#NephMadness 2026: AI Region
Submit your picks! | @NephMadness | @nephmadness.bsky.social | NephMadness 2026
Selection Committee Member: Lili Chan @LilCh4n
Dr. Lili Chan is Associate Professor of Medicine in the Department of Medicine, Barbara T. Murphy Division of Nephrology, and the Windreich Department of Artificial Intelligence and Human health at the Icahn School of Medicine at Mount Sinai located in New York City. Dr. Chan cares for patients with a variety of renal conditions ranging from acute kidney injury to end stage kidney disease. In addition to patient care, she is a NIH funded researcher with a focus on examining and identifying methods, including the use of machine learning and natural language processing, to improve outcomes in patients with kidney disease.
Selection Committee Member: Kuang-Yu Jen @KidneyPath
At NC State University, Dr. Autumn Harris investigates how kidneys regulate acid-base balance and what drives kidney disease progression. Her clinical trials seek earlier diagnosis and better treatments for chronic kidney disease in dogs and cats—advancing both veterinary care and comparative nephrology.
Writer: Neil Evans
Neil Evans is a nephrology fellow at UC Davis. Previously a hospitalist, his professional interests lie at the intersection of technology and clinical practice. He has developed patented machine learning tools for diagnostic support and is dedicated to exploring how integrated data systems can improve patient outcomes and medical education.
Writer: Aditya Yelamanchi
Aditya Yelamanchi is a first-year nephrology fellow at UC Davis in Sacramento, California. He completed his medical degree at the Chicago College of Osteopathic Medicine and internal medicine training at UChicago Northshore in Evanston, IL. His professional interests include AI, interventional nephrology and general nephrology. This is his first work in artificial intelligence but has been involved at ASN previously through poster presentations of bone mineral disorders and a case of DKA and metformin toxicity simultaneously.
Competitors for the AI Region
From microscope slides to clinic notes, nephrology is entering the algorithmic era.
Artificial intelligence is reshaping nephrology through the analysis of biopsy images and the interpretation of clinical text. Computational pathology and NLP enhance diagnosis, risk prediction, and research, but require high-quality data, validation, and strong governance for safe implementation.
Team 1: Computational Pathology
versus
Team 2: Natural Language Processing

Image generated by Matthew Sparks using ChatGPT at http://chat.openai.com, February 2026. After using the tool to generate the image, Sparks and the NephMadness Executive Team reviewed and take full responsibility for the final graphic image.
Team 1: Computational Pathology
What if the kidney biopsy could speak in pixels, and algorithms could listen?
Computational pathology harnesses image-based artificial intelligence to extract quantitative data from digitized kidney biopsies. In nephrology, these tools can identify glomerular lesions, quantify fibrosis, assess interstitial inflammation, and even predict outcomes with remarkable reproducibility. In contrast to traditional microscopy, AI does not fatigue or vary across observers, yielding scalable, standardized interpretation across centers. Yet difficulties remain: restricted annotated datasets, variability in staining and slide preparation, model bias, and regulatory concerns. Computational pathology promises precision and scalability, but it must earn trust through rigorous validation and thoughtful clinical integration.

Copyright: Bermek/ Shutterstock
Artificial intelligence (AI) provides a transformative lens for nephrology; just as glasses sharpen blurred edges, AI-driven tools reveal complex patterns in kidney disease that elude human vision. These innovations hold the potential to address long-standing limitations in diagnostic precision and therapeutic optimization. However, their integration into patient care demands attention to ethics, data transparency, safety, and validation across diverse populations.
- The Pathologist’s New Partner: From Interpretation to Quantification
The kidney biopsy report is a cornerstone of a nephrology diagnosis. A seasoned renal pathologist meticulously navigates a complex visual landscape using various modalities to identify patterns of morphologic injury. The integration of light, immunofluorescence, and electron microscopy data along with the clinical history and other laboratory values eventually leads to the critical diagnosis that guides our treatments.
While machines cannot replicate a renal pathologist’s nuanced, holistic judgment, they serve as a new lens for diagnostic efficiency. Within the digital pathology workflow, AI integration occurs at multiple stages of analysis. If natural language processing clarifies the documented echoes of the past, machine-vision provides the high-fidelity lens needed to see the biological reality of the present. Though importantly, the specific AI tool or model chosen must be aligned with the needs of the clinical task.
