#NephMadness 2023: IgA Nephropathy Region
Submit your picks! | NephMadness 2023 | #NephMadness | #IgAN
Selection Committee Member: Dana Rizk @DanaRizk1
Dana Rizk is Professor of Medicine in the Division of Nephrology at the University of Alabama at Birmingham. Her main clinical and research interests revolve around IgA Nephropathy focusing on multi-disciplinary translational projects of diagnostic and prognostic disease biomarkers. She is also site principal investigator on many IgAN clinical trials.
Writer: Dana Larsen @dana_m_larsen
Dana Larsen is a nephrologist at University of California, San Francisco, after completing her fellowship there while earning her Master of Arts in Education from the University of California, Berkeley, in 2022. Her research focuses on understanding how learners are engaging with Free Open Access Medical education (FOAMed) resources. She is a 2022 graduate of NSMC.
Competitors for the IgA Nephropathy Region
Team 1: Non-immunosuppressive Therapy in IgA Nephropathy versus Team 2: Supportive Care with Immunosuppression
Copyright: Ethan Daniels/Shutterstock
“Put me in, coach!” – Immunosuppression as a playmaker for the treatment of IgA nephropathy
This bracket highlights an age-old rivalry, like the ones between Indiana and Purdue or Duke and North Carolina. In its long history, each side has held the advantage at times, so this year’s match is sure to be a close one. The players are on the court; now it’s time for tip-off.
Overview
IgA nephropathy is the most common cause of primary glomerulonephritis worldwide (obligatory opening sentence). Peak incidence occurs in the second and third decade of life with a 2:1 male-to-female predominance in the United States versus a 1:1 sex distribution in Asia. While it was once considered “benign hematuria,” IgA nephropathy is now recognized as a major contributor to the global kidney health burden with 15%-20% reaching kidney failure within 10 years of kidney biopsy diagnosis and 20%-40% doing so within 20 years.
Defining the pathogenesis of IgA nephropathy is important for understanding the potential role of immunosuppression and supportive care measures. It is thought to comprise 4 key elements in a multi-hit process (Figure below).
The etiology of primary IgA nephropathy is still being elucidated, with current research seeking to identify triggers from amongst broad potential factors: infection, environment, etc. The disease is now identified as an autoimmune process that occurs in the genetically predisposed host. IgA nephropathy has also been associated with various clinical diseases including cirrhosis, inflammatory bowel disease, psoriasis, Sjogren syndrome, and HIV infection, and in those instances is referred to as secondary IgA nephropathy. IgA nephropathy can also occur in the context of systemic IgA vasculitis (previously known as Henoch Schonlein purpura) with dermal, gastrointestinal, and articular manifestations. While IgA vasculitis is more common in children, it tends to be a self-limiting disease when compared to adult onset.
The clinical presentation of IgA nephropathy is highly variable. Adults generally present with proteinuria, microscopic hematuria, hypertension, and elevated serum creatinine, whereas children and young adults most commonly present with macroscopic hematuria during an upper respiratory tract infection (“synpharyngitic hematuria”). Less than 10% of patients present with rapidly progressive glomerulonephritis (RPGN) or nephrotic syndrome. In IgA vasculitis with nephritis, well-described extra-kidney manifestations include palpable purpura, arthritis, and abdominal pain. Definitive diagnosis of IgA nephropathy requires a kidney biopsy. The hallmark of IgA nephropathy is IgA-dominant deposits in the glomerular mesangium, though it commonly also presents with C3 positivity (about 90% of cases) and IgG (up to 60%). Pathology specimens in IgA nephropathy and IgA vasculitis with nephritis are indistinguishable.
