#NephMadness 2026: Cerebronephrology Region
Submit your picks! | @NephMadness | @nephmadness.bsky.social | NephMadness 2026
Selection Committee Member: David Drew
Dr. David Drew is a Nephrologist and an Associate Professor of Medicine at Tufts University School of Medicine. He has been with the Division of Nephrology at Tufts Medical Center since 2011, joining the faculty in 2015. Dr. Drew has a longstanding research interest in the intersection of kidney disease and cognitive impairment, having published multiple manuscripts and invited reviews regarding kidney disease related cognitive impairment.
Writer: Colton Jensen
Colton Jensen is a second-year nephrology fellow at Johns Hopkins University. He completed his medical degree at the University of Iowa Carver College of Medicine and internal medicine residency at the University of Vermont. His professional interests include medical education, home dialysis, and kidney stones.
Competitors for the Cerebronephrology Region
The brain and kidneys may be far apart anatomically, but they share fragile microvasculature and tight autoregulation. This region dives into the Brain–Kidney Axis, where CKD, kidney failure, and dialysis intersect with cognition, cerebrovascular disease, and structural brain injury.
Team 1: Cerebronephrology in CKD
versus
Team 2: Cerebronephrology in ESKD

Image generated by Matthew Sparks using ChatGPT at http://chat.openai.com, February 2026. After using the tool to generate the image, Sparks and the NephMadness Executive Team reviewed and take full responsibility for the final graphic image.
Introduction
The brain and the kidneys are separated by numerous organs: the heart, the lungs, and the liver are positioned between them. Despite the physical distance, the kidneys and the brain are more closely linked than realized. The two organs share very similar physiologic characteristics and a decline in one can lead to a decline in the other. The relationship between the two has been coined the “Brain-Kidney Axis”. Both organs share a low vascular resistance system and are able to handle large volumes of blood flow. They are also able to maintain consistent blood flow across a large range of blood pressure through autoregulation. Additionally, chronic kidney disease can lead to cerebrovascular disease, disruptions in the blood brain barrier, and expose the brain to uremic metabolites. Conversely, brain injury can expose the kidneys to increasing sympathetic tone and can impact sodium and water balance. With this complex bidirectional relationship, kidney disease is associated with negative impacts on the brain, and the focus of this region is the relationship between chronic kidney disease (CKD) and cognitive impairment as well as the impact of end-stage kidney disease (ESKD) and kidney replacement therapy on the brain.
Hosted by Dr. Sam Kant, this Kidney Commute: Pit Stop episode explores the emerging concept of the brain–kidney axis. Drs. David Drew and Colton Jensen examine how chronic kidney disease and ESKD with dialysis contribute to cognitive impairment—an underrecognized complication affecting a large proportion of patients. From early microvascular injury, albuminuria, and hypertension in CKD to cerebral hypoperfusion, ischemia, and structural brain changes during dialysis, this discussion highlights the bidirectional relationship between kidney and brain health. Join us as we unpack where brain injury begins and what this means for earlier recognition and improved patient care.
Pitstop: NephMadness 2026: Cerebronephrology Region: Exploring the Intersection Between Kidney Disease and Brain Health
COMMENTARY BY DONALD MOLONY:
The Case for Intense Focus on the impact of CKD on the Brain
Team 1: Cerebronephrology in CKD
Cognition often slips quietly in CKD. This group explores how declining eGFR, albuminuria, hypertension, and microvascular disease drive early cognitive impairment—especially in executive function, attention, and memory—long before patients reach kidney failure.

Copyright: Eric Isselee/ Shutterstock
CKD affects roughly 1 in 7 adults in the US and impacts 34% of US adults 65 years or older. Approximately 32% of US adults 65 years or older have some degree of mild cognitive impairment or dementia. Nephrologists manage numerous complications of CKD with their patients: electrolytes, hypertension, anemia, bone mineral disease, hypervolemia, etc. but there are a significant number of patients that also have some degree of cognitive impairment that may go unnoticed!
Cognitive impairment refers to deficits in one or more core cognitive domains, including memory, learning, attention, and executive function. It exists along a spectrum; from mild cognitive impairment to more advanced, functionally limiting disease typically classified as dementia. Cognitive function is evaluated across distinct domains, such as executive function, memory, language, visuospatial ability, learning, and attention, each of which may be differentially affected in kidney disease.
