#KidneyWk 2015: Long-Term Efficacy and Safety of ZS-9 in the Treatment of Hyperkalemia
Late Breaking Poster: Long-Term (52-Week) Efficacy and Safety of ZS-9 in the Treatment of Hyperkalemia: Interim Results (as of October 10, 2015) From a Phase 3 Open-Label, Multi-Center, Multi-Dose Maintenance Study
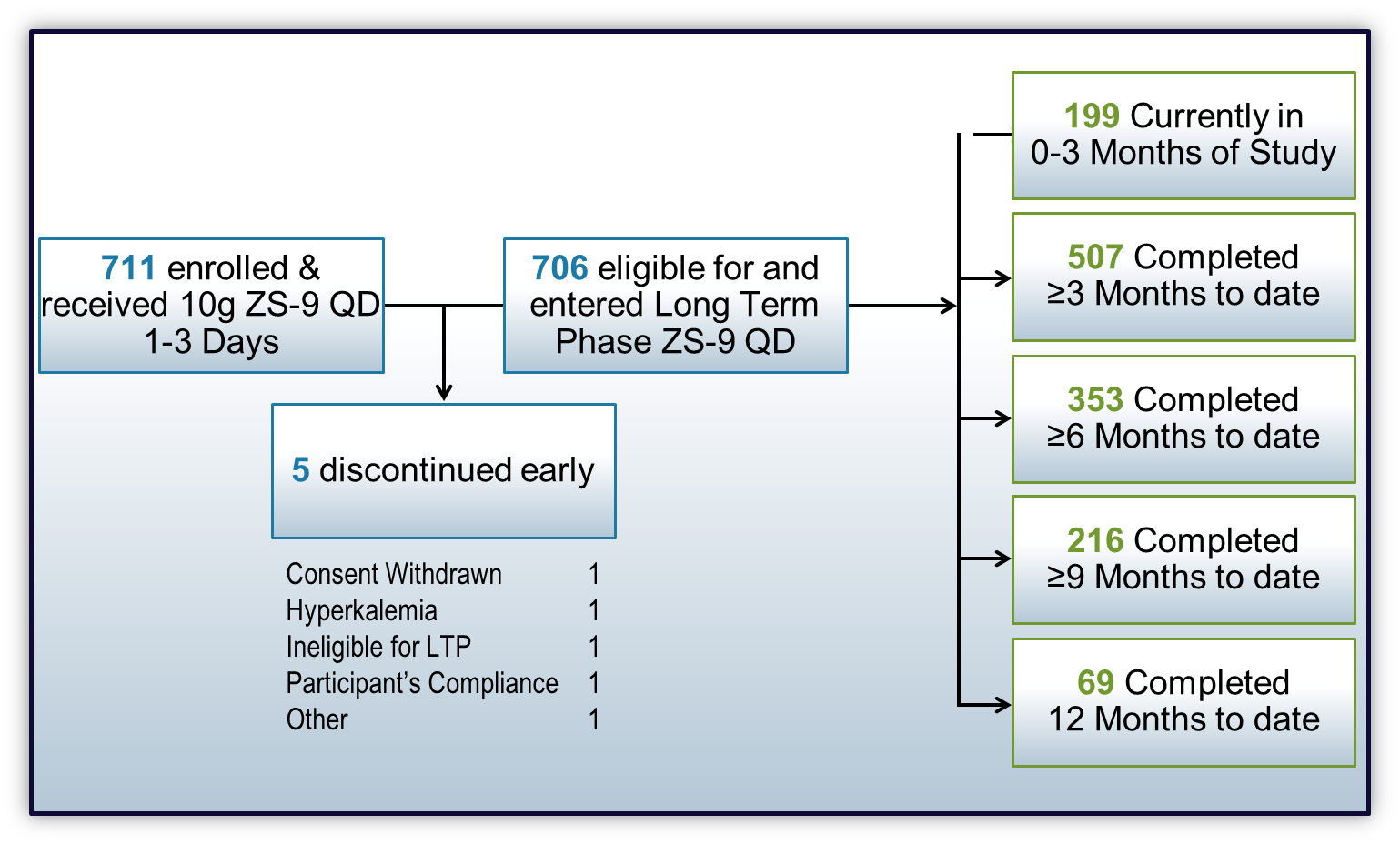
One of the late breaking posters was an interim analysis of the first 711 patient enrolled in the ZS005 Study. (Note: Dr. Lerma is one of the authors of this poster.) ZS005 is a global, multicenter trial designed to investigate the long-term safety and efficacy of ZS-9 in patients with hyperkalemia (potassium levels >5.0 mEq/L).
