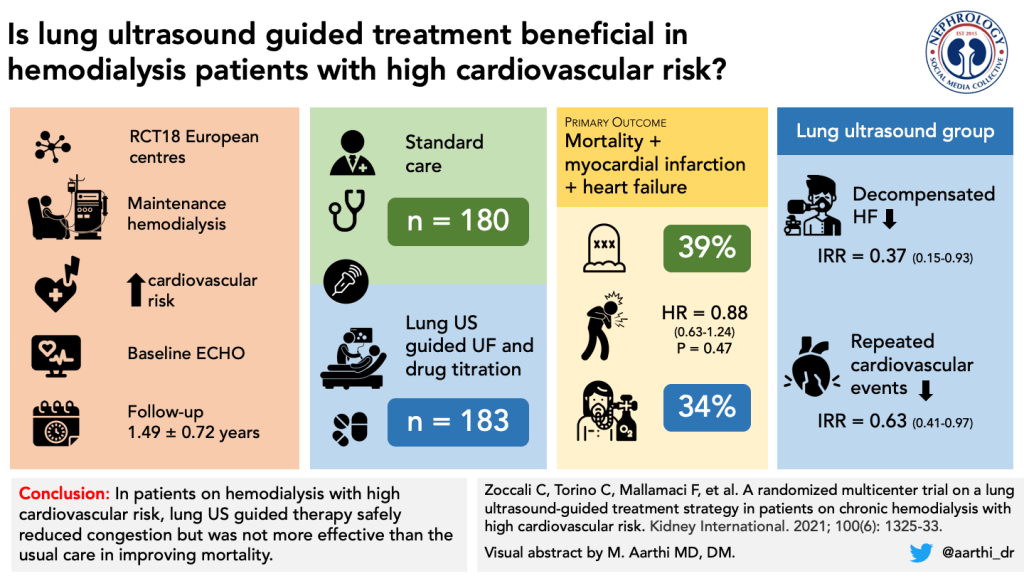
So while the data are still out there on POCUS’ benefits for maximizing ultrafiltration, can it help determine those who fall on the other end of the spectrum? Intradialytic hypotension (IDH), which has multiple definitions across the literature, is a common issue during dialysis that can be associated with higher all-cause mortality. There is some literature that IVC US may predict IDH in the outpatient dialysis population; however, further research is needed.
POCUS for Vascular Access
POCUS has also found a role in obtaining and evaluating vascular access. With respect to central venous dialysis access, it has been well documented that use of ultrasound improves safety and outcomes, to the point where it is now standard of care and recommended for insertion of every central venous catheter. But POCUS has a significant role in arteriovenous (AV) vascular access as well. It allows vessel mapping, assessment of maturation, real-time cannulation, and detection of complications such as infiltration or hematoma. The duplex ultrasound (DUS) has been integral for diagnostic imaging since the 1970s and with the introduction of color Doppler in 1978, it has been the gold standard diagnostic technique and modality of choice to assess for vascular pathologies. From a perioperative surgical standpoint, POCUS has been shown to lower primary failure rates, and reduce fistula abandonment.
Traditionally, the Rule of 6s (flow volume greater than 600 mL/min, a vein diameter greater than 6 mm, and a vein depth less than 6 mm from the skin) was the gold standard for assessing for AVF maturation, which on average takes 6-12 weeks with not insignificant failure rates. KDOQI has since moved away from the rule of 6s, leaving it up to clinical experience, and introducing language that demonstrates how POCUS can potentially benefit our management of AV dialysis access.
First, let’s examine if POCUS can help us speed up our time to first use. One study had a nephrology fellow prospectively perform POCUS of an AVF within four weeks of creation vs a retrospective standard of care cohort, with initiation of cannulation when the AVF had a diameter >6mm, was <6mm deep, and >6mm long. The POCUS cohort had significantly earlier cannulation (35.5 vs 63.3 days), shorter central venous catheter duration, and fewer infections without any difference in complications. While the literature on this subject remains thin, this study certainly provides a solid foundation for future research that POCUS-driven evaluation could lead to earlier time to first cannulation.
So how about other fistula-related outcomes? The data have been mixed on whether POCUS-guided cannulation reduces complications, with one study finding no significant difference in infiltration, while others found a reduction in complications such as oozing, hematoma and infiltrations. Direct visualization of the AVF/AVG may provide benefit as appropriate needle positioning was found to be suboptimal in 80% of traditional needle placements, demonstrating a potential nidus for acute or chronic vessel damage. Still further research is needed to tease out whether POCUS will ultimately improve longevity/reduce complications in arteriovenous access. What POCUS definitely achieves with respect to vascular access is accessible and teachable and leads to an improvement in satisfaction among both patients and staff, despite many of these studies showing that POCUS increased time for cannulation.
Peritoneal dialysis, another common form of renal replacement therapy, requires pre-procedural assessment prior to placing a peritoneal catheter. Ultrasound, once again, proves to be a valuable tool. It allows for careful assessment of the abdominal wall, peritoneal cavity, and vascular structures such as the inferior epigastric artery to ensure optimal placement and avoid any vascular injury. Ultrasound can also detect hernias, which would be a contraindication to percutaneous placement.
POCUS in Kidney Transplant
While POCUS has begun to be adopted in AKI and dialysis patients, its transition to post-kidney transplant patients has not been well studied. Given that some of the principles we have discussed could easily be translatable to a post-kidney transplant patient, it is important to note some important distinctions. Transplant kidney US is an important part of the post-kidney transplant workup, including evaluation of obstruction, perinephric fluid collections, or vascular complications such as transplant renal rrtery stenosis. While these all could, in theory, be evaluated by an experienced operator, transplant kidneys play by different rules–hydronephrosis happens and it’s not always pathologic. Until more data arises regarding its use, a formal transplant renal ultrasound should be ordered, especially given the scarcity of organs available.
Conclusion
POCUS offers nephrologists something we have never had before– a real-time window into our patients that sharpens our clinical judgement. But for all of its promise, two principles remains paramount. First the tool is only as good as its operator. Image acquisition and interpretation take skill, practice, and honest self assessment, so while we have outlined the many uses for POCUS in our patient population, the safest approach is simple: If you’re not confident in what you are seeing, seek other means of getting that information, whether it be through a colleague or a formal study. Second, POCUS is a diagnostic tool, and what one does with the information is more important than the tool itself. Still, as POCUS is just beginning to reshape how we care for patients, the final call is yours: Which contender, POCUS in AKI or ESKD, earns a place in the next round?
– Executive Team Members for this region: Jeffrey Kott @jrkott27 – @jrkott27.bsky.social and Anna Burgner @anna_burgner – @annaburgner.bsky.social | Meet the Gamemakers
How to Claim CME and MOC
US-based physicians can earn 1.0 CME credit and 1.0 MOC per region through NKF PERC (detailed instructions here). The CME and MOC activity will expire on June 1, 2026.








Leave a Reply