#NephMadness 2022: Parasites Region
Submit your picks! | NephMadness 2022 | #NephMadness | #Parasites
Selection Committee Member: Arvind Conjeevaram @arvindcanchi
Arvind Conjeevaram is a Consultant Nephrologist and Kidney Transplant Physician in Bangalore, India. Consequent to his interest in nephrology education, he set up #ECNeph, a nephrology case discussion forum on Twitter, and won the Nathan Hellman Social Media Award in 2017, an award given for the dissemination of nephrology worldwide through social media. He has been the Past Chair of the @ISNeducation team and Lead on the Social Media Committees of various conferences.
Writer: Mythri Shankar @nephromythri
Dr Mythri Shankar is an Assistant Professor at the Institute of Nephro-urology, Bengaluru, India. She is a National Gold Medalist in DNB Nephrology for the year 2017. She is a graduate of Glomcon Fellowship (2020), NSMC Internship (2021), and winner of NephJC CJASN Best Visual Abstract of 2021.
Competitors for the Parasites Region
Malaria vs Leishmaniasis
Schistosomiasis vs Filariasis

Copyright: Kateryna Kon / Shutterstock
Parasites have plagued mankind for ages. While over 300 parasites are linked to human disease, only about 20 cause kidney dysfunction. Out of those, four tropical parasites strike a particular fear in our hearts (and kidneys). Malaria, our top seed, is going against the trio of Neglected Tropical Diseases: schistosomiasis, leishmaniasis, and filariasis. These diseases receive little attention from the developed world. Endemic areas are characterized by extreme poverty and immense human suffering, while countries successful in eliminating these infections undergo substantial social and economic boost. Let’s stop the neglect and plunge into the fascinating world of nephro-parasitology!

Visual Abstract by @nephromythri on Barsoum
Malaria vs Leishmaniasis
Malaria
“Where malaria prospers most, human societies have prospered least.” – Elsheikha and Sheashaa in Epidemiology, Pathophysiology, Management and Outcome of Renal Dysfunction Associated With Plasmodia Infection (Parasitology Research)

Copyright: Kateryna Kon / Shutterstock
History
Malaria is a prehistoric zoonosis which greeted humans when we evolved. Paleo-parasitologists found malarial antigen in some of the oldest Egyptian mummified remains from 3200 BCE. Compatible symptoms were described in the oldest Chinese medical script from 2700 BCE, Nei Ching. The Indian Vedic literature in 1500s BCE proclaimed malaria the “king of diseases”. In the 400s BCE, the Epidemics by Hippocrates supplied a detailed account of periodic fever. Classical antiquity brought us the expression “dog days of summer” (when Sirius, the Dog star, is high in the sky), associated with ill health because of periodic fevers of malaria. Historians suspect that the falciparum malaria epidemic of 79 CE could have contributed to the fall of Rome. The name itself, mal’aria, means bad air in Latin, and the first intervention for disease control was drainage of swamps across the entire city of ancient Rome by emperor Nero. Alphonse Laveran deduced that malaria was not caused by miasma but a parasite, and made his Nobel prize-winning discovery in 1880. Tu Youyou, a Chinese chemist, received the Nobel Prize in 2015 for discovering the most effective treatment for malaria, artemisinin compounds, inspired by the traditional 4th century Chinese “Handbook of Prescriptions for Emergencies.”
Epidemiology

Infographic by @krishnadoctor1 based on information from the CDC
Malaria is endemic to the tropics. It is caused by protozoa of the genus Plasmodium. The major species to torment humans are P. falciparum (the most common and dangerous), P. vivax, P. ovale and P. malariae. Kidney involvement is most likely in P. falciparum and P. malariae infections. Up to 95% of malaria cases occur in Africa, the rest in South and South-East Asia and Latin America. With an incidence of 200-300 million cases, malaria results in a quarter million deaths every year. As testament to public health efforts, endemic areas are receding and disappearing. Sri Lanka was declared free of malaria in 2016, Paraguay in 2018, and Algeria and Argentina in 2019. In total, 39 countries have become malaria-free in recent years.
Anopheles mosquitoes spread the parasite as they bite, usually between dusk and dawn. Other rare modes of transmission include contaminated needles, organ transplantation, blood transfusion, nosocomial transmission, and congenitally acquired malaria.
Life Cycle

