#NephMadness 2026: IgA Nephropathy Region
Submit your picks! | @NephMadness | @nephmadness.bsky.social | NephMadness 2026
Selection Committee Member: Laura Mariani @LauraMarianiMD
Dr. Mariani is an Associate Professor and clinical researcher at the University of Michigan. She is the Assistant Nephrology Fellowship Director and enjoys teaching glomerular disease to trainees at all levels. Her research program includes several observational studies in glomerular diseases where she works to identify novel markers of disease progression and clinical trial outcomes.
Writer: Alessandra Tomasi @AleTomasiMD
Alessandra Tomasi is a second-year nephrology fellow at Duke University in Durham, NC. She completed her medical education at UNC Chapel Hill, and her internal medicine residency as well as chief residency at Mayo Clinic in Rochester, MN. She plans to pursue a career in academic nephrology, with a focus on glomerular disease and medical education.
Competitors for the IgA Nephropathy Region
IgA Nephropathy is totally having its main character era right now, and the vibes are stellar. On one side, you’ve got Team Complement Inhibitors pulling up like, “We’re here to shut down that overactive cascade, stay mad.” Meanwhile, Team B‑Cell Modulators is rolling in with the “let’s fix the source” energy. It’s giving rivalry, it’s giving plot twist, and honestly the nephrology world is just grabbing popcorn while these two squads battle it out for who’s going to be the real MVP of IgA nephropathy therapy.
Team 1: B-Cell Modulators
versus
Team 2: Complement Inhibitors
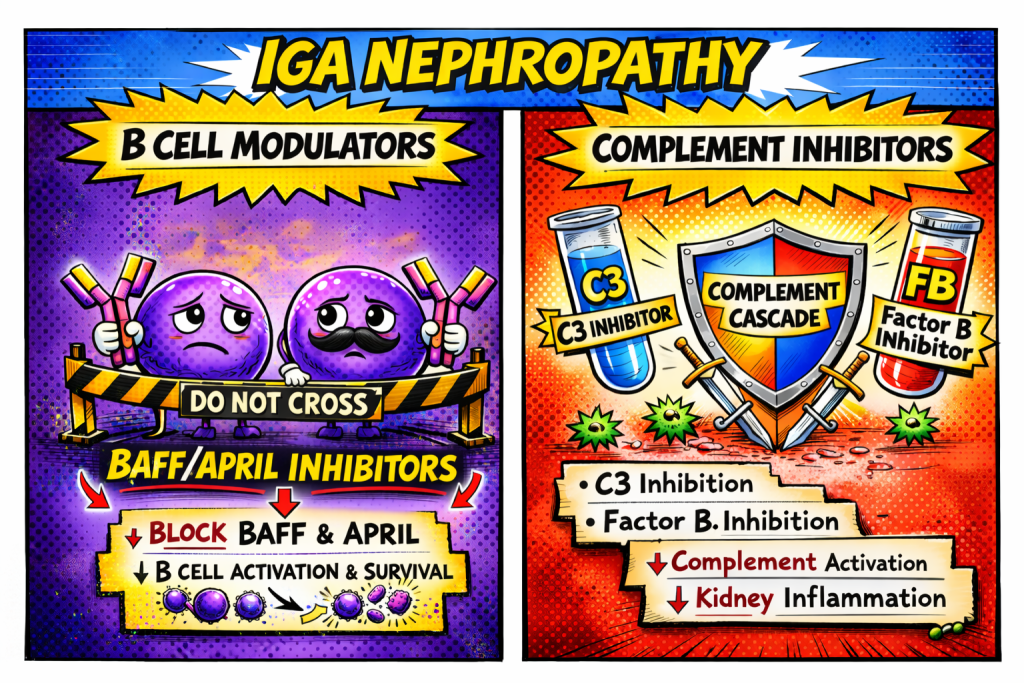
Image generated by Matthew Sparks using ChatGPT at http://chat.openai.com, February 2026. After using the tool to generate the image, Sparks and the NephMadness Executive Team reviewed and take full responsibility for the final graphic image.
Is it déjà vu? Or already time for an update?
IgA nephropathy had its moment in the spotlight during NephMadness in 2023. At the time, the debate was immunosuppression versus non-immunosuppressive therapy. The Blue Ribbon Panel picked non-immunosuppression!