The relationship between cognitive impairment and CKD is closer, and more clinically relevant, than many appreciate. In a cross-sectional study in patients 55 years or older with CKD and diabetes, 48% were found to have some degree of neurocognitive impairment. In another cross-sectional study, an analysis of the BRINK cohort found that 48% of patients with CKD had some degree of cognitive impairment. CKD is also one of the biggest risk factors for developing cognitive impairment. In a longitudinal 6-year study, CKD was the 3rd biggest risk factor for developing mild cognitive impairment (MCI) and dementia behind only stroke and the use of anxiolytics.
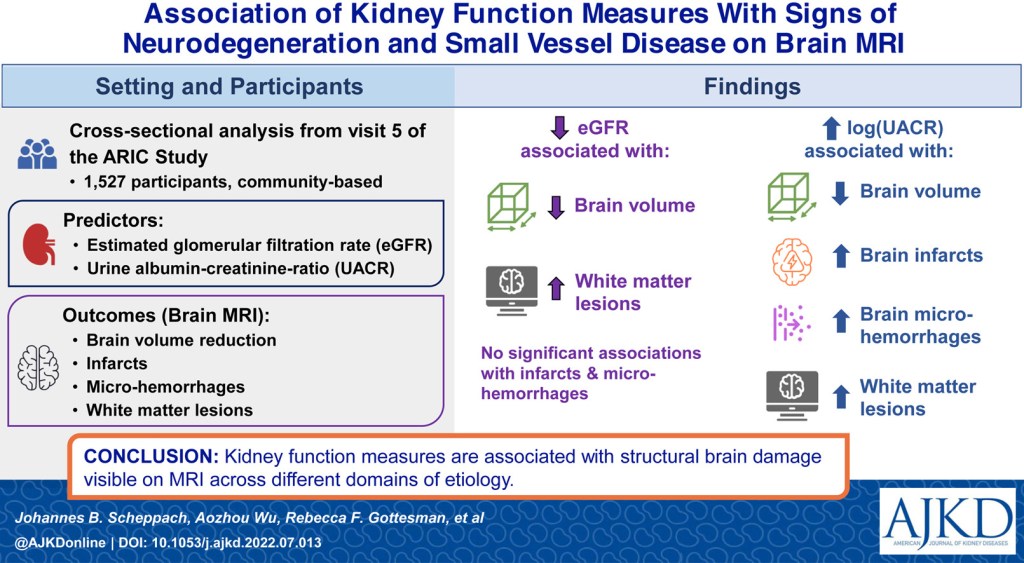
The risk of developing cognitive impairment in CKD has been shown to increase with lower eGFR, higher albuminuria, and higher systolic blood pressure. Within the Chronic Renal Insufficiency Cohort (CRIC), lower eGFR was associated with an increased risk of cognitive impairment. In one analysis, 17% of total patients in the study were found to have some degree of cognitive impairment. The risk of cognitive impairment increased with eGFR < 30 mL/min/1.73 m² compared to eGFR 45-59 mL/min/1.73 m² . This risk was attenuated after adjustment for hemoglobin levels. In the CRIC COG study, patients with more advanced CKD performed worse on cognitive testing compared to patients with milder CKD.In this cross-sectional study, participants with eGFR < 30 mL/min/1.73 m² had higher rates of clinically significant impairment in global cognition, naming, attention, executive function, and delayed memory compared with those with eGFR 45–59 mL/min/1.73 m², while category fluency was not significantly different. Finally, within The Health, Aging, and Body Composition cohort, the risk of developing cognitive impairment was higher in patients with CKD compared to patients without CKD and the risk increased with eGFR < 45 mL/min/1.73 m² compared to eGFR 45-59 mL/min/1.73 m² .