Infographic by @nephromythri based on information from the CDC
Common Clinical Manifestations
Severe malaria is caused by P. falciparum or, in Southeast Asia, by zoonotic P. knowlesi. It is a medical emergency. Severe manifestations include encephalopathy, seizures, respiratory failure, shock, hemolytic anemia, coagulopathy, liver failure and, of special interest to us, acute kidney injury (AKI).
Uncomplicated malaria can be caused by any of the plasmodium spp. Patients have fevers, anemia and splenomegaly. The classic malarial periodic fevers are now rare.
Chronic malaria is the specialty of P. malariae. Additionally, P. vivax and P. ovale relapse via hypnozoites which can snooze in the liver for months to years. Even P. falciparum can recrudesce due to treatment resistance. Manifestations can be fevers, anemia, hepato-splenomegaly, and, important for us, immune complex nephritis due to chronic antigenic stimulation.
Kidney Involvement in Severe Malaria
Severe malaria is likely to cause AKI in adults (up to 30-40%) and children (50%). Malaria-induced AKI is the third most common cause of AKI in children from Nigeria, with a mortality rate of 5% to 25%. The typical manifestation is oligo-anuric AKI with metabolic (lactic) acidosis, hyponatremia and hyperkalemia.
Pathogenesis of kidney injury in severe malaria is complex and includes mechanical factors, immune mediation, cytokine release, acute phase reactants, and hemolysis-induced acute tubular injury (“blackwater fever”). A unique mechanism is impairment of kidney microcirculation by sequestration of parasitized RBC, leading to tissue hypoxia.

Infographic by @nephromythri adapted from da Silva, Junior, et al
Histopathology of AKI in severe malaria is acute tubular necrosis, often with deposition of hemosiderin, urinary casts, mononuclear interstitial infiltrate and interstitial edema.
Some reports show acute interstitial nephritis, or even glomerulonephritis with mesangial hypercellularity and IgG/IgM/C3 deposits in mesangial and subendothelial locations.
Collapsing FSGS with Hemolytic Uremic Syndrome were also described.
Kidney Involvement in Chronic Malaria
Chronic P. malariae infection, or “quartan fever” (classically recurs every 4th day), is notorious for causing quartan nephropathy. Manifestations vary based on immuno-genetic factors of the host from mild and transient proteinuria to nephrotic syndrome and progression to kidney failure even after elimination of parasites.
Histopathology of quartan nephropathy is MPGN, occasionally with crescents. Immunofluorescence may show various degrees of IgG, IgM, C3, and P. malariae antigen presence along the capillary loops. Electron microscopy shows subendothelial deposits and the characteristic thickening and splitting of the basement membrane. The lesion may progress to glomerular sclerosis.

Typical glomerular lesion attributed to quartan malaria. (Left) Mesangiocapillary: membranoproliferative lesion with focal basement membrane thickening (H&E stain). (Right) Silver stain shows basement membrane thickening with intramembranous “lacunae” of immune deposits. Figure 4 from Barsoum, AJKD, © National Kidney Foundation.
Diagnosis
Malaria should be high on the differential in any febrile resident of or visitor to an endemic area. Children from these areas may have anemia without fever. The gold standard for diagnosis are Giemsa-stained blood smears. Thick smears are sensitive for the detection of parasites, whereas thin smears are used to identify parasite species and measure parasite density. Rapid diagnostic tests are essential in low resource settings. Currently available tests detect plasmodium antigens, like plasmodium LDH or aldolase, and more are being developed.
Treatment
Severe malaria can be fatal within a few hours of presentation, hence early initiation of treatment is essential. First-line treatment of severe malaria is artemisinin-based combination therapy. Artemisinins do not require dose adjustment for dialysis. Where artemisinins are not available, intravenous quinine remains an option. One “proof of principle” study from South Asia showed that the use of acetaminophen (1 g every 6 hrs x 72 hrs) in febrile patients treated with artesunate was renoprotective.
Prevention and Control
WHO designated April 25th as World Malaria Day to promote political support and investment in malaria eradication. According to WHO, malaria can be controlled by a 4-prong strategy of personal protection, mosquito control, chemoprophylaxis, and vaccination.
In the News: A New Vaccine
A recent trial evaluated the new R21-Matrix M malaria vaccine in children, and found an efficacy of 74-77% after three doses.
Leishmaniasis
“That is where the sandfly lives, a tiny creature, even smaller than a mosquito, which feeds at dusk and makes no sound in flight.” – Irene Nunez in Who Cares About Kala Azar? (Médecins Sans Frontières Blog)