Fast forward three years. The world of immunosuppression for this disease continues to grow, and is now ready for a comeback.
Part of what has allowed for this comeback is the adoption by drug regulators of proteinuria reduction as a reasonably likely surrogate end point for treatment efficacy in IgA nephropathy, providing a basis for accelerated approval by the FDA for new therapies. What this means is that, for this relatively rare disease, clinical trials became more feasible. With proteinuria reduction shown to reflect treatment effect on kidney failure, this can provide a basis for accelerated approval. Following approval, the benefit is verified with a post-marketing confirmatory trial.
Accelerated approvals have thus paved the way for exciting new therapy options. But before we dive into our competitors for NephMadness 2026, let’s remind ourselves of the pathophysiology of IgA nephropathy, and take a tour of the recent clinical updates.
As we learned last time, IgA nephropathy is the most common cause of primary glomerular disease worldwide. It is thought to develop following a four-hit hypothesis:
- the production of galactose-deficient IgA1 (Gd-IgA1),
- the production of IgG and IgA autoantibodies against Gd-IgA1,
- the formation of immune complexes containing IgG, IgA, and Gd-IgA1, and
- mesangial deposition of these circulating immune complexes.
Understanding this underlying pathophysiology is key to recognizing the potential role for disease-modifying therapies, including immunosuppression. More to come.
While IgA nephropathy is common, few patients present with severe disease manifestations such as rapidly progressive glomerulonephritis (RPGN). More commonly, individuals present with a milder disease form. This can be highly variable, ranging from synpharyngitic gross hematuria, to microscopic hematuria, proteinuria, hypertension, and elevated serum creatinine levels. Diagnosis is ultimately confirmed via kidney biopsy, which characteristically shows immune complex deposition in the mesangium with IgA dominance on immunofluorescence.
At the time of biopsy, the Oxford classification, also termed the MEST-C score, can be used as an initial assessment of disease severity. It is a histopathologic scoring system that facilitates quantification of kidney damage based on 5 key components: Mesangial hypercellularity, Endocapillary hypercellularity, Segmental glomerulosclerosis, Tubular atrophy/interstitial fibrosis, and Crescent formation. The International IgAN Prediction Tool can then be used to combine the MEST score (the -C component is not included) with additional clinical data, including eGFR, proteinuria, blood pressure, and demographic information, to help determine prognosis in adults with IgA nephropathy for up to two years following initial biopsy. Together, these histologic and clinical findings can help guide treatment decisions.
So let’s talk management. In accordance with the recently updated (and very timely) 2025 KDIGO guidelines for IgA nephropathy, treatment of this disease considers several key goals:
- stopping the production of pathogenic IgA and associated immune complex formation as well as resultant kidney injury,
- reducing glomerular hyperfiltration and proteinuria, and
- managing hypertension.
Non disease-specific interventions to manage blood pressure and reduce glomerular hyperfiltration and proteinuria include lifestyle modifications, renin-angiotensin system (RAS) inhibitors, sodium-glucose cotransporter 2 (SGLT-2) inhibitors, and/or selective endothelin (ERA) or dual endothelin angiotensin receptor antagonists (DEARA).
With regard to endothelin receptor antagonists, the DEARA sparsentan and selective endothelin type A antagonist atrasentan both boasted meaningful reductions in proteinuria in patients with IgA nephropathy in the PROTECT and ALIGN trials, respectively, paving their way towards the forefront of disease management. You can check out the recaps from NephMadness 2023 for more details. But to summarize, the PROTECT trial demonstrated a 50% reduction in proteinuria from baseline at 36 weeks in patients taking sparsentan compared to 15% in those taking irbesartan, and the ALIGN trial found that patients taking atrasentan experienced a 38% decrease in proteinuria compared to 3% in those taking placebo. The PROTECT trial also notably showed a total 2-year slope eGFR of −2.9 mL/min/1.73m2 per year in the treatment group, compared to -3.9 mL/min/1.73m2 per year in the irbesartan group. These findings ultimately led to full FDA approval for sparsentan and accelerated (though not yet full) approval for atrasentan. It’s important to note, due to the risk of hepatotoxicity with endothelin receptor antagonists, the FDA requires patients on sparsentan to be enrolled in the Risk Evaluation and Mitigation Strategy (REMS) program for regular liver function testing. This is not the case for those on atrasentan, though liver function monitoring still needs to occur. Importantly, both DEARA and ERA medications are fetotoxic and should not be used in pregnancy. And, both studies excluded patients on immunosuppressive therapy.