In addition to reduced eGFR, albuminuria has also been associated with impaired cognition. In the Australian Diabetes, Obesity, and Lifestyle (AusDiab) cohort study, baseline elevated albuminuria predicted worse memory function at 12-year follow up but baseline eGFR did not predict decline in memory function or processing speed. In the study, elevated albuminuria at baseline was associated with reduced scores for cognitive testing compared to patients without albuminuria, but after attenuating for cardiovascular risk factors only the difference in the California Verbal Learning Test was statistically significant. Decreased eGFR did not predict memory or processing speed at the 12-year follow up. An analysis of The Maastricht Study in the Netherlands demonstrated higher levels of albuminuria were associated with decreased information processing speed whereas eGFR cystatin C-Cr did not. Albuminuria may be associated with these changes as it is a marker of underlying microvascular disease.
Hypertension in CKD has been linked to a higher risk of developing cognitive impairment. Within the CRIC cohort, patients with an eGFR > 45 mL/min/1.73 m² and elevated baseline systolic blood pressure were found to have an increased risk of developing cognitive impairment based on the mini mental status exam (MMSE). Even acute kidney injury (AKI) has been recently linked to increased risk of developing dementia and MCI. As part of a retrospective cohort analysis of the SPRINT trial, AKI was determined to be a risk factor for developing dementia even with the majority of patients recovering from the AKI. Collectively, these findings suggest that vascular injury may represent the common pathway linking kidney dysfunction to cognitive decline.
Cognitive impairment in CKD likely arises from multiple converging mechanisms, with cerebrovascular disease emerging as a key contributor. CKD confers a heightened risk of small vessel pathology, and the shared microvascular architecture and autoregulatory demands of the brain and kidney may explain this parallel vulnerability. In the setting of small vessel disease, impaired microvascular perfusion is thought to reduce neuronal activity and contribute to subcortical cognitive dysfunction. Moreover, even when global cerebral blood flow is preserved, or compensatorily increased, cerebral oxygen delivery may remain inadequate in CKD, particularly in the presence of anemia, further amplifying neuronal vulnerability.
The glymphatic system of the brain has also been hypothesized as playing a role in cognitive impairment in CKD. The glymphatic system is the waste clearance system of the central nervous system (CNS). This system can be negatively affected by hypertension and diabetes and there is evidence of decreased glymphatic system function in CKD that does not improve with kidney replacement therapy. Uremic toxins are also potential contributors to cognitive impairment in CKD. Though overt uremia does not typically manifest until eGFR is less than 10 to 15 mL/min/1.73 m², metabolites accumulate at earlier stages. One possible metabolite that could contribute to cognitive impairment is 4-hydroxyphenylacetate. In a study with both a discovery cohort and a replication cohort, patients underwent cognitive testing and had uremic metabolites measured pre-dialysis. Of the uremic metabolites measured, 4-hydroxyphenylacetate had a statistically significant association with impaired executive function in both the discovery cohort and the replication cohort. Finally, there are likely genetic factors that predispose patients with CKD to cognitive impairment. In pediatric nephrology, there is a stronger connection to genetic causes of CKD whereas only 10% of adult patients with CKD have an identified genetic cause. There are some genetic diseases that cause both cognitive impairment and CKD such as Fabry disease, tuberous sclerosis, Joubert syndrome, and Lowe syndrome. At present, more research is needed to elucidate the connection.
The clinical manifestations of cognitive impairment in CKD are vast. In a systematic review and meta-analysis covering cognition and CKD, the manifestations of cognitive impairment in CKD varied by stage of CKD. Overall, performance on cognitive testing worsened as CKD stages advanced in numerous facets of cognition. Individuals with eGFR < 60 mL/min/1.73 m² performed worse across several cognitive domains: orientation and attention, language, reasoning, executive function, memory, and global cognitionrelative to those with preserved kidney function. This held true when the eGFR cutoff was lowered to 45 mL/min/1.73 m² and 30 mL/min/1.73 m².
Since the manifestations of cognitive impairment in CKD is variable, it is difficult to know which cognitive testing should be performed to screen for MCI in CKD. There is currently no single dedicated screening test for patients with CKD. A recent systematic review focused on this question. In the systematic review, 10 studies were included in the analysis. Of these studies, 3 were focused on patients with CKD, 6 on patients on hemodialysis (HD), and 1 on patients on peritoneal dialysis (PD). Within the studies, the screening instruments used were the MOCA, MMSE, 3MS, M-ACE, and the Kidney Disease Quality of Life-Cognitive Function subscale. A total of 1,801 patients were screened and 995 patients were found to have cognitive impairment. The systematic review found that the MOCA was the best overall screening instrument for patients with CKD for cognitive impairment. The additional benefits of the MOCA are that it takes 10 minutes to administer and can be administered in over 100 languages.