Image from CDC Public Health Image Library / Frank Collins, James Gathany (Public Domain)
History
Leishmania was very much a presence in the ancient world. A molecular paleopathology study of Egyptian and Nubian mummified remains dating back 4000 years identified mitochondrial DNA of leishmania kinetoplast. Interestingly, ancient people in Asia and Middle East knew that cutaneous ulcers from the sandfly bites could only happen once, and inoculated ulcer exudate into the buttocks of small children to protect them from disfiguring facial scars. This rudimental vaccination practice was used for millennia and, under the name of leishmanization, was popular through the 20th century. Now, with CRISPR gene editing technologies, a safer second-generation leishmanization is making its way to clinical trials.
In the 19th century, an epidemic of kala-azar, or visceral leishmaniasis, ravaged the Indian states of Bengal and Assam, wiping out a third of the population. Kala-azar (black disease) is named for the hyperpigmentation of the skin, historically a feature of this infection. In the early 20th century, a Scottish pathologist, William Boog Leishman, and an Irish physician, Charles Donovan, both working in India, discovered the parasite in autopsies of affected soldiers within a few weeks of each other, giving Leishmania donovani its name.
Epidemiology

Infographic by @brian_rifkin based on information from the CDC
Leishmaniasis is a Neglected Tropical Disease; it is a zoonosis caused by the intracellular protozoa from the genus Leishmania. It is transmitted by the bite of a female sandfly. There are several clinical manifestations depending on the location of the infected macrophages: cutaneous, muco-cutaneous, and visceral. We are primarily concerned with visceral leishmaniasis (VL), which affects liver, spleen, bone marrow and, of course, the kidneys. VL is caused by L.donovani and L.infantum.
There are between 1 and 2 million cases of leishmaniasis yearly. It is widespread in tropical and subtropical regions, however, more than 90% of cases come from just 13 countries. Most leishmaniasis cases are asymptomatic, but VL is fatal without treatment. Risk factors for symptomatic and severe illness include poor nutritional conditions, host immunogenetic factors, co-infection with HIV, and age <5 years (especially L. infantum).
Life Cycle

Infographic by @nephromythri based on information from the CDC
Common Clinical Manifestations
Visceral leishmaniasis starts with early symptoms (malaise, fatigue, fever, weight loss), progresses to hematological manifestations (pancytopenia, hypergammaglobulinemia), along with mononuclear phagocyte system activation (hepato-splenomegaly). Late symptoms are liver dysfunction and cachexia.
Kidney Involvement in VL
Renal manifestations of VL are common and consist of tubular or glomerular effects as well as drug nephrotoxicity. A cohort study examined 50 patients with VL and found that roughly half had abnormal urinary findings. Low grade proteinuria, less than 1 g per day, was common (57%). Of the 7 patients biopsied, tubulo-interstitial involvement was found in all, and proliferative glomerulonephritis in 5. The kidney disease subsided within weeks of treatment.
A cross-sectional study of 55 patients with chronic VL found proximal tubular dysfunction with tubular proteinuria and glucosuria, as well as nearly universal hyponatremia and frequent hypokalemia, hypocalcemia and hypomagnesemia. Two-thirds of the patients had complete or partial proximal renal tubular acidosis. One-third of the patients had decreased GFR.
Glomerular disease in VL is possible, in forms of nephrotic syndrome, acute glomerulonephritis and rapidly progressive glomerulonephritis.
Most of the drugs used for anti-leishmanial therapy are nephrotoxic. Amphotericin B is notorious for causing multiple electrolyte abnormalities as well as acute kidney injury; antimonial compounds can cause ATN.
Histopathology: the most common reported lesions are interstitial nephritis and proliferative glomerulonephritis. Rarely, glomerular lesions can represent segmental necrotising glomerulonephritis or secondary AA amyloidosis.
Diagnosis
Diagnosis of VL can be made by a number of means: visualization of amastigotes on a blood smear or histologic specimen; detection of parasitic DNA in blood or tissues; in vitro culture (microtiter plates are used successfully in India with sensitivity of 60%-85%). Serological tests, ELISA and IFA, are useful in endemic areas with high clinical suspicion of VL where specialized laboratories are not available.