Stratification of relative risk for disease progression in patients with IgA nephropathy is crucial in management decisions. Clinical stratification can be determined at time of biopsy via the International IgA Nephropathy Prediction Tool (IIgAN-PT), which calculates the five-year risk of a 50% decline in eGFR or progression to kidney failure using the following information:
- There is a decreased risk of progression with < 1g/day proteinuria and perhaps even greater risk reduction for those with <0.5g/d
- Hypertension > 140/90 mmHg is predictive of a worse outcome
- Decreased eGFR at the time of diagnosis and during the course of disease is predictive of worse outcomes
- Pacific Asian origin has been associated with an increased risk of end-stage renal disease (adjusted HR 1.56)
- Histologic features are evaluated using the Oxford classification system
The Oxford classification system was developed in 2009 by the International IgA Nephropathy Network. Aiming to standardize IgA nephropathy grading and provide histological risk stratification to accompany clinical risk, the Oxford score initially comprised 4 key categories: mesangial hypercellularity, endocapillary hypercellularity, segmental glomerulosclerosis, and tubular atrophy and interstitial fibrosis (MEST). Each has independently been validated to be predictive of kidney outcomes in independent studies, though the largest validation trial (the VALIGA cohort) did not confirm endocapillary hypercellularity as an independent risk factor for progression. The impact of crescents (C) remains challenging in this classification system due to the exclusion of RPGN from the original classification development cohort. By combining four large cohorts retrospectively, however, the presence of crescents was found also to be a significant and independent predictor of kidney failure risk. The overall metric is validated in both adults and children with a minimum requirement of an adequate biopsy specimen of 8 glomeruli. Patients with eGFR <30 mL/min/1.73 m2 were excluded and the primary outcome of risk stratification was progression to kidney failure or a decrease in eGFR by more than 50%.