The management of cognitive impairment in patients with CKD focuses on prevention and controlling blood pressure and albuminuria. Within the CRIC cohort, patients were at an increased risk of incident cognitive impairment with elevated systolic blood pressure and eGFR > 45 mL/min/1.73 m². Focusing on controlling blood pressure in patients with early CKD may decrease their future risk of developing MCI.
Controlling albuminuria may also reduce the risk of developing MCI. Evidence specifically addressing albuminuria and cognitive outcomes in CKD is sparse; however, studies have explored whether lowering albuminuria in patients with preserved eGFR affects the development of mild cognitive impairment. In a post hoc analysis of the ONTARGET/TRANSCEND studies, patients with albuminuria during initial screening were more likely to have reduced MMSE scores. Additionally, patients who developed albuminuria at 5 years were at an increased risk of having cognitive decline, and patients with albuminuria on initial screening who were treated with RAAS blockade were at decreased risk of decreased MMSE scores at 5 years compared to patients on placebo. Reduced eGFR was not a requirement of the study and only 24.6% of patients had eGFR < 60 mL/min/1.73 m² in the post hoc analysis.
Team 2:
Cerebronephrology in ESKD
Dialysis fixes uremia but stresses the brain. This group tackles how hemodialysis and peritoneal dialysis affect cerebral blood flow, intracranial pressure, stroke risk, and white matter integrity, shaping cognitive outcomes in patients with ESKD.
Image adapted by chatGPT from: kenonl/ Shutterstock
Now that we have reviewed the impact of CKD on cognitive impairment, we will switch gears to patients with ESKD and the impact of kidney replacement therapy on the brain. Kidney replacement therapy results in reductions in serum osmolality, intravascular volume, and mean arterial pressure. These reductions are a necessary consequence to manage uremia, hypervolemia, and hypertension in patients with ESKD. Unfortunately, these benefits may have negative impacts on the brain by inducing transient ischemia and fluctuations in intracranial pressure.
The first impact we will explore is dialysis disequilibrium syndrome. Dialysis disequilibrium syndrome is defined by the acute development of neurologic symptoms during a hemodialysis treatment. Symptoms can range from mild (headache and nausea) to severe (seizures and coma). The symptoms are caused by the development of cerebral edema, which is attributed to the rapid clearance of urea from the intravascular space and slower diffusion of urea across the blood brain barrier (BBB) with subsequent net movement of water into the brain. Another proposed mechanism is paradoxical brain acidosis, wherein intracerebral pH decreases with hemodialysis despite the blood pH increasing. The rapid correction of pH in the intravascular space results in a rapid rise of PCO2 that forms carbonic acid in the brain and lowers brain pH. The lower pH results in displacement of intracellular cations and a subsequent rise in intracerebral osmolality, ultimately resulting in the net movement of water into the brain. A final hypothesis focuses on idogenic osmoles where there is an increase in intracerebral osmoles as a compensatory mechanism in the setting of increase in intravascular hyperosmolality. The reduction in serum osmolality following hemodialysis results in net water movement into the brain and causes cerebral edema.
Patients with ESKD are also at high risk for developing cerebrovascular disease (CVD), including stroke. In a 2003 study, incident hemodialysis and peritoneal dialysis patients were found to have a higher rate of hospitalization for stroke compared to the general population adjusting for age and gender. The increased risk was seen for both ischemic and hemorrhagic strokes. Additional evidence comes from the CHOICE study, a prospective cohort study designed to quantify the incidence of cerebrovascular disease in incident hemodialysis patients. Patients were enrolled from 1995-1998 and followed until the end of 2004. During this time, the overall incidence of CVD was 4.9 events/100-person-years with CVD defined as nonfatal (hospitalized stroke and carotid endarterectomy) and fatal (stroke leading to death). Ischemic stroke was the most common event within the cohort. Finally, a systematic review and meta-analysis explored the incidence of stroke in patients on HD vs patients on PD. From fifteen studies, the meta-analysis demonstrated a statistically significant decreased risk of hemorrhagic stroke for patients on PD compared to HD but overall no difference in ischemic stroke or all types of stroke.