Light microscopy of bone marrow specimen – Macrophage containing amastigote with a nucleus (red arrow) and a kinetoplast (black arrow). Kinetoplast is characteristic for the diagnosis of Leishmaniasis. Image from CDC/DPDx (Public domain)
Treatment of Visceral Leishmaniasis
Untreated, severe VL has a mortality rate of 95%. The most effective agent is liposomal amphotericin B. Combination therapies are preferred, and armamentarium includes amphotericin B, antimonial compounds,paromomycin and oral miltefosine, depending on geographical resistance patterns.
Prevention and Control
There is always a wildlife reservoir, as Leishmaniasis is a zoonotic infection. This complicates disease control measures. Vector control to reduce transmission by sandflies is the way to go, as well as vaccine development.
In the News: APOL1 Correlations
Trypanosome Lytic Factor (TLF) is a high-density lipoprotein (HDL) complex that provides innate immunity against certain kinetoplastid parasites such as trypanosomes through the cation channel-forming protein APOL1. Recent animal models have demonstrated that it can also kill the intracellular leishmania promastigotes inside the acidic vacuole of macrophages. TLF seems to have evolved since ancient times and may provide innate immunity against Leishmaniasis in humans with APOL1 gene polymorphisms. This finding adds leishmaniasis to the sleeping sickness as another tropical disease which could have exerted selection pressure for the rise of effective but nephrotoxic APOL1 gene variants.
Schistosomiasis vs Filariasis
Schistosomiasis
“Water poison attacking man…
Like a poisonous insect, but invisible…”
Ge Hong in Zhouhou Beiji-fang (China, 400 CE)

Copyright: Oleksandr Yakoniuk / Shutterstock
Schistosomiasis is an age-old disease caused by the trematode flukes. How old is age-old? An intact schistosome egg was found in a 6000-year-old human pelvic skeletal sediment from a settlement in the Euphrates river valley, topping a 5000-year-old Egyptian adolescent as the earliest known case of human schistosomiasis. Hematuria, the predominant symptom, was so common in ancient Egyptian boys that it was viewed as “male menstruation”, a sign of puberty. One paleopathology study detected a schistosome antigen in 65% of the mummies tested. High prevalence of this disease in Northern Africa since pharaonic times is attributed to irrigation projects in the Nile delta providing ideal conditions for “the schistosome parasite, the aquatic snail intermediate host, and the human definitive host converge in space and time in surface waters”. Schistosomes have a reputation for infecting soldiers, famously, in Napoleon’s campaign to Egypt, the China -Taiwan conflict (literally settled by the fluke), and the Vietnam War.
Alternate names are “bilharziasis” (pays homage to Theodor Bilharz who identified the parasite in 1852), and “snail fever.”
Epidemiology

Fighting Schistosomiasis in Uganda. Scenes from the banks of Lake Victoria in Mukuno District, central Uganda. Image from RTI International/Katie G. Nelson (CC BY-NC-ND 2.0)
Worldwide, more than 200 million people have schistosomiasis, with the highest prevalence found in Sub-Saharan Africa. Approximately 120 million have symptoms, 20 million have severe disease, and 100,000 die each year. It is one of the Neglected Tropical Diseases.
Endemic areas are predominantly rural, with bodies of freshwater where snails thrive. Children can be infected while bathing or playing in lakes and streams contaminated with cercarial larvae. In children, the parasitic load gradually increases until adolescence.
Travelers can be infected following a single exposure. For instance, an outbreak of genito-urinary schistosomiasis was observed near the Cavu River in France. Investigation showed that French tourists were infected in Senegal and brought the parasite back home, where it then spread to non-travelers.
Each human schistosome species requires a specific species of snail to complete its life cycle.

Infographic by @CTeodosiu based on information from the CDC
Morphology
Adult male and female flukes are 10-20 mm tubes with rudimentary oral and ventral suckers. Males are short and stout while females are long and slender. The name schistosome (split body) refers to male’s gynecophoral canal, in which the female lies folded.