Treatment goals in all patients with IgA nephropathy are to slow down eGFR loss. Higher levels of proteinuria have been associated with more rapid progression of kidney disease in patients with IgA nephropathy, and reduction of proteinuria has been shown to be protective. In clinical practice, a resolution in microscopic hematuria is also targeted, though the use of this as a marker for treatment efficacy is unproven.
The opponents in this matchup both start the game with their supportive therapy treatments on the court. The decision on whether (and when) to bring immunosuppression off the bench and the amount of playtime allotted will be critical in determining the winner of this matchup.

Copyright: Dariia Belkina/Shutterstock
Team 1: Non-immunosuppressive Therapy in IgA Nephropathy
Supportive therapy has been the tried-and-true treatment for IgA nephropathy, with excellent data to support its use. Summarized in the table below, these treatments and supporting data highlight just how integral these agents are for IgA nephropathy treatment.
Summary of supportive care treatments for IgA nephropathy:
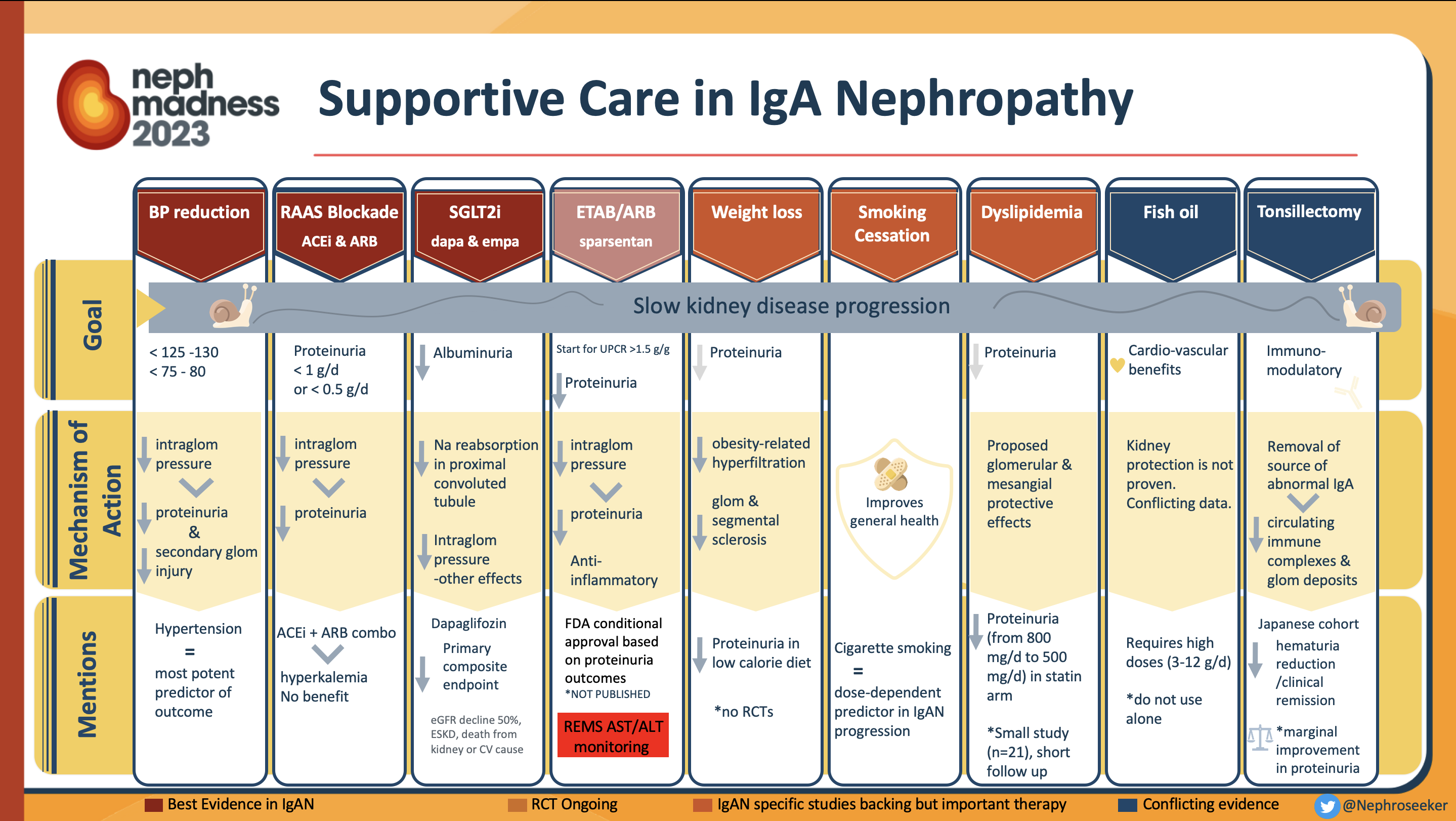
As the backbone for treatment in all patients with IgA nephropathy, optimized supportive care was considered the ultimate victor in the STOP-IgAN trial, which assessed supportive care with and without the addition of steroids. During the 6 month run-in phase, all patients received comprehensive supportive care including renin-angiotensin system (RAS) blockade to a target blood pressure less than 125/75 mm Hg. Approximately one-third of participants (94/309) achieved remission and were no longer eligible for enrollment, thus showing the power of supportive care in this disease process. You read that right, 30%! Furthermore, patients in the supportive care arm of the trial had an annual eGFR decline of only 1.6 mL/min/1.73 m2, which was about the same as in the immunosuppression arm split into two groups.
- Those with eGFR >60 mL/min/1.73 m2 received intravenous methylprednisolone with oral prednisone alone (average eGFR 73 mL/min/1.73 m2)
- Those with eGFR 30-59 mL/min/1.73 m2 received prednisone with cyclophosphamide followed by azathioprine (average eGFR 36 mL/min/1.73 m2)
Notably, the overall rate of eGFR decline in either group was significantly lower when compared with prior trials in IgA nephropathy, and was thought to be related to aggressive supportive care in this trial.