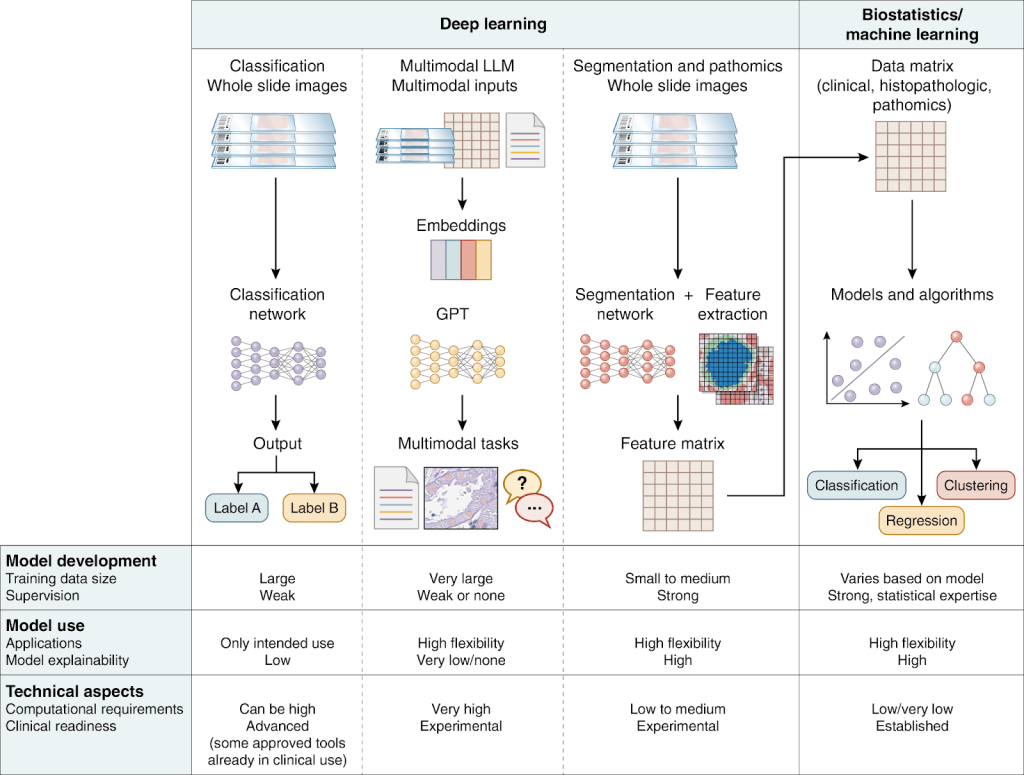
Different tasks and frameworks in computational pathology and their respective advantages and limitations. GPT, generative pretrained transformer; LLM, large language model. From “Precision nephrology: current status and future perspectives of artificial intelligence in kidney diseases” by Hölscher et al. (2025), Kidney International. Available under CC BY 4.0.
- Virtual Biopsies: In the U.S. renal transplant system, the United Network for Organ Sharing (UNOS) policy recommends integrating procurement biopsies, Kidney Donor Profile Index, and donor characteristics to assess organ quality. However, the logistical delay in obtaining pathology leads to prolonged cold ischemia time, where each extra hour may increase the risk of graft failure and mortality.
To address this, Yoo et al. (2024) created a machine-learning “virtual biopsy” system. Without a transplant kidney biopsy, the ensemble of machine-learning models with the eleven non-invasive parameters achieved a Multi-Area Under the Receiver Operating Characteristics Curve of 0.83 for arteriosclerosis and interstitial fibrosis and tubular atrophy (IFTA), and 0.77 for arteriolar hyalinosis. This tool can serve as a useful adjunct to centers with 24-hour pathology coverage and a rapid triaging tool for those without it. This approach empowers clinicians to expedite allocation of high-quality grafts without the delays inherent in biopsy processing and interpretation.
- Quantitative Accuracy: While the human eye often struggles to precisely quantify sclerosis or fibrosis, AI provides objective, reproducible quantification in minutes. Ginley and Jen et al. (2021) demonstrated this across biopsies from six institutions, where the model’s agreement with expert pathologists was quantitatively indistinguishable (ICC 0.94). Yi et al. (2021) achieved 72% accuracy in categorical IFTA grading using 67 whole-slide images. Deep learning models can rapidly segment key structures like glomeruli and tubules, providing measurements of percentage glomerulosclerosis or the extent of IFTA. This enables standardized diagnoses and provides a deterministic metric to track how a patient’s disease burden changes over time.
- Automated Triage: AI integration allows automatic triage of digital biopsies immediately upon scanning. Cases with a high probability of acute rejection can be flagged for priority review, while those with more benign features are placed lower in the queue. This smart system allows pathologists to prioritize their efforts toward the most complex and critical cases.