Adult male and female flukes. Adapted from Servier Medical Art (CC BY 2.0)
Life Cycle

Infographic by @nephromythri based on information from the CDC
Clinical Manifestations
Clinical syndrome of schistosomiasis is consequent to granulomatous immune reaction to eggs and schistosomal antigens. The disease can range in severity from asymptomatic to severe, depending on the intensity of the parasitism, immune response, and location of the eggs.
The infection can be divided into an initial and late phase:
- Initial phase corresponds to the entry of cercariae into the skin causing an allergic reaction or dermatitis (swimmers itch). The cercariae penetrate the skin, enter the veins and finally, having developed into schistosomula, the lungs. Their passage through the lungs results in cough, fever, and eosinophilia. Acute schistosomiasis syndrome, or Katayama fever, develops several weeks following the infection. It is characterized by malaise, gastrointestinal symptoms, fever, urticaria or angioedema, and eosinophilia.
- Late phase: The chronic form (>6 months), is characterized by fibrosis of the affected organs manifesting as portal hypertension, pulmonary hypertension, ascites, esophageal variceal rupture. It is characterized by granulomatous inflammation, high levels of proinflammatory cytokines, and eosinophilia.
Genitourinary Schistosomiasis
Genitourinary Schistosomiasis is caused by S.haematobium.
- Initial phase: During early stages of infection, eggs are excreted in the urine, resulting in hematuria. Blood is usually seen at the end of voiding, which is called “terminal hematuria.” In severe cases, blood may be seen throughout the void. Men may also present with hemospermia.
- Late phase: Urinary bladder inflammation occurs 3 to 4 months following primary infection and can present with hematuria and dysuria. On cystoscopy, hypertrophic nodules, polyps, and deposited eggs are visible. The lesions are reversible if treated prior to fibrosis and calcification.
- Long standing infection can cause fibrosis or calcification in the bladder wall, squamous cell bladder carcinoma, urinary obstruction leading to acute and chronic kidney disease; infertility and increased risk of HIV transmission. Ultrasound can demonstrate irregularities in the bladder wall, hydronephrosis, polyps and tumors. CT urogram or IV pyelogram can demonstrate ureteral strictures.

Image from CDC Public Health Image Library / CDC/ Dr. Edwin P. Ewing, Jr. (Public Domain)

Bladder calcifications in schistosomiasis. Image from Wikimedia / Jmarchn (CC BY-SA 3.0)
Schistosomal Nephropathy
Schistosomal glomerulopathy is a feature of intestinal and hepato-splenic schistosomiasis (chronic infection by S. mansoni, S. japonicum or S. mekongi). Only rarely is it caused by S. haematobium. Schistosomiasis can also cause granulomatous tubulointerstitial nephritis. The incidence of glomerulopathy in S. mansoni is 5% in hepato-intestinal form, but increases to 15% in hepato-splenic form due to impaired antigenic clearance. Proteinuria is reported in 20% of patients.
Pathogenesis of glomerular injury is initiated by antigens released by the parasites into circulation. Progression to advanced stages and glomerulosclerosis is due to the following factors:
- Hepatic fibrosis: This impairs the clearance of the antigens from portal circulation and it reaches the systemic circulation via intra- and extrahepatic porto-systemic shunts.
- Increased circulating IgA: Patients with schistosomiasis have increased mucosal production of IgA. Due to hepatic fibrosis there is impaired clearance of these IgA, which eventually reaches systemic circulation and gets deposited in the kidney.
- Autoimmunity: Experimental animals and infected patients can develop anti-DNA or anti-phospholipid antibodies, rheumatoid factor.
- Coinfection: Coinfection with Salmonella, Hepatitis B, Hepatitis C, HIV modifies glomerular structural functions
- Genetic factors: may influence susceptibility to infection, progression to liver fibrosis, type and progression of glomerulonephritis.