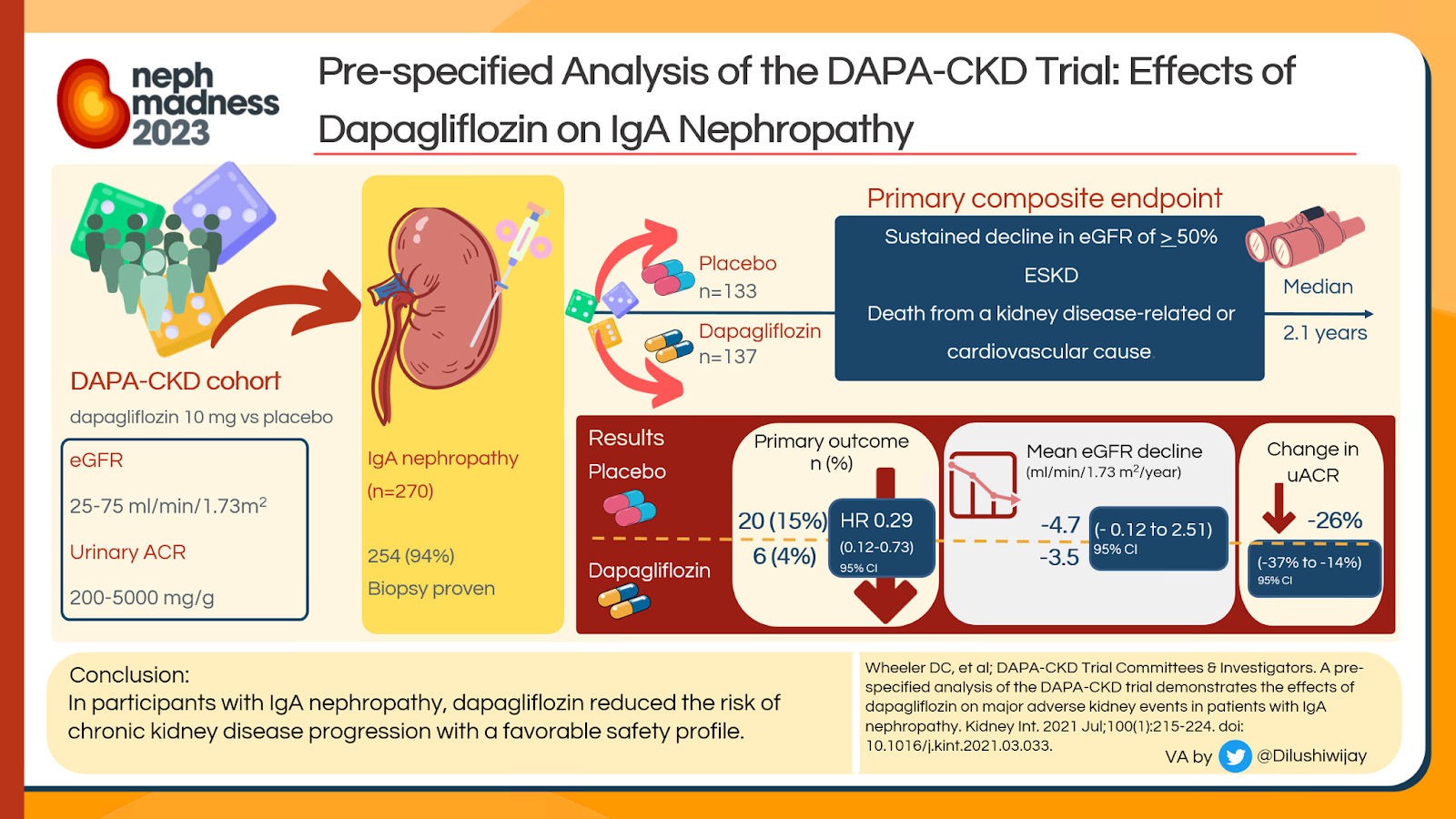
There are several novel agents recently or currently under investigation in addition to this all-star supportive therapy of RAS blockade and blood pressure control. We’ll start with the recently published data on sodium-glucose co-transporter 2 (SGLT2) inhibitors. DAPA-CKD, published in October 2020, enrolled 270 patients (of 4,303 total) with IgA nephropathy (diagnosed by kidney biopsy in 94%) both with and without diabetes. The eGFR was 25-75 mL/min/1.73 m2 and urine albumin-creatinine ratio (UACR) 200-5000 mg/g for those enrolled. Overall, treatment with the SGLT2 inhibitor dapagliflozin was associated with a significant reduction in the primary composite endpoint of 50% reduction in eGFR, kidney failure, or death. In a pre-specified analysis of DAPA-CKD, patients with IgA nephropathy treated with dapagliflozin had decreased in UACR by 26%, mean rates of eGFR decline (-3.5 mL/min/1.73 m2 in dapagliflozin vs -4.7 mL/min/1.73 m2 in controls), and decreased primary outcome events (HR 0.29, CI 0.12-0.73) compared to those treated with placebo. As of November 2022, EMPA-KIDNEY became the largest randomized control trial of patients with IgA nephropathy. Published in a systematic review and meta-analysis in the Lancet, SGLT2 inhibitors were found to decrease the risk of kidney disease progression by 40% (95% CI 0.46-0.78) across patients with any type of glomerular disease. No heterogeneity was identified amongst those with IgA nephropathy, focal segmental glomerulosclerosis, or other glomerulonephritis.
