How Does Aldosterone Activation Contribute to CKD?
Christopher Naranjo @NaranjoNephro
Dr. Christopher Naranjo is a Nephrology Fellow at the University of Miami. He is passionate about hypertension, glomerular diseases, and general medical education.
Efren Chavez Morales @efrenchm1
Efren Chavez, MD, MS is a nephrologist and hypertension specialist at University of Miami. He has an interest in hypertension and hereditary kidney diseases. His research is focused on evaluating treatment patterns, responses and long-term clinical outcomes of patients with hypertensive crisis.
In 2019, the prevalence of chronic kidney disease (CKD) worldwide was estimated to be 843.6 million cases with increased incidence among individuals with diabetes mellitus, hypertension, and the elderly. CKD is recognized as one of the leading causes of death worldwide. An epidemiological study published in the Lancet predicts that CKD will become the 5th most common cause of years of life lost worldwide by 2040. Risk factors such as albuminuria and reductions in estimated glomerular filtration rate (eGFR) have been associated with increased mortality that is independent of cardiovascular risk and therefore have become the target for intervention. New therapies are showing promise in delaying CKD progression. Recently, developments in the understanding of aldosterone receptor activation and the effect on progression of CKD has directed attention to the use of mineralocorticoid receptor antagonists (MRAs) as a potential therapy to slow CKD progression.
Mineralocorticoid receptor (MR) stimulation is fundamental to maintain sodium and potassium balance, and in the anatomic regulation of blood pressure and volume status. The major triggers for aldosterone release are hyperkalemia and stimulation of the renin-angiotensin-aldosterone system (RAAS) caused by volume depletion. The response to MR stimulation in principal cells of the distal tubule is shown in Figure 1. In In addition, there is incremented activity of NCC transporter in the distal convoluted tubules.
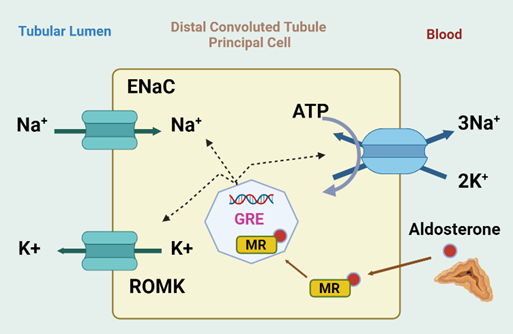
Figure 1. Effect of MR activation in distal nephron. In the kidneys, aldosterone activates the MR in the principal cells of the distal nephron which leads to increased Na+ reabsorption and K+ excretion. The aldosterone-MR complex binds to glucocorticoid-responsive elements (GRE) that activate the apical epithelial sodium channel (ENaC) and the basolateral Na+-K+ ATPase by different regulatory pathways. In response to hyperkalemia, the aldosterone-mediated Na reabsorption via ENaC drives K secretion via ROMK. Created with BioRender.com.
Other tissues also express the MR, such as cardiomyocytes, vascular cells, podocytes, fibroblasts and inflammatory cells. Therefore, these are also responsive to aldosterone signaling.
Over the past decade, emerging data has linked aldosterone and MR activation with increased risk of renal and cardiovascular injury, including progression of CKD, coronary artery disease, stroke, heart failure, atrial fibrillation, arterial stiffness and the metabolic syndrome. In the kidney, MR activation in non-epithelial cells leads to the activation of inflammatory and fibrotic pathways and has deleterious effects on podocytes and mesangial cells. In experimental models of CKD in T2DM, pharmacological inhibition of the MR reduced albuminuria, kidney fibrosis, glomerular lesions and inflammation. Clinical studies have shown that MRAs have an anti-albuminuric effect in diabetic kidney disease (DKD) and are associated with a decrease in adverse cardiovascular outcomes. However, incidence of hyperkalemia among patients taking MRA has limited the use of these agents over the long term, especially among inexperienced providers.
In 2006, Epstein et al showed that the addition of low doses of eplerenone (50-100 mg/d) to enalapril significantly reduced albuminuria in patients with type 2 diabetes mellitus (T2DM). By week 12, UACR was reduced by 7.4% in the placebo group, by 41% in the group on 50 mg of eplerenone and by 48.4% in the group on 100 mg of eplerenone. Mehdi et al administered spironolactone to patients with type 2 diabetes mellitus and microalbuminuria who were already taking lisinopril. This drug lead to a greater reduction of proteinuria compared to placebo.
A systematic review of eight studies in adult patients with diabetic nephropathy secondary to either type 1 or 2 diabetes mellitus who were taking standard of care treatment with an angiotensin converting enzyme inhibitor (ACEI) or an angiotensin II receptor blocker (ARB) showed that adding an MRA (spironolactone or eplerenone) as an adjunct therapy further decreased albuminuria by 23-61% compared to RAAS blockade monotherapy. However, the proportion of patients who discontinued treatment due to hyperkalemia was 17% in the MRA group versus 5% in the placebo group.