- Virtual Staining: Several studies have shown the ability to create virtual histochemical or immunohistochemical stains from unstained or standard hematoxylin and eosin (H&E)-stained slides. Typically, these algorithms employ generative adversarial network-based predictive models to computationally generate whole slide images of the stain. For example, Haan et al. were able to apply this technique to kidney biopsies to generate virtual special stains, including periodic acid-Shiff (PAS), trichrome, and Jones stains. They showed that the virtual stains proved statistically equivalent to traditional histochemical stains when used by renal pathologists in terms of diagnostic accuracy.
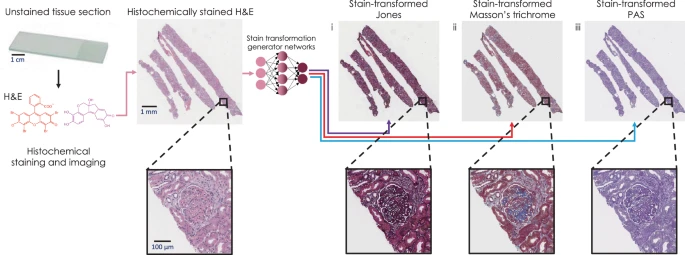
Overview of deep learning-based H&E stain transformation into special stains. Histochemical staining of H&E is digitally transformed using a deep neural network into JMS (Jones Methenamine Silver), MT (Masson’s Trichrome), and PAS (Periodic Acid-Schiff). From Deep learning-based transformation of H&E stained tissues into special stains by Haan et al. (2021), Nature Communications. Available under CC BY 4.0.
Virtual immunohistochemical staining can also be generated and translated to identify-specific cell types. For example, Govind et al. were able to use immunohistochemical stains for WT1 and p57 to label and identify podocytes. Using these stains as ground truth, a generative adversarial network (GAN)-based algorithm was made to predict pococytes. Although the significance of podocyte morphology and counts is currently unclear and too tedious for routine clinical diagnosis, the automation of evaluating these features may prove to have some prognostic relevance in the future.
- Adaptive Dialysis: From Reactive to Proactive Care
The greatest potential of machine-vision is evident in dynamic real-time clinical environments such as dialysis units. Although hemodialysis is fundamental to modern nephrology, current practice often depends on single-time-point assessment and retrospective methods including Kt/V calculations for dialysis adequacy, relative blood volume and bioimpedance. These approaches offer only a partial representation of the patient’s physiological status. This lack of real-time, patient-centric data is a major contributor to intradialytic hypotension (IDH), the most common hemodialysis complication. IDH is a significant risk factor for repeated organ injury, hospitalization, and mortality. It leads to cardiovascular morbidity and mortality, increased dementia, and loss of residual renal function. Text-based models use natural language processing (NLP) to parse old notes that rely on subjectivity. On the other hand, machine-vision can dynamically detect sub-clinical hemodynamic shifts before being detected by intermittent blood pressure (BP) cuff drops or reported by the patient.
Home-based care is often perceived as discharging patients with limited support, raising concerns about their wellbeing and outcomes. However, AI devices improve home care monitoring. By utilizing smartphones (owned by 90% of Americans) to capture photos of their face and peritoneal dialysis (PD) effluent, patients are empowered to use the “new glasses” of machine-vision to bring this remote landscape into clear focus. A machine-vision model can now analyze faces for edema, while another inspects the PD fluid for fibrin strands and early cloudiness. By bypassing the documentation gap of traditional monitoring, machine-vision models can identify peritonitis a median of 2.6 days before clinically apparent. In the CATCH study, an optical sensor system generated 140 notifications in 243 patients, of which 96 didn’t meet peritonitis criteria, highlighting both early detection and false-alert burden. This form of home-based, AI-assisted monitoring not only aligns with emerging clinical implementation guidelines but improves quality of individualized care by encouraging patient autonomy.
- Kidney Stone Disease: The Art of the Stone
The complexity of kidney stone disease makes it a prime candidate for machine-vision. While computed tomography (CT) imaging is the current standard for diagnosis, interpretation remains an art form. This process often involves considerable uncertainty, potentially leading to sub-optimal therapeutic decisions. Machine-vision promises to turn the scan into a precise predictive tool.
- Predicting Stone Composition: AI models trained on thousands of CT scans with known stone compositions can now predict the type of kidney stone. For example, a recent study validated that an AI model can predict stone composition from CT images with robust accuracy. This is vital, as a uric acid stone might dissolve with medical therapy, while a calcium oxalate stone may require lithotripsy or surgical removal. Machine-learning analyzes not just the Hounsfield units, but also radiomics, and the texture, shape, and heterogeneity of the stone on a CT scan, accurately identifying stone type. This is a critical distinction that can save a patient from an unnecessary and costly procedure.