Additional novel agents, endothelin receptor inhibitors, are currently under investigation. These agents have been proposed to decrease kidney cell injury and fibrosis in experimental models and reduce proteinuria in addition to maximal renin-angiotensin system blockade, though known side effects of sodium retention and heart failure have resulted in limited use. Following on the heels of SONAR, atrasentan, a selective endothelin A receptor antagonist, is currently being investigated as a treatment for IgAN in a phase 3 trial, Atrasentan in Patients with IgA Nephropathy (ALIGN). A multicenter randomized controlled trial (RCT), ALIGN seeks to assess change in proteinuria in 320 patients after 24 weeks and change in eGFR after 2.6 years of study drug versus placebo. Uniquely, it is enrolling additional subjects currently on stable doses of SGLT2 inhibitors for secondary analysis. ALIGN is anticipated to conclude in December 2025. Additionally, the Effect and Safety of Sparsentan in the Treatment of Patients With IgA Nephropathy (PROTECT) trial compares sparsentan (a novel dual-acting angiotensin receptor blocker and endothelin type A receptor antagonist) with irbesartan using a primary outcome of change in 24-hour urine protein to creatinine ratio (UPCR) after 36 weeks. PROTECT aims to enroll 380 patients with biopsy-proven IgA nephropathy considered at high risk for progression with eGFR greater than 30 and greater than 1g/d proteinuria who have already had 12 weeks of RAS blockade, with results anticipated in August 2023. On February 17, 2023, the FDA granted conditional approval for the use of sparsentan in patients with IgAN at high risk of disease progression with UPCR≥ 1.5 g/g despite optimized medical care.
Sparsentan (FILSPARI) is a once-a-day oral medication that acts as a dual endothelin and angiotensin II receptor antagonist. In the phase 3 PROTECT trial, sparsentan was associated with 49.8% reduction in proteinuria from baseline at 36 weeks compared to 15.1% in the Irbesartan comparator arm. The conditional approval was based on these primary outcome results. Because of the risk of hepatotoxicity with endothelin receptor antagonists, FDA required FILSPARI-treated patients to be enrolled in REMS program for monthly liver function testing for the first year of therapy and then quarterly thereafter.
As exciting as all these existing and emerging therapies are, we must give due respect to our opponent by further examining immunosuppression in IgA nephropathy. Before doing so, however, we give some last considerations, specifically reasons to avoid immunosuppression in the treatment of IgA nephropathy:
- Immunosuppressives have well-known side effects to avoid – Complications from glucocorticoid therapy are well-described and include the following: infection, diabetes and hypertension exacerbation, osteoporosis, avascular necrosis, peptic ulcers, hyperlipidemia, myopathy, cataracts and suppression of the HPA axis.
- Use of immunosuppression does not predict outcomes – The combination of MEST and cross-sectional clinical data at biopsy was able to predict adverse outcomes independent of treatment with immunosuppression.
- IgA nephropathy still recurs during transplant – A 23% risk of recurrence of IgA nephropathy in kidney transplant recipients has been reported despite high doses of immunosuppressives aimed to prevent this.
- Immunosuppression has a poor track record – Several immunosuppressive agents, including rituximab and CNIs, have failed to show any benefit in IgA nephropathy despite their widespread use in other immune-mediated kidney diseases.