So, what about immunosuppressive interventions? Last time, steroids (including prednisone and budesonide) were gaining traction— in fact, budesonide has since received full FDA approval following results from the NefIgArd trial. Additional medications including mycophenolate, cyclophosphamide, and azathioprine were starting to be discussed following the completion of a few major (albeit limited) trials. And rituximab and calcineurin inhibitors remained on the sidelines.
But what’s been new since 2023 in the realm of IgA nephropathy? That’s right, it’s now time for our 2026 competitors to take the stage: introducing B-cell modulators versus complement inhibitors.
It’s the “New England Journal of IgA” these days, and we’re just living in it! In this special NephMadness edition, hosts Kenar Jhaveri and Koyal Jain are joined by Matt Sparks and Ale Tomasi to break down the heavy hitters in the IgA Nephropathy Region. Matt’s dog also joins us for a special, possibly biased cameo. We’re moving past “ACE first, think later” and diving into the upstream battle: B-cell modulators (BAFF/APRIL inhibitors) versus Complement inhibitors. Whether you’re team “Hit Zero” or team “Alternative Pathway,” this episode covers the latest trial data from ORIGIN, VISIONARY, and APPLAUSE to help you fill out your bracket.
Episode 12: NephMadness Special! IgAN (Again?!): New B-Cell Targets vs. Complement Inhibitors
COMMENTARY BY JONATHAN BARRATT:
IgA Nephropathy – The Bench is Loaded, but is There a Clear Star?
Team 1: B-Cell Modulators
Team B‑cell Modulators in IgA Nephropathy is anchored by the first FDA‑accelerated approved APRIL inhibitor, sibeprenlimab, which achieved a ~50% reduction in proteinuria and represents a true disease‑modifying milestone for IgA nephropathy. Alongside sibeprenlimab, next‑generation BAFF/APRIL‑targeting agents such as atacicept, telitacicept, and povetacicept are advancing through late‑phase development and further validating the upstream B‑cell–driven biology of IgAN. Together, these therapies signal a transformative shift away from nonspecific immunosuppression toward precise, mechanism‑based treatments that offer real hope for altering the long‑term course of IgA nephropathy..

Copyright: Ana Aguirre Perez/ Shutterstock
“April showers bring… more antibodies” – Proverb (adapted)
Let’s start with some definitions. B-cell Activating Factor (BAFF) and A PRoliferation-Inducing Ligand (APRIL) are cytokines. They are both members of the tumor necrosis factor (TNF) superfamily. And they each act as key regulators of B cell development and survival.
BAFF is produced by T cells as well as various immune cells of the innate system. While B cells are not dependent on BAFF for survival while in the bone marrow, once they are released into the periphery they become responsive to BAFF. Specifically, BAFF binds to three receptors: B-cell maturation antigen (BCMA), transmembrane activator and calcium-modulator and cyclophilin ligand interactor (TACI), and BAFF receptor (BAFF-R). BCMA is primarily found on plasma cells, TACI is expressed on mature B cells as well as activated plasma cells, and BAFF-R is found mostly on B cells. In its active role, BAFF facilitates peripheral B cell selection and survival, as well as helps maintain follicular- and marginal-zone B cell populations. Pathologically, BAFF can also help promote the inappropriate survival of auto-reactive B cells, such as in the case of lupus and— you guessed it— IgA nephropathy.
APRIL is similarly produced by T-cells, as well as innate immune cells such as macrophages and dendritic cells. Once released from the bone marrow into the periphery, B cells rely on APRIL for key processes including antibody class-switching and ultimately their differentiation into antibody-secreting plasma cells. In its active role, APRIL binds to multiple different receptors to facilitate the above, including BCMA and TACI (similarly to BAFF).