Patients with hyperkalemia are given ZS-9 10 g TID during the 24- to 72-hour Acute Phase of the study. Those who achieve normokalemia, are then given 5 g once daily with the ability to titrate dose in 5-g increments or decrements, if needed, to maintain normokalemia. The primary endpoint is safety and tolerability, while the secondary endpoint is the proportion of patients with average serum potassium ≤5.1 mEq/L between months 3-12.
Mean potassium levels at baseline in the study were 5.6 mEq/L with over 60% of patients having baseline potassium levels ≥5.5 mEq/L.


- 99% of patients achieved normokalemia in the Acute Phase on 10 g TID of ZS-9
- Mean potassium levels were maintained at 4.6 mEq/L throughout the 12-month Long Term Treatment Phase
- The primary efficacy endpoint was met with 87- 92% of patients maintained at an average serum potassium ≤5.1 mEq/L between month 3 and month 12
- Normokalemia was maintained with patients receiving once-daily 5 g (64%), 10 g (30%), or 15 g (5%) doses of ZS-9.
- There were no deaths in the Acute Phase; 5 deaths (0.7%) occurred in the Long Term Phase but none were considered to be related to study drug
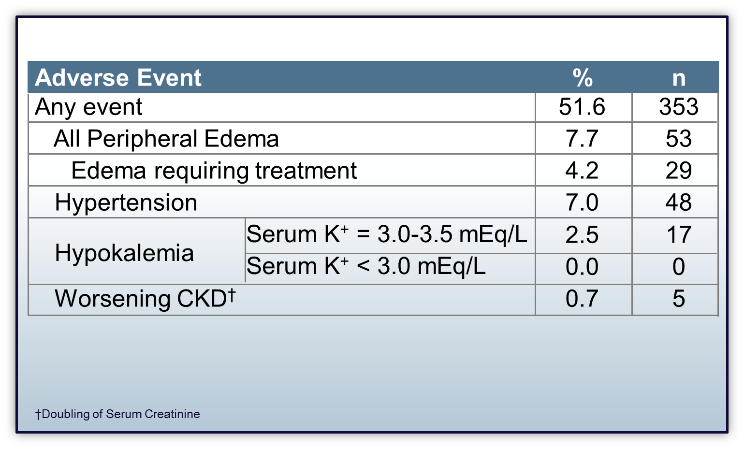
- The rate of peripheral edema was 53/684 (7.7%); only 29/684 (4.2%) patients required treatment with diuretics
- 26/29 (90%) patients that required treatment had prior history of edema
- Exposure corrected rates of edema:
- 0-6 months = 0.00057
- 6-12 months = 0.00015
- No significant change from baseline at 3, 6, 9 and 12 months in weight or blood pressure
- The rate of hypokalemia (serum K+ < 3.5 mEq/L) was 2.5%; all cases were mild
- There were no changes in serum Mg+2, Ca+2, and Na+
Conclusions
- In patients with hyperkalemia, ZS-9 rapidly normalized serum potassium levels and maintained serum potassium levels at 4.6 mEq/L with once daily treatment for up to 52 weeks, with >90% of patients maintained with 5 or 10 grams of ZS-9
- Improvement in metabolic acidosis was observed and consistent with previous studies
- Exposure corrected edema rates were consistent with HARMONIZE placebo group (0.00089) and do not increase with duration of ZS-9 therapy
- Rates of edema and hypertension were consistent with the patient population over this time frame
- There was a low rate of gastrointestinal (GI) adverse events (<5%) with ZS-9 treatment
- These findings are supportive of long-term use of daily ZS-9 for management of hyperkalemia.
ZS-9 is an insoluble, non-absorbed zirconium silicate with a clearly defined three-dimensional crystalline lattice structure that was designed to preferentially trap potassium ions. The potassium selectivity of ZS-9 enables high in-vitro binding capacity for potassium ions even in the presence of other competing ions. Two previous blogs have covered ZS-9 http://wp.me/p1IgOp-1j1 and http://wp.me/p1IgOp-1D9. Another related blog post: http://wp.me/p1IgOp-1Qs.
All images courtesy of and post written by Dr. Edgar Lerma, AJKD Blog Advisory Board member.
Check out all of AJKD Blog’s coverage of Kidney Week 2015!

Leave a Reply