Perhaps most convincingly, in its 2012 clinical practice guidelines on glomerulonephropathy, KDIGO recommended a trial of steroid therapy for those patients with IgA nephropathy and persistent proteinuria greater than 1 g/day despite 3 months of optimized supportive care. The revised 2021 KDIGO guidelines now recommend inclusion in a clinical trial if possible prior to consideration of glucocorticoid therapy which “may reduce the risk of kidney failure” due to “uncertainty over the safety and efficacy of existing immunosuppressive treatment choices.” The evidence for supportive care is strong, and with new therapies continuing to be developed and evaluated, there are numerous arguments for avoiding immunosuppression and its known risks in the treatment of IgA nephropathy. This team leaves it all on the court.
Check out this podcast episode of ISN Global Kidney Care featuring Roberto Pecoits-Filho, Dana Rizk, and Dana Larsen:
Discussion on IgA Nephropathy
COMMENTARY BY SHIKHA WADHWANI:
Prediction: IgA Nephropathy the Favorite to Win

Copyright: khuncho24/Shutterstock
Team 2: Supportive Care with Immunosuppression
“You miss 100% of the shots you don’t take.” – Wayne Gretzky
While acknowledging the benefit of supportive care for all with IgA nephropathy, ongoing research into immunosuppressive therapies may hold the key to improved outcomes for this heterogeneous disease.
The therapeutic Evaluation of Steroids in IgA Nephropathy Global (TESTING) study followed the STOP-IgAN trial (as discussed in the supportive treatment section above) and provides substantial grounds for the use of corticosteroids, after study modifications. This investigator-initiated study, funded by government agencies in Australia and China, also began with a 3-month run-in of maximally tolerated RAAS blockade. Those patients with persistent proteinuria greater than 1g/d and GFR 20-120 mL/min/1.73 m2 were eligible for study enrollment. The initial study, TESTING High Dose (2017), was terminated due to significantly increased risk of serious adverse events in the steroid arm, specifically infections (11 vs 0 with two deaths). At the time of study discontinuation, an early analysis performed on 262 patients after 2.1 years identified potential early efficacy for steroids with a decreased composite outcome of kidney failure, death, or 40% loss of eGFR (15.9% vs 5.9%; HR 0.37). A revised protocol, TESTING Low Dose (combined), raised the lower limit of GFR inclusion to 30 mL/min/1.73 m2, lowered the dose of steroids (a reduction in methylprednisolone from initial 0.6-0.8 mg/kg/d to 0.4 mg/kg/d), and included the routine use of Pneumocystis jirovecii (PJP) prophylaxis. The trial resumed with the new protocol and was completed in May 2022. At the completion of the revised protocol, the combined results were statistically powered to show significance in the primary endpoint a composite of 40% eGFR decline, kidney failure (by dialysis or transplant), or death due to kidney disease over the 4.2-year follow-up. The steroid arm did better with respect to the primary outcome, with a HR of 0.58 (95% CI, 0.41-0.81) for full-dose and 0.27 (95% CI, 0.11-0.65) for reduced-dose methylprednisolone. The authors did, however, concede a significant increase of serious adverse events in the steroid arm, mainly with high-dose therapy.
Why different outcomes between TESTING and STOP-IgAN? Potential explanations for differences have been hypothesized to be secondary to underlying patient characteristics and baseline eGFR decline. Notably, the rate of eGFR decline in the supportive care arm of both studies was considerably different, with a mean yearly eGFR loss of 1.6 mL/min/1.73 m2 in STOP-IgAN vs 6.95 mL/min/1.73 m2 in TESTING which may have contributed to the lack of benefit in the steroid arm of STOP-IgAN. Given that participants in TESTING entered with greater baseline proteinuria (2.5 g/day in TESTING vs 1.8 g/day in STOP-IgAN), the reduction in proteinuria between these studies highlights the importance of this marker for kidney function preservation and suggests that steroids may be important for kidney preservation for those with greater than 2 g/day proteinuria.