Recall from our introduction the 4-hit hypothesis that underlies the pathophysiology of IgA nephropathy. Steps 1, 2, and 3 involve the production of an aberrant IgA molecule (Gd-IgA1), the production of IgG and IgA autoantibodies against Gd-IgA1, and the formation of immune complexes containing IgG, IgA, and Gd-IgA1. It stands to reason that perhaps selective targeting of BAFF and/or APRIL will prevent these hits from happening effectively. Let’s look at the data so far.
First, atacicept. A fully human recombinant fusion protein provided as a once-weekly subcutaneous injection, atacicept targets both BAFF and APRIL. An initial phase 2 (ORIGIN) trial enrolled 113 study participants with IgA nephropathy, who received either 25 mg, 75 mg, or 150 mg of atacicept, or placebo. The study showed a 25% reduction in proteinuria at 24 weeks (p=0.037). Notably, at the end of 36 weeks, sustained reduction in proteinuria persisted, with a mean decrease by 35% (p=0.004); additionally, the authors measured a significant decrease in circulating Gd-IgA1, by 60% (p<0.0001) in patients taking atacicept. Notably, serum levels of IgG, IgA, and IgM decreased following treatment as well, by 35.5%, 63.5%, and 74.6%, respectively. These levels stabilized after week 24, and the study showed no occurrences of hypogammaglobulinemia.
Participants were subsequently enrolled in an open-label extension study during which they received 150 mg of the drug for an additional 60 weeks. This included patients who were switched from placebo. This phase 2b extension trial further demonstrated that treatment with atacicept resulted in sustained reduction in proteinuria (by 52%), hematuria, and circulating Gd-IgA1 levels at 96 weeks, with stabilization of eGFR (mean eGFR slope 0.6 mL/min/1.73 m2).
The phase 3 ORIGIN trial is ongoing, for which an interim analysis has been published. A total of 203 patients were included in this prespecified analysis (106 in the atacicept and 97 in the placebo group), and the data demonstrated a 45.7% reduction in proteinuria (urine protein/Cr ratio) in the atacicept group, compared to 6.8% in the placebo group. Additionally, resolution of hematuria (defined as a reduction to negative or trace dipstick findings) occurred in 81% of patients in the atacicept group compared to 20.7% in the placebo group. Importantly, 99% of patients in this trial were receiving angiotensin-converting enzyme (ACE) inhibitor or angiotensin-receptor blocker (ARB) therapy, and 53% were taking SGLT2 inhibitors. Of note, observed adverse effects in the atacicept cohort were mild or moderate in severity. More to come on the eGFR slope.
Next, sibeprenlimab. Unlike atacicept, this monthly subcutaneous injection is a humanized monoclonal antibody that targets APRIL alone. Its initial phase 2 trial (ENVISION) enrolled 155 patients who underwent randomization to receive 2 mg/kg, 4 mg/kg, or 8 mg/kg of sibeprenlimab once monthly for 12 months, or placebo. The study showed up to a 62% reduction in proteinuria (depending on dose administered), with as low as a 0.2 mL/min/1.73m2 decline in eGFR during the 12 month study period.
Also underway is a phase 3 (VISIONARY) study for which an interim analysis has been published. Among the 320 patients included (152 in the sibeprenlimab and 168 in the placebo group), at 9 months there was a 50% reduction in proteinuria (urine protein/Cr ratio) in those receiving the study drug compared to a 2% increase in proteinuria in the placebo group, with similar safety profiles. Additionally notable was the demonstrated 67% reduction in pathogenic Gd-IgA1 in patients receiving sibeprenlimab. Hematuria was significantly reduced in patients taking sibeprenlimab, from a prevalence of 78.3% to 19.8%, compared to no change in the placebo group (70.8% prior to treatment, 69% after receiving placebo). Importantly, nearly 98% of patients in this trial were receiving ACE inhibitor or ARB therapy, and 42% were taking SGLT2 inhibitors.
Both atacicept and sibeprenlimab are novel to the treatment of IgA nephropathy, showing significant promise with their targeted BAFF and/or APRIL inhibition. As we continue to study and elucidate the roles of BAFF and APRIL in IgA nephropathy, the therapeutic benefits of these targeted treatments will undoubtedly continue to be clear. As of this year, the FDA has granted accelerated approval for sibeprenlimab, and atacicept is under FDA review.