Beyond the increased risk for CVD, patients on dialysis are at increased risk for structural brain changes. In one study, 30 patients on HD underwent MRI to assess for cerebral atrophy and were screened for cognitive impairment. They were compared to a group of healthy controls with similar age, sex, and cerebrovascular risk factors. Nine of the 30 patients on HD had a prior stroke, and 80% of the patients on HD were found to meet the criteria for dementia based on their scores on MMSE and Mattis Dementia Rating Scale along with the DSM-IIIR definition of dementia. The MRI showed that patients on HD were found to have a greater burden of cortical atrophy, ventricular atrophy, and white matter hyperintensities. Additionally, in a cross-sectional cohort study assessing brain abnormalities on MRI of patients with ESKD on HD and controls, patients on HD were found to have more white matter disease and cerebral atrophy along with a higher prevalence of small-vessel and large-vessel infarcts compared to the control group without kidney disease.
Another study utilized diffusion tensor imaging (DTI) to obtain detailed imaging of white matter tracts in the brain and compared the images between patients receiving hemodialysis and healthy controls of similar age. DTI provides measured water displacement within the brain and typically reports a measure called fractional anisotropy. Fractional anisotropy (FA) quantifies the directional diffusion of water within white matter tracts and serves as a marker of microstructural integrity. Higher FA values reflect preserved white matter architecture, while lower values indicate structural disruption. In hemodialysis patients, FA was reduced in frontotemporal pathways, the genu of the corpus callosum, and the fornix, consistent with more advanced white matter injury.
One final study focused on patients with ESKD on peritoneal dialysis compared to patients with CKD and not on dialysis. In this study, 62 patients on PD and 69 patients with CKD underwent brain MRI then underwent a second brain MRI two years later. In the initial MRI, patients on PD were found to have lower gray matter volume but there was no difference in white matter volume. For the second MRI, 34 of the original patients in the PD group and 61 patients in the CKD group participated. The patients on PD had a greater decrease in the measured gray matter volume compared to the patients with CKD.
Another impact of dialysis on the brain is cerebral blood flow and cerebral oxygen delivery. In patients with ESKD, cerebral blood flow at baseline is increased due to underlying anemia. Despite the increased cerebral blood flow, cerebral oxygen delivery is ~15% lower as the increased cerebral blood flow is not able to compensate for the underlying anemia in ESKD. At present, the only way to measure systemic pressure, including cerebral pressure, is blood pressure monitoring during dialysis. Some studies have used advanced techniques to monitor cerebral blood flow and cerebral oxygenation during dialysis. In one pilot study, the relationship between dialysis-associated hypotension and cerebral oxygenation was assessed. For the study, cerebral oxygenation was monitored with near-infrared spectroscopy (NIRS) during hemodialysis sessions while blood pressure was monitored with volume-clamp blood pressures on a finger cuff. NIRS measures the differential absorption of near-infrared light by oxygenated and deoxygenated hemoglobin to provide continuous, noninvasive estimates of regional hemoglobin oxygen saturation within tissues approximately 2–3 cm beneath the skin. Because near-infrared light can penetrate the skull, NIRS enables monitoring of cerebral oxygenation, particularly within the frontal cortex. Blood pressure was measured continuously via a finger BP cuff or oscillometry every 15 minutes on the arm. During a median of 12 consecutive hemodialysis sessions of 54 patients (total of 583 sessions), 23.5% of sessions featured a drop in cerebral oxygen by at least 15% while only 31.9% of these episodes were symptomatic.
In another prospective cohort study, cerebral blood flow (assessed as mean flow velocity) during hemodialysis was measured by transcranial doppler ultrasound of the cerebral artery. During the study, mean flow velocity decreased on average of 10% and correlated with ultrafiltration volume and rate of change of weight during hemodialysis. It did not correlate with absolute blood pressure values. As part of the study, cognitive function testing was performed during dialysis, off of dialysis, and 12 months after the initial testing. During dialysis, patients performed worse in executive function and time/tasking switching. Finally, mean flow velocity change also correlated with global cognitive decline based upon MOCA scores over the course of the 12 month study along with white matter hyperintensities on brain MRI.