Infographic by @nephromythri based on information from Barsoum
Classification of Schistosomal glomerulopathy (SGN) endorsed by the African Association of Nephrology (AFRAN)
- Class I: Mesangioproliferative glomerulonephritis as a result of mesangial deposition of parasitic antigens and antibodies. Usually asymptomatic with mild proteinuria and/or microscopic hematuria, normal GFR. Benign course and complete recovery is typical with treatment.
- Class II: Proliferative exudative glomerulonephritis, which occurs in cases of co-infection with Salmonella. Nephrotic/nephritic syndrome. Reversible with simultaneous treatment of Schistosomiasis and Salmonella.
- Class III: Membranoproliferative glomerulonephritis, predominant SGN lesion in the 1970s.
- Class IV: Focal Segmental Glomerulosclerosis, sometimes associated with high risk APOL1 genotype. Predominant lesion since successful treatments of chronic schistosomiasis. Classes III and IV are associated with liver cirrhosis and reflect high levels of antigenemia shunted to the kidneys due to portal hypertension. Clinically patients present with nephrotic range proteinuria, hypertension, and progresive decline in eGFR despite antiparasitic treatment.
- Class V: Rarely, AA kidney amyloidosis due to persistent infection. Presentation is with nephrotic syndrome +/- reduced GFR. The course and prognosis are similar to other causes of secondary amyloidosis.
- Class VI: Cryoglobulinemic Glomerulonephritis due to HCV co-infection. Many glomerular lesions are simultaneously present such as mesangial proliferation, fibrinoid necrosis, capillary thrombosis. Immunosuppression may be tried in these cases, along with HCV treatment and anti-parasitic therapy.
Rarely, membranous nephropathy has been seen in patients with schistosomiasis and nephrotic range proteinuria. There is scant evidence for a pathogenic link, hence not included in AFRAN classification.
Diagnosis
We should clinically suspect schistosomal glomerulopathy in patients with a history of schistosomal infection or those coming from an endemic area, especially with evidence of chronic liver disease. Definitive diagnosis requires identification of schistosomal antigens on kidney biopsy, which is currently possible only in specialized laboratories. Urine and/or stool microscopy is rarely useful since schistosomal glomerulopathy is a late complication occurring after the worms have ceased laying eggs. Submucosal rectal or bladder biopsy can demonstrate old calcified eggs. Serological tests target circulating antibodies or parasitic antigens. The antigens are species specific. The most reliable serological test is FAST-ELISA.
In confirmed disease, mild cases with proteinuria < 1g/day can be monitored for resolution of symptoms following antiparasitic therapy without kidney biopsy. Severe cases with suspicion of schistosomal glomerulopathy should undergo kidney biopsy to rule out other glomerular diseases.
Patients with chronic hepatosplenic schistosomiasis and liver cirrhosis with normal kidney function should be screened annually as they are at high risk for developing glomerulopathy.
Treatment
Treatment of underlying parasitic infection with praziquantel or oxamniquine if resistant. Praziquantel is safe and side effects are mild. Oxamniquine may cause nausea, dizziness and urticarial reactions.Immunosuppression is not required except for life threatening Class VI glomerulopathy.
Control and Prevention
Some of the preventive measures are to minimize contact with contaminated freshwater, mass treatment with praziquantel in endemic areas, water sanitation programs and vaccine development.
In the News: TRP Channel in Schistosomes
The mechanism of action of the first line drug, praziquantel, was recently elucidated to be via a schistosome TRPM channel, and resistance to this drug – to genetic variants of this channel, opening the door to developing more effective treatments.
Filariasis
“I will examine some mosquitoes that have bitten a patient to see if they suck up the filariae”. Charles Bancroft, in a letter to T. Spencer Cobbold, 1877.

Copyright: CoolKengzz / Shutterstock
History:
A 4000-year-old Egyptian statue of Pharaoh Mentuhotep II with leg edema is considered the earliest evidence of filariasis. Our old favorite Ebers Papyrus provides the earliest written records of this malady. Around 600 BCE, an Ayurvedic manuscript, Susruta Samhita, described a swollen limb as a symptom of filariasis. The term “elephantiasis”, coined by Celsus in his review of Greek and Roman medical knowledge 2000 years ago, has now been used by Chinese, Asian, Indian and Arabic physicians for millenia. A 16th century Dutch merchant and historian wrote about people from the Indian state of Goa with a leg “as thick as elephant’s” from the knee down. The causative filaria, Wuchereria bancrofti, is named after physicians who identified this worm and its life cycle in the 1800s.
Epidemiology:

Infographic by @sophia_kidney based on information from the CDC
Filariasis is caused by nematodes which inhabit the lymphatics and subcutaneous tissue of the definitive host (human). Mosquito vectors transmit the infection.
Two thirds of patients affected with lymphatic filariasis are in Asia. The epidemiological pattern is shifting due to the implementation of mass drug administration. Many countries have been declared non-endemic, such as Trinidad and Tobago, Costa Rica, and Suriname. From 2000 to 2018, the total number of cases has decreased from 200 million to 50 million. However, in areas where MDA has not been implemented, the infection rates are still high. The COVID pandemic has delayed the elimination program.
According to the WHO, 36 million people are currently disfigured and seriously incapacitated by chronic lymphatic filariasis.The burden of infection increases proportionately with the time spent in the endemic area. A third of children under 5 from endemic areas are infected. By the third or fourth decade most adults are infected. The mosquito vectors for Wuchereria are Culex, Aedes, Mansonia, Anopheles and Coquillettidia, and the vectors for Brugia are Aedes and Mansonia.
Life Cycle

Infographic by @nephromythri based on information from the CDC
Common Clinical Manifestations
Two-thirds of the infected are asymptomatic, some will have subclinical features such as abnormal lymphatic drainage and lymphatic dilation, scrotal lymphangiectasia, and microscopic hematuria/proteinuria. Acute manifestations vary from acute adenolymphangitis to filarial fever to tropical pulmonary eosinophilia (hyperresponsiveness to the filarial antigens). Chronic manifestations include lymphedema, hydrocele, and kidney disease.
Pathogenesis is influenced by the extent of exposure to mosquito bites, quantity of filarial worm antigens in the lymphatics, host immune response, and secondary infections. Early in the infection, adult worms cause subclinical lymphatic obstruction without inflammatory response.
A fascinating story is that filarial worms require an endosymbiotic rickettsia-like organism, Wolbachia, in order to survive. Wolbachia are the most widespread intracellular bacteria in the animal kingdom, and most of the mosquitoes are infected. Being indispensable to the filarial hosts, Wolbachia are an attractive target for filariasis control. Severe inflammatory reactions in treatment of filariasis are attributed to release of Wolbachia into the circulation.
Kidney Manifestations
Chyluria, a rare but dramatic complication, is caused by lymphatic obstruction, formation of lymphatic varices and lymphatic-renal shunting, when intestinal lymph leaks into the renal collecting system. Renal fornices are the most common sites of rupture, but fistulas can be found anywhere along the urinary tract. Due to extensive loss of lipids and proteins in chyluria, these patients are at risk of malnutrition, immunodeficiency, and anemia. Chylous clots can cause obstruction and renal colic. There is usually bilateral lower limb swelling, as well as scrotal swelling and pain, which can be positional or exertional. Chylous urine has a milky appearance, which disappears on a fat free diet. Upon standing, it separates into layers of clot, fat (90% triglycerides), and debris (wbc, rbc). Since chylous urine contains lymphatic proteins, “nephrotic range proteinuria” will be found. Chyluria has to be confirmed by the presence of urinary chylomicrons.
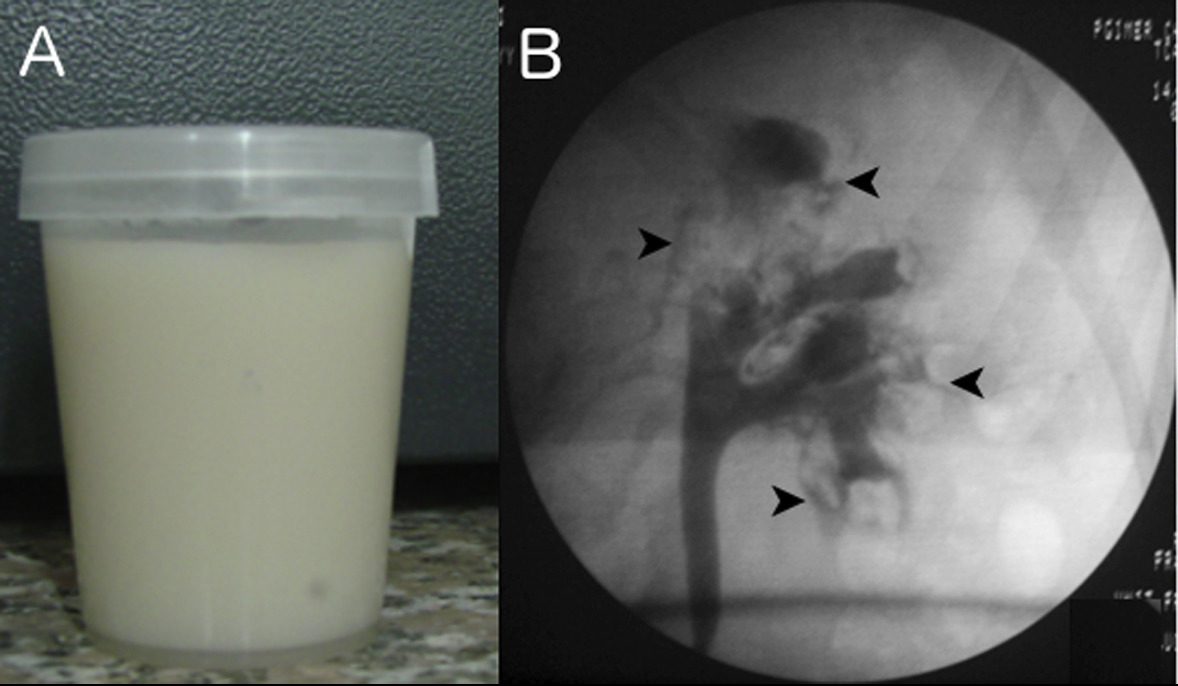
Kidney disease in filariasis. (A) Milky white urine in a patient with chyluria due to filariasis. (B) Retrograde pyelogram in the same patient shows retrograde filling of lacteals (arrowheads). Figure 4 from Jha and Prasad, AJKD, © National Kidney Foundation.
Proteinuria and hematuria can also be seen in Bancroftian filariasis with microfilaremia. In one prospective study of hospitalized microfilaremic patients, these manifestations were present in almost half, transiently increased following initiation of anti-filarial therapy, and resolved by 3 to 4 weeks. A noninvasive technique (SDS-PAGE) was studied in an endemic area in Kerala, India, to differentiate between tubular and glomerular proteinuria in kidney involvement in filariasis. Tubular type of proteinuria was predominant in asymptomatic microfilaria carriers, and glomerular type of proteinuria was seen in symptomatic, chronically ill patients, and carried worse prognosis. In corroboration of tubular to glomerular disease spectrum, experimentally infected rodents had renal interstitial inflammation and tubular degeneration, while dogs with chronic filarial infection had glomerulopathy due to in situ immune complex formation.
In humans, the most dramatic biopsy finding is microfilaria in kidney tissue.