Team 2: Complement Inhibitors
Complement activation is a key driver of injury in IgA nephropathy, making complement blockade a highly rational therapeutic target. Iptacopan, the first approved complement inhibitor for IgAN, slows eGFR decline and reduces proteinuria by selectively inhibiting the alternative pathway. Emerging agents such as pegcetacoplan, vemircopan, ravulizumab, and cemdisiran further expand a rapidly growing toolkit aimed at targeting multiple complement pathways.

Copyright: fengdr2020/ Shutterstock
“Everybody likes a complement.” – Abraham Lincoln (adapted).
The complement system is made up of several plasma proteins that help create a cascade of inflammation, protection, and destruction.
Recall that there are three key pathways for complement activation: classical, lectin, and alternative. The classical pathway is initiated following antibody-antigen binding, the lectin pathway by the binding of mannose-binding lectin to mannose on bacterial and viral surfaces, and the alternative pathway can be initiated spontaneously as a direct response to foreign or pathogenic material. All three of these ultimately lead to the same effector molecules, and to a shared (terminal) complement cascade.
Evidence of complement deposition in glomeruli affected by IgA nephropathy is common, and increasing literature is starting to clarify the role of complement activity in the development of this disease. Specifically, the alternative and lectin pathways have been most implicated, and to this end, the role of complement inhibitors in the management of IgA nephropathy has gained significant traction.
Enter iptacopan. An oral complement factor B inhibitor (CFB), iptacopan works specifically by blocking the alternative complement pathway: inhibition of CFB prevents the formation of C3 convertase (C3bBb), thereby inhibiting the cleavage of complement protein C3. Without these steps, inflammation is dampened.
The APPLAUSE trial looked at exactly this: 443 patients with biopsy-proven IgA nephropathy were enrolled, of whom 222 received 200 mg of iptacopan twice daily, and the remaining 221 received placebo. Of note, >98% of patients were receiving ACE inhibitor or ARB therapy, and approximately 13% were taking SGLT2 inhibitors. Individuals were followed for 9 months, at which point the study revealed that proteinuria levels were significantly lower in those receiving iptacopan, by 38% (p<0.001). Also noted was that there was no significant difference in adverse events between the two groups.
Iptacocan received accelerated approval by the FDA in 2024 for the treatment of IgA nephropathy. So what do we need to know about how to use this medication safely?
Importantly, the inhibition of complement significantly increases one’s risk of infection, especially by encapsulated bacteria. This is due to the crucial role our complement system plays in fighting off these infections: the membrane attack complex (a terminal product of complement activation) directly lyses bacteria, and complement proteins C3b and C4b act as tags to opsonize bacterial surfaces for recognition and clearance by phagocytic cells. Without these intact mechanisms in place, the risk for severe infection increases. To this end, patients receiving iptacopan should complete or update their vaccinations against Streptococcus pneumoniae and Neisseria meningitidis, ideally at least 2 weeks before their first dose of the medication. If treatment is felt to be more urgent and cannot be delayed, patients should take prophylactic antibiotics while starting their treatment and completing the necessary vaccine series. There is an increased risk for haemophilus influenzae type B (Hib) infection as well; however, current guidelines do not recommend repeat vaccination.
All in all, the complement studies are growing. And several ongoing trials are starting to evaluate the efficacy of additional complement-targeting medications such as pegcetacoplan (a C3 inhibitor) and ravulizumab (a long-acting C5 inhibitor similar to eculizumab), among others. This is, and will continue to be, an exciting area to watch for future developments.
With our growing armamentarium for the treatment of IgA nephropathy, additional data will certainly be needed to help drive decision-making and therapeutic selection. Initially this will likely include more subgroup analyses, and eventually give way to larger, targeted randomized trials.
And who knows, maybe next year IgA nephropathy will make yet another appearance in NephMadness 2027. Same place, same time?
– Executive Team Members for this region: Matt Sparks @Nephro_Sparks – @nephrosparks.bsky.social and Dia Waguespack @d_r_waguespack | Meet the Gamemakers
How to Claim CME and MOC
US-based physicians can earn 1.0 CME credit and 1.0 MOC per region through NKF PERC (detailed instructions here). The CME and MOC activity will expire on June 1, 2026.







Leave a Reply