- Guiding Treatment Decisions: Beyond composition, AI can also analyze the stone’s shape, location, and the patient’s anatomy, thereby predicting response. A recent study developed a model with over 90% accuracy in predicting the stone-free status after a single session of shock wave lithotripsy. A clinical decision-support system could take a patient’s CT scan, predict a 20% success rate for lithotripsy based on the stone’s location and density, and then recommend ureteroscopy instead with an estimated 95% predicted success rate, providing straightforward evidence for the most effective intervention.
- Predictive Risk Assessment: AI can also identify patients at elevated risk for future stone formation. By analyzing subtle signs of hyperoxaluria or other metabolic abnormalities on CT and combining it with 24-hour urine collection, the system could alert the nephrologist to initiate preventative measures. This moves us from a reactive, symptom-based model to a proactive, preventative one, fundamentally changing how we manage this common disease.
- The Logistical Imperative: Making It a Fixture, not a Fad
For machine-vision to be truly effective, it must become a seamless part of our daily workflow to aggregate statistically relevant datasets and build effective models that can be used for better patient care.
- Electronic Medical Record and Data Integration: AI tools must integrate with existing electronic medical record (EMR) systems through a robust extract, transform, load (ETL) framework. This requires the development of standardized application program interfaces and data exchange protocols to allow for the seamless transfer of visual data, AI-generated insights, and clinical recommendations back into the patient’s chart. Currently, inter-EMR communication remains challenging. Without this integration, technology risks becoming an isolated tool with limited clinical utility.
- Validation and Clinical Workflow: Before AI-powered diagnostic or predictive tools become widespread, they must undergo rigorous validation in a clinical setting. This process involves proving their accuracy, reproducibility, and safety in diverse patient populations. This validation process is critical because AI models can confidently perform poorly when given sub-optimal or incomplete data. Also, clinical workflows must be adapted to incorporate these new insights. For example, a flagged biopsy should seamlessly be added to a pathologist’s queue for immediate review, and a predictive alert for IDH must be a clear and actionable signal for the care team.
- Data Security and Privacy: Visual data, which can contain sensitive patient information, necessitates robust data security measures. Platforms must have end-to-end encryption, multi-factor authentication, de-identified and strict access controls to ensure patient privacy is protected at every step, from image capture to analysis and reporting.
- Calibrating the Lens: Addressing the Equity Gap
While machine-vision has the potential to democratize expertise, these “new glasses” are only as effective as their calibration. To ensure this technology provides a clear view for all, we must address the Digital Determinants of Health (DDOH). In rural and under-resourced communities, DDOH includes limited access to affordable high-speed broadband, and limited digital literacy and technical knowledge, creating a refractive error in care delivery. Without a clear strategy to overcome these fundamental infrastructural barriers, this technology risks further entrenching existing access disparities.
- Clinical Guardrails: The very data used to train AI models can contain biases that reflect historical healthcare disparities. To ensure machine-vision is a force for good, we must be vigilant in implementing ethical leashes. The World Health Organization (WHO) and other bodies are now releasing guidance on how to manage these risks. At its core, AI software needs a foundational commitment to patient privacy and explicit, informed consent. Frequent model audits, intentionally curated benchmark datasets to assess biases, required human review, and constant retraining are essential to mitigate bias and model drift, ensuring that the technology is an equalizer, not a perpetuator of inequality.
- The Horizon: Multimodal AI and Integrated Insights
Nephrology is shifting from single-task automation toward integrative predictive analytics. By consolidating data streams, AI provides useful biological predictions. Its utility will depend on rigorous monitoring of the development lifecycle, from raw data acquisition to real-world deployment, and continual re-evaluation.
- Integrated AI for Deeper Insight: Imagine a system that takes a patient’s kidney biopsy image, their genomic data, real-time dialysis vitals, and their electronic health record, and synthesizes these data. This integrated approach could uncover disease subtypes we do not currently recognize, predict an individual’s response to specific immunosuppressants, or forecast the precise rate of eGFR decline. This approach moves beyond single-task automation toward true predictive analytics for personalized medicine.
- Accelerating Research and Drug Discovery: The application of machine-vision can also accelerate research itself. AI models can analyze vast libraries of biopsy images and genetic data to identify new therapeutic targets or drug candidates. By digitizing and systematically analyzing this information, we can pinpoint novel pathways of disease progression and design effective therapies.
- Training the Next Generation: Beyond the direct clinical benefits, machine-vision and AI will revolutionize medical education. Trainees could use interactive platforms to practice interpreting digital biopsy slides with real-time feedback from an AI mentor. The system could highlight key pathological findings and provide links to relevant literature. This would offer a standardized and scalable way to train the next generation of nephrologists and pathologists, ensuring a consistent level of expertise across the globe.

Flowcharts of artificial intelligence technology during training and deployment. From “Artificial intelligence in renal pathology: Current status and future” by Feng and Liu (2023), Biomol Biomed. Available under CC BY 4.0.