One final study utilized [15O]H2O PET-CT to measure cerebral blood flow during hemodialysis. During the study, cerebral blood flow was measured three times during a session of hemodialysis for 12 patients: before, during, and at the end of the session. The second measurement was on average obtained 30 minutes into the dialysis session. On average, the cerebral blood flow globally was reduced. Additionally, cerebral blood flow decreased in all 6 regions of the brain studied (frontal, temporal, occipital, parietal, thalamus, cerebellum). Different variables were analyzed to see if they correlated with the decrease in cerebral blood flow, and higher tympanic temperature, greater ultrafiltration, and lower pCO2 all correlated with the decrease in cerebral blood flow.
Hemodialysis also raises intracranial cerebral pressure (ICP), which can theoretically cause recurrent brain injury. During hemodialysis, ICP rises due to mechanisms described earlier in the dialysis disequilibrium section. Notably, there are some strategies to potentially mitigate these changes. Increasing the dialysate sodium can limit the rate of change of the osmotic gradient across the BBB. Decreasing the dialysate bicarbonate can limit the increase in pH of the blood. Finally, lowering the dialysate temperature may help limit the decrease in mean arterial pressure (MAP) during hemodialysis. In a retrospective cohort study, patients admitted for an acute brain injury requiring ICP monitoring who underwent renal replacement therapy were analyzed. A total of 13 patients were included in the analysis and underwent either intermittent hemodialysis (N=4) or continuous renal replacement therapy (N=9). The mean baseline ICP was 11.9 mm Hg to a mean peak ICP 21 mm Hg during dialysis. The average ICP also increased to 15.2 mm Hg during dialysis. There was no difference in modality for the peak or average ICP but the time to peak ICP was faster in HD compared to CRRT. In a recent secondary analysis of the HD-REMODEL trial, patients underwent a 3T MRI during hemodialysis to measure cerebral water content and were matched to controls who underwent MRI not on dialysis. The participants who underwent dialysis had an increase in cerebral water compared to the controls which persisted even after dialysis was finished.
Finally, patients with ESKD on renal replacement therapy are at increased risk for developing cognitive impairment compared to patients without ESKD. In a cross sectional study of the COGNITIVE-HD cohort, the cognitive function of patients on hemodialysis was assessed across multiple domains. A total of 676 adult patients were assessed and 474 completed assessments across all 5 domains studied: perceptual-motor function, language, executive function, learning and memory, and complex attention. Each domain was assessed using two neuropsychological tests. The mean age of participants was 70.9 years. Of the 474 patients who completed all testing, 71% of patients demonstrated cognitive impairment in at least one domain and 4.9% demonstrated cognitive impairment in all 5 domains. Of the 5 domains tested, the most common impairment was complex attention with 48.8% of patients with an impairment. In another cross-sectional study, 314 patients on HD underwent cognitive testing. Patients on HD were found to have worse executive function but no difference in memory performance compared to population norms. In another longitudinal cohort study, hemodialysis patients studied over a median 2.1 years demonstrated a decline in executive function on neurocognitive testing.
In summary, the Cerebronephrology region demonstrates the complex relationship between the brain and kidneys and the brain-kidney axis. Cognitive impairment is an underrecognized complication of chronic kidney disease, and kidney replacement therapy results in transient ischemia and fluctuations in ICP in order to manage the complications of ESKD. Greater understanding of these interactions and further exploration is imperative to improve care for patients.
– Executive Team Members for this region: Dia Waguespack @d_r_waguespack and Jeffrey Kott @jrkott27 – @jrkott27.bsky.social | Meet the Gamemakers
How to Claim CME and MOC
US-based physicians can earn 1.0 CME credit and 1.0 MOC per region through NKF PERC (detailed instructions here). The CME and MOC activity will expire on June 1, 2026.





Great work writing this region.
I sense that the game is afoot though. The DDS visual abstract has been switched with the VALIANT trial. A sneaky move by TeamC3G!