Additionally, STOP-IgAN showed some benefit with regard to immunosuppression. Despite no significant difference in the proportion of patients with a reduction in their eGFR greater than 15 mL/min/1.73 m2 after 3 years, there were more patients in the steroid arm who met full clinical remission criteria, defined as UPCR less than 200 mg/g and stable eGFR less than 5 mL/min/1.73 m2 from baseline (17% vs 5% p=0.01). As these were not primary outcomes, the clinical significance of this has yet to be determined.
Whether you’re sold on steroids for IgA nephropathy or not, team immunosuppression has a deep bench of supporting players. Additional immunosuppressive agents have and are currently undergoing further investigation, giving this team the chance to pull off an upset win.
In an attempt to decrease steroid dose and thereby decrease side effects while still realizing immunosuppressive benefits, mycophenolate mofetil (MMF) has been tried as a treatment for IgA nephropathy. The 2023 MAIN trial was an RCT of 170 patients randomized to either MMF (for 1.5 years) in addition to optimized supportive care or optimized supportive care alone. It found a decrease in the primary composite outcome (doubling of serum creatinine, ESKD, or death due to cardiovascular cause). Secondary outcomes also demonstrated a reduction in the progression of chronic kidney disease, defined as mean eGFR loss without an increase in serious adverse events. Prior to this study, only three prospective trials had evaluated the benefit of MMF vs placebo, with only the largest of those showing a potential role for MMF as a steroid-reducing agent. While no difference in proteinuria reduction at 6 and 12 months was seen when MMF was combined with steroids for induction therapy, fewer adverse events (such as diabetes) were noted.
Other strategies are being developed in order to decrease the adverse risk of steroids in IgA nephropathy treatment. A notable one is the targeted-release steroidal formulation (TRF) of budesonide. This formulation is supposedly aimed at the Peyer patches of the distal ileum where B cells are primed to produce abnormally glycosylated Gd-IgA1. TRF-budesonide seeks to intensify steroid exposure at this site while limiting systemic exposure. A phase 2b double-blinded RCT of 16 mg vs 8 mg of TRF-budesonide vs placebo, NEFIGAN, enrolled patients with persistent proteinuria greater than 0.75 g/day or UPCR > 500 mg/g and eGFR ≥ 45 mL/min/1.73 m2 after a 6-month run-in of RAS inhibition, and found that those receiving budesonide had a 24% decrease in UPCR compared with 3% decrease in placebo group, as well as stable eGFR vs 9.8% reduction in eGFR in the placebo group. Furthermore, no serious infections were reported. As also discussed by NephJC, the follow-up phase 3 trial, NefIgArd, using the 16 mg TRF-budesonide dose, confirmed these findings from the 2b trial with 199 patients with 27% reduction (absolute change from baseline 221 mg/g) in UPCR at 9 months and 3.87 mL/min/1.73 m2 difference vs placebo in eGFR preservation. This has led to conditional approval by the Food and Drug Administration in the United States and European Medicines Agency in Europe of TDF-budesonide (under brand names Tarpeyo and Kinpeygo, respectively). The final results of the trial on eGFR outcome at 12 months follow-up are still being awaited. Adverse events were similar across all treatment groups. Notably, both 16 mg TRF-budesonide and placebo had two serious treatment-emergent adverse events recorded by blinded investigators—DVT and progressive kidney disease in the TRF-budesonide group and increased proteinuria and progressive kidney disease in the placebo group. Classic steroid-associated side effects of weight gain, hypertension, and worsening of diabetes were not seen during the 12-month trial period, though patient-solicited corticosteroid-related symptoms (including swelling of the ankles, acne, striae, depression, and hair loss) were more common in the TRF-budesonide group and most patients who discontinued the treatment experienced corticosteroid-related adverse events.