Conclusion: The Case for Machine Vision
Multimodal sensing breakthroughs may democratize high-quality patient care. With thoughtful implementation and rigorous calibration, these tools could move from experimental novelty to clinical fixture, simultaneously reducing healthcare costs and elevating the standard of care. NLP remains a valuable filter for the after-images of the past, but it is limited by the noise of human documentation and the silence of its absence. In contrast, machine-vision witnesses the clinical present with high-fidelity, capturing the biological reality. By addressing the digital determinants of health at the design level, we ensure our digital lenses raise the quality of care not for a few, but bring a clear path into sharp focus for all.
Team 2: Natural Language Processing
Every nephrology clinic note holds hidden data. If only we knew how to read between the lines.
Natural Language Processing (NLP) enables computers to interpret unformatted clinical text, from biopsy reports to dialysis notes and transplant summaries. In nephrology, NLP can identify CKD phenotypes, extract trends in proteinuria, detect medication toxicity, and support real-time clinical decision-making. Its strength lies in obtaining insights from narrative data that would otherwise remain inaccessible for research or quality improvement. However, variability in documentation, ambiguous terminology, concerns about data confidentiality, and the risk of contextual misinterpretation pose significant limitations. NLP transforms words into structured knowledge, but accuracy depends on careful model design and clinical oversight.

Copyright: Gorodenkoff/ Shutterstock
Introduction
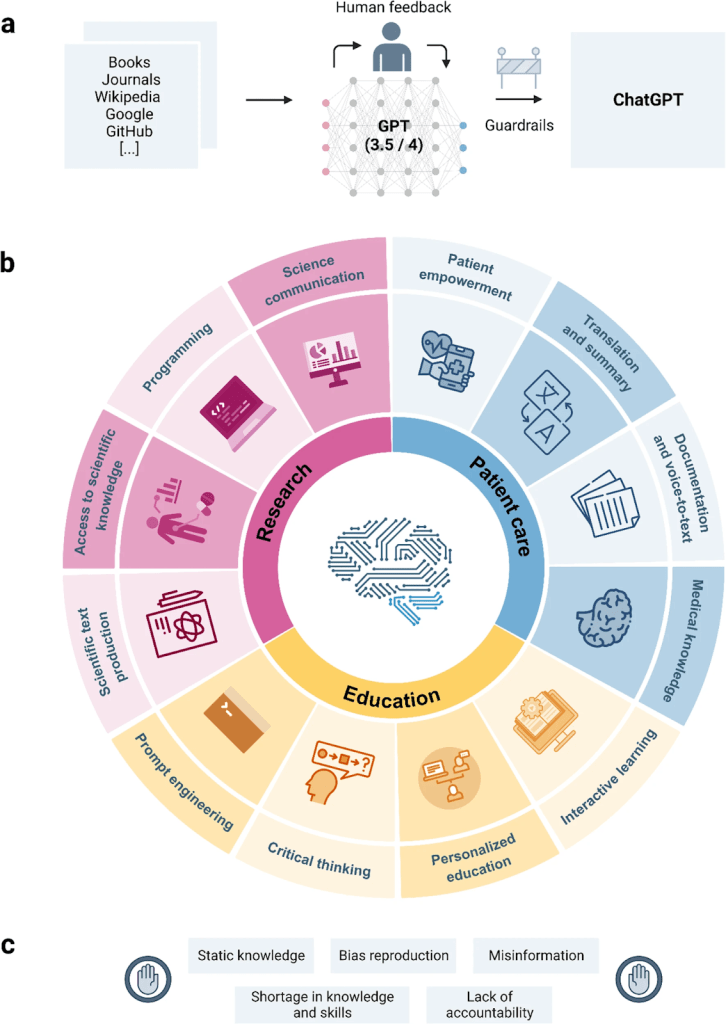
Clinicians can spend as much as 36 minutes charting per patient visit, 6.2 “pajama minutes” (EMR time between 5:30 pm and 7 am), and another 7.8 minutes of inbox management per patient visit according to one study based on 309 PCPs published in JAMA. This study also analyzed current team-based approaches, such as the role of pharmacy technicians in medication refills. However, this time spent doesn’t necessarily translate into optimized patient-focused care. In an evolving time of complex EMR, NLP can provide relief for clinicians, nurses, and ancillary staff by efficiently coordinating patient care. NLP is a subset of artificial intelligence used to understand human language. Within NLP, generative AI refers to models that produce new text based on learned language patterns, with large language models (LLMs) representing a modern class of NLP capable of understanding and generating human language. For nephrologists, this advancement in free text analysis could be used in myriad applications, as shown in the images below. In this post, we will discuss specifically predictive models for chronic kidney disease (CKD), identification of patient symptomatology on maintenance hemodialysis (HD), and reducing administrative burnout.
