#NephMadness 2026: POCUS – Stop Guessing, Start Scanning
Submit your picks! | @NephMadness | @nephmadness.bsky.social | NephMadness 2026
Abhilash Koratala @NephroP
Abhilash Koratala, MD, FASN, FNKF, is a nephrologist–intensivist and Associate Professor at the Medical College of Wisconsin. A global leader in POCUS and Doppler-based hemodynamics, he has delivered 150+ national and international invited lectures and was named ASN Distinguished Educator in 2024.
Competitors for the POCUS Region
Team 1: POCUS in AKI
vs
Team 2: POCUS in ESKD
Image generated by Matthew Sparks using ChatGPT at http://chat.openai.com, February 2026. After using the tool to generate the image, Sparks and the NephMadness Executive Team reviewed and take full responsibility for the final graphic image.
When nephrology is consulted for acute kidney injury (AKI), everyone assumes we’re the volume whisperers. Hypovolemic? Congested? “You guys can tell, right?” But if we are honest with ourselves, much of what we rely on is far from precise. The classic physical exam signs lack sensitivity. Intake-output charts are imperfect. Daily weights vary with timing and technique. Vital signs offer only fragments of a much larger hemodynamic story. So we label someone “dry” or “wet,” adjust fluids or diuretics, and hope we are right. Sometimes we are. Sometimes we are confidently wrong. That is where point-of-care ultrasound (POCUS) changes the game. Instead of inferring from indirect clues, we can actually see the story unfold.
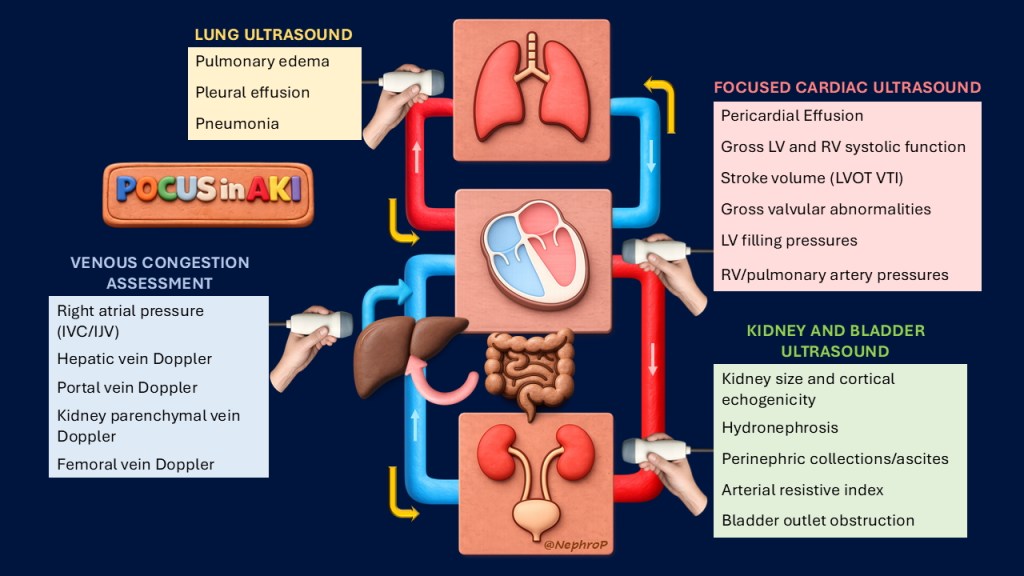
Many still equate nephrologist-performed POCUS in AKI with kidney ultrasound alone. And in fairness, by the time we are consulted, a renal ultrasound has often already been done to rule out obstruction. But that does not make it irrelevant. It identifies hydronephrosis and structural abnormalities, and when baseline creatinine is unknown, renal size and cortical thickness help assess chronicity. Kidney size may also influence biopsy decisions. When interpreting kidney ultrasound, a structured approach prevents oversight; the SECONDS checklist exists for a reason. And the evaluation should not stop at the kidneys. The bladder deserves attention too. A Foley catheter does not exclude urinary obstruction. Malposition or blockage can still occur, and a quick bladder POCUS can prevent premature assumptions about the cause of anuria.