Summary of immunosuppression agents for IgA nephropathy:

Several novel immunosuppressive agents are under investigation for IgA nephropathy treatment at this time. Leflunomide is an immunosuppressive agent currently used in rheumatoid arthritis and is thought to reduce glomerular deposition of autoantibodies and immune complexes. Currently, a phase 4 trial of leflunomide in addition to low-dose glucocorticosteroids and conservative treatment is ongoing. Additionally, multiple trials are underway investigating the role of complement inhibitors including narsoplimab (lectin pathway inhibitor), ipatacopan LNP023 (factor B inhibitor), cemdisiran (RNAi for C5), and avacopan (C5a receptor antagonist). In disrupting the formation of immune complexes by the alternative complement pathway, these agents were specifically developed to target the pathophysiologic mechanism of IgA nephropathy. Bortezomib, approved for multiple myeloma, is a proteasome inhibitor targeting plasma cells. It has been proposed to modulate IgA nephropathy by depleting antibodies produced by these plasma cells. Bortezomib recently completed a pilot study with 8 patients and is currently in a phase 4 trial with 11 patients. Finally, despite the failure of rituximab in mitigating B-cell dependent pathways for IgA nephropathy progression, two B-cell modulating therapies are currently being evaluated. VISIONARY, a phase 3 trial, seeks to evaluate the effect of sibeprenlimab, a monoclonal antibody against a proliferation-inducing ligand (APRIL) which is a proliferation-inducing ligand cytokine thought to be involved in B cell maturation. VISIONARY will add sibeprelimab to patients on max standard of care therapy to evaluate the impact on proteinuria and eGFR, and is anticipated to complete in 2026. Atacicept, a pharmacologic antagonist for both APRIL and the cytokines B lymphocyte stimulator (BLyS), aimed at reducing B cell activation and class switching, will be evaluated by ORIGIN (Atacicept in Subjects with IgA Nephropathy). Anticipated to complete 2024, ORIGIN will assess change in proteinuria after 36 weeks as well as galactose deficient (gd)-IgA1 levels.
These existing and ongoing trials supportive of immunosuppression for IgA nephropathy all focus on primary IgA nephropathy; less is known about other forms. For IgA nephropathy with apparent minimal change disease, there are convincing arguments that immunosuppression is beneficial, and KDIGO 2021 continues to recommend treatment with corticosteroids as well as steroid-sparing agents for long-term steroid dose reduction. Similarly, in patients with systemic IgA vasculitis also affecting the kidney, anecdotal reports have shown promise for the use of rituximab, though overall there is very little data to guide treatment of these IgA nephropathy-plus entities.
Additional arguments for immunosuppression:
- Kidney histology improves after steroid exposure – A repeat kidney biopsy study in 2002 included 35 patients with IgA nephropathy treated with combination tonsillectomy and steroid pulse with resultant remission in microscopic hematuria. They found improvement in all glomerular lesions excluding global sclerosis and tubulointerstitial markers.
- Retrospective analyses of those treated with steroids suggest improvement – 184 patients from the VALIGA cohort were found to have improved outcomes when treated with corticosteroids compared to patients with matched propensity scores. Outcomes were measured by ESKD progression, 50% decline in kidney function. and proteinuria.
- Existing trials exclude “high-risk” patients – Existing data must be interpreted for specific study populations only. The STOP-IgAN trial, for example, excluded patients with persistent proteinuria > 3.5 g/d. Improved risk stratification and incorporation of “high-risk” patients in trials may identify those patients in whom benefits might outweigh the risks.
With major 2022 publications for both opponents in this bracket, as well as several trials in the pipeline, this rivalry is heating up on the court, and whoever wins today may or may not be the clear victor next year. Should immunosuppression be employed, or are the developments in supportive care alone sufficient while avoiding treatment-related toxicities for IgA nephropathy?
– Executive Team Members for this region: Matt Sparks @Nephro_Sparks and Tim Yau @Maximal_Change
How to Claim CME and MOC
US-based physicians can earn 1.0 CME credit and 1.0 MOC per region through NKF PERC (detailed instructions here). The CME and MOC activity will expire on June 1, 2023.
Submit your picks! | #NephMadness | @NephMadness | #IgAN







Leave a Reply