Natural Language Processing background
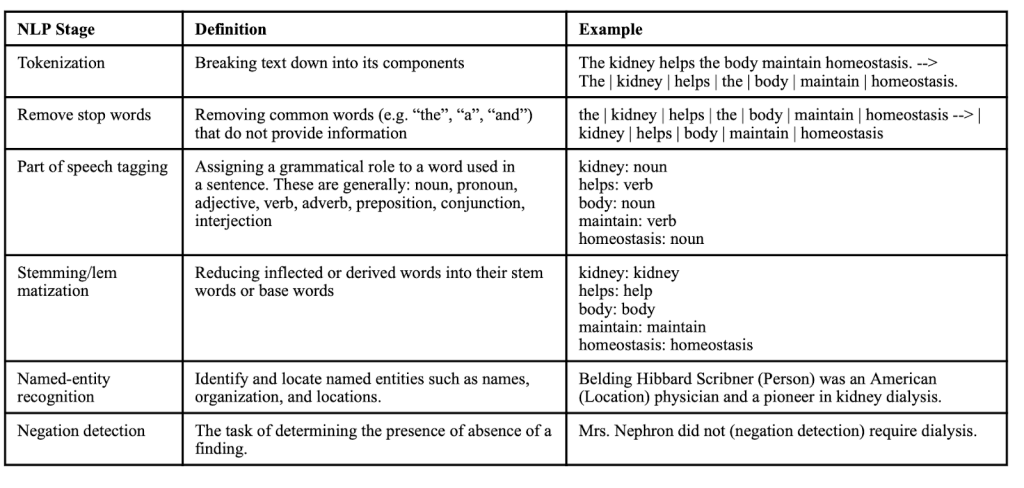
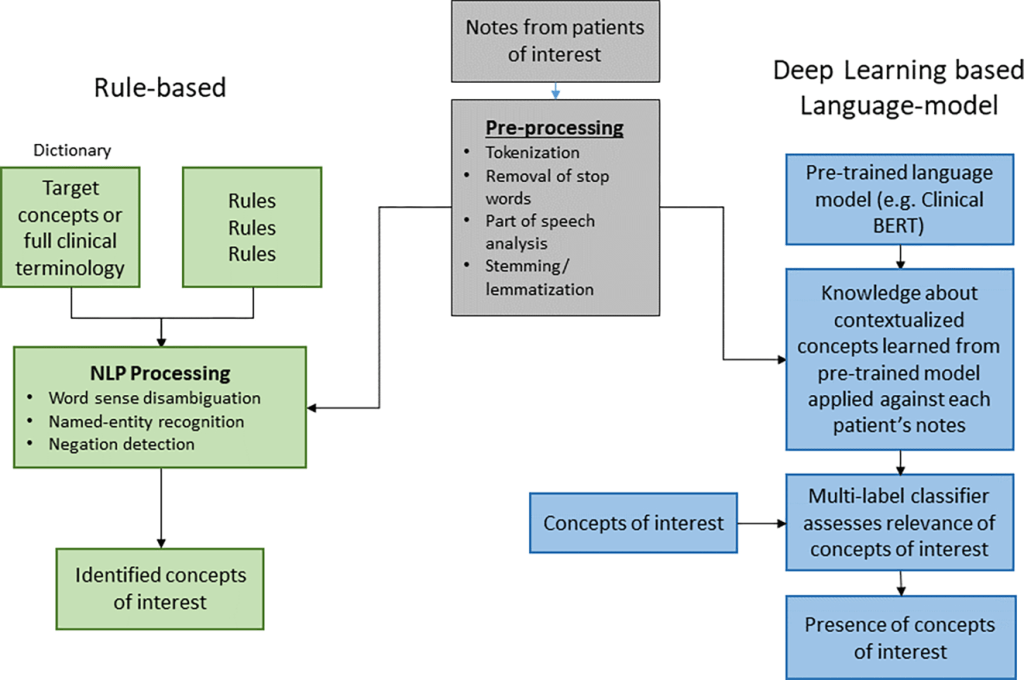
NLPs analyze linguistic constructs of texts such as sentence structure, synonyms, abbreviations, negation, and inflections. The goal of NLP is to identify concepts from text and filter out noise. Traditionally, older models relied on rule-based algorithms which use hard grammatical rules to infer meaning from the text. These can be used in a limited dataset with consistent sentence structure (i.e., simplified templates), but do not “learn” from variation (yes, Skynet has not yet become self aware, fortunately). Examples of some of the technical rules are described below.