The newer non-steroidal MRAs have similar efficacy but a better safety profile and a decreased risk of hyperkalemia compared to steroidal MRAs. Finerenone demonstrates higher selectivity to MR, has a shorter half-life with no active metabolites, no significant central nervous system (CNS) penetration, and no sexual side effects when compared with spironolactone and eplerenone (Table 1).
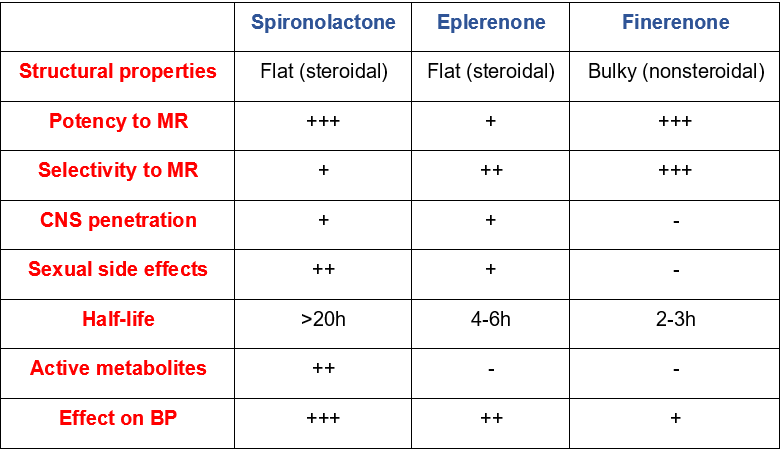
Table 1. Pharmacologic characteristics of spironolactone, eplerenone and finerenone. Adapted from Kintscher et al. under the terms of the Creative Commons Attribution License (CC BY 4.0) (https://creativecommons.org/licenses/by/4.0/)
The Finerenone in Reducing Kidney Failure and Disease Progression in Diabetic Kidney Disease (FIDELIO-DKD) and Finerenone in Reducing CV Mortality and Morbidity in Diabetic Kidney Disease (FIGARO-DKD) studies have assessed the impact of MRAs on clinical outcomes.
The FIDELIO-DKD study included 5,734 patients with DM2 and CKD with albuminuria randomized to get either finerenone or placebo. Finerenone decreased the primary outcome (renal events) by 18% and secondary outcome (CV events) by 14% compared to placebo after a median follow up of 2.6 years. The primary outcome was a composite of incident end stage kidney disease, continuous decline of ≥40% of basal eGFR or kidney-related deaths. The secondary outcome was a composite of incident CV-related deaths, non-lethal myocardial infarction or stroke, and heart failure-related hospitalizations.
The FIGARO-DKD study included 7,437 patients with DM2 and CKD with albuminuria randomized to get either finerenone or placebo. Finerenone reduced the primary outcome by 13% compared to placebo, which was a composite of incident death, non-lethal myocardial infarction or stroke and heart-failure related hospitalization.
Incidence of hyperkalemia for finerenone versys placebo groups in FIDELIO-DKD and FIGARO-DKD trials are 18.3% and 10.8% versus 9% and 5.3% respectively.
A pooled analysis which followed the publication of FIDELIO and FIGARO demonstrated that the primary contribution to cardiovascular benefit from Finerenone comes from the prevention in hospitalizations related to heart failure in patients with T2DM and CKD. The success of FIDELIO and FIGARO cannot be understated. Collectively the evidence towards cardiovascular and kidney protection was observed as early as 1 and 12 months, respectively, after initiation of Finerenone and persisted for the 3-year duration of the trials, which has led to drug approval by the FDA. Furthermore, when comparing Finerenone and spironolactone in patients with HFrEF and CKD, the degree and incidence of hyperkalemia was less among patients taking Finerenone.
Given the current available data, the risk-to-benefit ratio regarding the use of non-steroidal MRA has become noteworthy. These drugs confer significant renal and cardiovascular protection. The risk of hyperkalemia persists with the use of non-steroidal MRAs, but it may be lower compared to steroidal MRAs. Other strategies to decrease the risk of hyperkalemia, such as dietary counseling, avoidance of polypharmacy and the use of potassium binders are of paramount importance. In the near future, the results of FIND-CKD will shed light on the effect of Finerenone on non-diabetic CKD with albuminuria. The CONFIDENCE trial will compare the effectiveness of either Finerenone or empagliflozin alone and in combination versus placebo on reduction of albuminuria in patients with CKD and T2DM. We have gained an additional tool to combat CKD progression. As our understanding develops, our patients truly stand to benefit from this shift in treatment paradigm.
–Post prepared by Christopher Naranjo @NaranjoNephro and Efren Chavez Morales @efrenchm1
To view Epstein et al (FREE), please visit AJKD.org.
Title: Aldosterone, Mineralocorticoid Receptor Activation, and CKD: A Review of Evolving Treatment Paradigms
Authors: Murray Epstein, Csaba P. Kovesdy, Catherine M. Clase, Manish M. Sood, and Roberto Pecoits-Filho
DOI: 10.1053/j.ajkd.2022.04.016


Leave a Reply