AKI patients often receive empiric fluids almost reflexively. But hemodynamic AKI is not synonymous with hypovolemia. While POCUS cannot directly evaluate renal autoregulation or reliably detect microcirculatory abnormalities, it provides a real-time window into the macrocirculatory forces that influence kidney perfusion. A focused cardiac exam lets us assess gross ventricular function, chamber size, and pericardial effusion at the bedside. In experienced hands, Doppler measurement of LVOT VTI provides a practical estimate of stroke volume and forward flow. And that matters. A small, collapsible IVC does not necessarily indicate volume depletion. It may reflect euvolemia or even a high cardiac output state, such as the hyperdynamic circulation seen in cirrhosis. If we do not assess forward flow, we risk mislabeling the physiology. Consider cirrhosis. A small IVC should not reflexively prompt albumin administration. In a high output state, once Terlipressin raises afterload, this additional preload may be enough to tip the balance toward pulmonary edema. Why take that risk without understanding the full hemodynamic picture? At the same time, a normal mean arterial pressure does not guarantee adequate renal perfusion. Renal perfusion pressure depends on the difference between arterial and venous pressures. If central venous pressure is elevated, renal outflow is impaired. Venous congestion alone can drive kidney dysfunction, even when the forward flow is preserved.
Pulmonary congestion is another piece of the puzzle, especially when deciding whether to give fluids. If the lungs are already wet, more fluid may cause harm rather than help. The challenge is that many hospitalized patients have silent congestion, especially when their activity level is far below baseline. This is where lung ultrasound shines, but also where nuance matters. B-lines are not synonymous with cardiogenic pulmonary edema. They can also appear in acute respiratory distress syndrome, pneumonia, or interstitial lung disease. So, if lung ultrasound shows congestion and you are unsure whether it is cardiogenic or pneumogenic, cardiac Doppler can help. By assessing transmitral inflow and mitral annular velocities, it is possible to estimate left ventricular filling pressures and better understand whether elevated left-sided pressures may be contributing to the picture. In cirrhosis, this becomes even more relevant. Emerging data suggest that Doppler-derived diastolic parameters may predict non-responsiveness to Terlipressin in hepatorenal syndrome. In other words, careful hemodynamic phenotyping is not academic. It may change who benefits from therapy.
Coming back to the IVC, what do we do if POCUS suggests elevated central venous pressure? Holding fluids and/or giving diuretics is reasonable. But what next? This is where venous excess Doppler ultrasound, or VExUS, adds depth. It helps determine whether that elevated pressure is actually being transmitted to the organs, in other words, whether the kidneys are truly “feeling the congestion”. By showing real time flow changes and grading their severity, VExUS shifts the assessment from assumption to objective confirmation. It also allows dynamic monitoring. As decongestive therapy takes effect, venous waveforms change, offering visible proof that congestion is improving. Few bedside tools provide that kind of immediate, intuitive feedback. In AKI, especially in cardiorenal syndrome, that feedback can make a huge difference.
At the end of the day, everything comes back to the hemodynamic circuit. Kidney ultrasound is foundational. It rules out obstruction and gives us insight into chronicity. But most real decisions in AKI revolve around the balance between forward flow, venous pressures, and extravascular fluid. Muddy brown casts do not give us a free pass. They do not shield the kidney from ongoing hemodynamic injury, and they certainly do not eliminate the need to assess the circulation while we provide supportive care. And let’s be honest. As nephrologists, we do not want to drift into irrelevance in the ICU by defaulting to “fluid removal per ICU team” simply because we are not comfortable assessing hemodynamics ourselves.
So, in the POCUS in AKI region of NephMadness, this is not about gadget enthusiasm. It’s about sharpening how we think. It’s about moving from assumption to physiology we can actually see. And for a specialty that prides itself on speaking the language of physiology, what could be more fitting than that?
This year, POCUS is not just in the conversation. It is coming for the title.
– Guest Post written by Abhilash Koratala
As with all content on the AJKD Blog, the opinions expressed are those of the author of each post and are not necessarily shared or endorsed by the AJKD Blog, AJKD, the National Kidney Foundation, Elsevier, or any other entity unless explicitly stated.
Click to read the POCUS Region
Submit your picks! | @NephMadness | @nephmadness.bsky.social | NephMadness 2026



Leave a Reply