The introduction of transformers revolutionized NLP by replacing sequential processing with the attention mechanism which evaluates relationships between all words simultaneously. One early example of such a pretrained transformer algorithm that is open source is the Bidirectional Encoder Representation from Transformers (BERT). This is the technology that has enabled LLMs such as ChatGPT and Gemini. This has set the stage for development of more focused models, as shown below.
NLP Uses in Nephrology
In nephrology, rule-based NLP can help identify progression of CKD, extract outcomes from AKI, and highlight patient-centered symptoms, leveraging the fact that much of kidney care is documented in narrative text rather than images. LLMs are more flexible and can be used to enhance patient-provider communication, synthesize medical literature, and improve provider workflows. The image below depicts current AI architecture broadly and how it can be applied to medicine.
Novel Predictors which may Identify Progression of CKD using EHR
Improvements in early detection and prognostication can lead to improved outcomes, frequent monitoring, and appropriate referrals. The presence of longitudinal data and clinical narratives were used to develop a risk prediction model that identified progression of CKD stage III to IV using heterogenous data. The main question of the study was how to elicit both data points in the prediction model. This study looked into several different statistical models, one of which — Laboratory Test and Text Kalman Filter (LTKF) — incorporated text-based factors from discharge summaries, outpatient primary care, and specialty notes. The study used free text and an unsupervised topic modeling technique called, latent Dirichlet allocation (LDA), for about 50 topics. Variables included well-known risk factors for CKD including diabetes, heart failure, and dialysis. This model had the highest concordance with the classic eGFR model with a 0.849 concordance correlation coefficient (CCC) after performing internal validation.
Another study examined the relationship of thousands of concepts extracted from clinical notes and the development of ESRD and revealed 129 concepts of positive and negative predictors via Cox regression. In this particular study, concepts were extracted via a two step process. First, negated phrases (process of determining presence of an absence of an item) were removed via the NexEx negation engine using Python. Second, notes were processed via the National Library of Medicine’s MetaMap software (program developed at the National Library of Medicine to map biomedical knowledge consisting of concepts classified by semantic type, and hierarchical and non-hierarchical relationships among concepts).
Based on positive predictors, the kidney failure risk equation (KFRE) or Tangri score (predictive model for progression of CKD to kidney failure) was calculated adjusting for multiple hypothesis testing. The concept of proteinuria had a HR of 2.16 (95% CI, 1.51 to 3.10; q<0.001) and nephrotic range proteinuria had an HR of 5.09 (95% CI, 2.48 to 10.40; q<0.001). Other notable positive predictors included concepts related to heart failure (95% CI, 1.65 to 3.43; q<0.001) and hyperkalemia (95% CI, 1.43 to 3.63; q<0.004); such conditions and complications are expected to be seen with ESRD a true risk factor. While all these concepts should not be taken without careful consideration of association versus correlation and in the setting of large volume hypothesis testing, there is opportunity for further investigation for these predictors that may not be so commonly associated with progression of CKD (fast food, history of psoriasis, ascorbic acid).
Better Identifying Patient Symptoms with NLP
Let’s step back and think about some of the difficulties faced by nephrology on a day-to-day basis: your dialysis patient has a sparsely populated chart that does not give the latest treatments. Here is one such place where NLP can help. Can charts even give us up-to-date information on a patient’s symptoms and ability to fine tune treatments, possibly even better than physicians who do not assess patients every treatment?
One study of 97 ambulatory dialysis patients looked at symptom studies collected from patients, nurses, and physicians. An NLP algorithm was developed from the patients’ EHR progress notes (written by physicians, nurses, nutritionists, and social workers) and validated the performance of the algorithm vs manual chart review and patients’ surveys. The most common reported symptoms included fatigue (61%), cramping (59%), dry skin (53%), muscle soreness (43%), and itching (41%). Results were noted on the validated Dialysis Symptom Index that asks patients about the presence and severity of 30 different symptoms from 0-4. The algorithm had high sensitivity and specificity compared to manual chart review. However, it found less than 50% of symptoms as compared to what patients reported on their surveys (i.e., 15% of fatigue, 39% of cramping). While this study is limited by its reliance on progress notes from HD units, it demonstrates a clear use case for NLP to identify patient-centered outcomes that cannot be captured through imaging.
Underdocumentation of CKD
As CKD rates grow with an aging population, there is still a gap between detection of CKD and appropriate referral. An estimated 30% (32 of 107 patients) with verified moderate CKD lacked appropriate documentation, with 22% (24 of 107) patients receiving regular primary care follow up. As a community, we are responsible not only for providing preventative care but also rating risk factors prompting early referrals. A lexical classification was developed by nephrologists using 39 medical terms to identify CKD in a note.
In this study, notes were categorized using two separate methods (classifier and word count) with CKD documentation or not. Results showed high specificity (99.8% and 98.8%, respectively) and sensitivity (95.4% and 99.8%, respectively) with either method. The implication of this study is that with more validation for a population- based study (taking into account socioeconomic status, race, gender) it could be possible to identify a CKD tool that incorporates already documented risk factors to prompt early CKD. Even in this model, there was a calculated 96.8% positive predictive value (meaning 4% could be incorrectly identified CKD) and 94%-99% negative predictive value (meaning missed patients that would not have proper classification).
Reducing Administrative Burnout
Rising messaging and documentation demands are key contributors to physician burnout. NLP can help mitigate the workload by triaging in-basket messaging with a reasonable level of accuracy. A ChatGPT-generated simulation correctly triaged 93% of messages when evaluated by two different nephrologists. Ambient AI scribing, using an AI tool to listen to patient-provider conversations and automatically generate a medical note, is rapidly being adopted across healthcare systems. In an earlier study at a large healthcare system, they found that primary care physicians who used ambient AI had statistically significantly larger decreases in time spent in the EHR outside of 7 a.m. and 7 p.m. and in time spent in notes during appointments. Clinicians rated the quality of the notes to be high, with average transcript scores of 48 out of a possible 50 points. There were two recent randomized controlled trials evaluating ambient AI. One compared Microsoft Dragon Ambient eXperience (DAX) Copilot or Nabla vs. control. They found that Nabla users had decreases in time-in-note while there was no change in the DAX or control group. There were improvements in some quality of life indicators such as burnout and physician task load. In another trial, the use of ambient AI led to significant reductions in work exhaustion/interpersonal disengagement and work outside of work. Together, these findings highlight why NLP-based applications may deliver more immediate and scalable benefits than imaging-focused AI, by directly reducing clinician workload, improving workflow efficiency, and alleviating documentation burden.
Improving Provider-Patient Communication
Provider-patient communication often contains medical jargon that may be difficult for patients to understand. Tailoring after visit summaries and discharge summaries to a patient’s reading level and language can facilitate their care. In one study, researchers used a LLM to improve the readability of 50 hospital discharge summaries. They found that the LLM discharge summaries significantly lowered the Flesch-Kincaid Grade Level (6.2 [0.5] vs 11.0 [1.5]; P < .001) and significantly improved the Patient Education Materials Assessment Tool understandability scores (81% vs 13%; P < .001). However, these were not perfect discharge summaries and two physician reviews indicated 18 discharged summaries had safety concerns including omissions and hallucinations indicating the need for continued human oversight.
Personalizing Kidney Disease Education
Precision education tailors instruction to an individual learner’s level of knowledge and needs. LLMs are proving to be effective study tools in medical education, including for question generation. A systematic review identified eight studies that described using LLMs to generate medical questions. Overall, five of the eight studies were able to generate valid medical questions.
LLMs are able to review and synthesize literature and guidelines. LLMs such as OpenEvidence, an AI-powered platform that leverages retrieval-augmented generation which grounds the response to peer-reviewed journals and guidelines, is used daily by40% of physicians in the United States. In one study, researchers had four physicians review OpenEvidence clinical decision making for five patient cases. Physicians rated the AI output to have high clarity (3.55±0.21), relevance (3.75±0.43), support (3.35±0.22), and satisfaction (3.60±0.38) using a scale that ranged from 0 to 4. LLMs can help clinicians stay up to date with rapidly expanding medical literature. However, we must be careful not to become over reliant on LLMs as this can lead to deskilling early on in training and unskilling for more advanced clinicians. In particular, we must be careful that trainees who have not yet developed their clinical reasoning are using AI to support their learning rather than replacing it.
Conclusion
NLP has enabled us to care for patients in ways that were previously not possible. A recently published review discusses the expansion of AI applications in the field of nephrology. We now have the ability to analyze big data and recognize patterns that would not have been possible with standard statistical models. AI can augment the workflow of a nephrologist burdened with heavy clinical volume, alert fatigue, messaging, and other daily tasks. NLP tools can help us communicate with patients better and tailor education to each person’s literacy, language, and informational needs. Despite existing limitations, NLP’s capacity to unlock meaning from text underscores the importance of words in understanding and improving kidney care.
– Executive Team Members for this region: Ana Catalina Alvarez-Elías @catochita – @catochita.bsky.social and Jeffrey Kott @jrkott27 – @jrkott27.bsky.social | Meet the Gamemakers
How to Claim CME and MOC
US-based physicians can earn 1.0 CME credit and 1.0 MOC per region through NKF PERC (detailed instructions here). The CME and MOC activity will expire on June 1, 2026.









Leave a Reply