Microfilaria in a peritubular capillary – can you spot it? Image courtesy of Dr. Alok Sharma, Director of Dept. of Renal Pathology & Electron Microscopy, Dr. Lal PathLabs Ltd, New Delhi, India

Microfilaria in a glomerular capillary – can you spot it? Image courtesy of Dr. Alok Sharma, Director of Dept. of Renal Pathology & Electron Microscopy, Dr. Lal PathLabs Ltd, New Delhi, India
The most complete information on clinical-histopathologic correlation comes from a case series of 94 patients at a lymphatic filariasis clinic. Fourteen were found to have proteinuria and hematuria unexplained by other comorbidities, some had slightly decreased GFR, and 2 also had chyluria. Kidney biopsies were done on 9 patients, 6 showed mesangioproliferative changes and mesangial immune deposits with IgG +/- C3, 2 had endocapillary proliferation, and 1 showed immune deposits in capillary loops. This suggested immune-mediated glomerulopathy secondary to either circulating or in-situ formed immune complexes with filarial antigens. Follow up information on these patients was not provided.
Diagnosis
We should suspect filariasis in individuals from endemic areas presenting with acute manifestations (fever, eosinophilia, acute adenolymphangitis) or chronic manifestations (chyluria, hydrocele, lymphedema). Additional features can be eosinophilia, elevated IgE, microscopic hematuria, and/or proteinuria.
Definitive diagnosis is made by any one of the following: detection of filarial antigen in circulation (W.bancrofti only), demonstration of microfilaria or microfilarial DNA in the blood, detection of adult worms in lymphatics, or demonstration of microfilaria or adult worms in the tissue biopsy or cytological specimens.
Ultrasound may show continuous motion of living worms -“filarial dance” sign; lymphoscintigraphy is helpful to determine lymphatic anatomy.
W.bancrofti, B.malayi and B.timori can be differentiated based on their morphologic characteristics on blood smear.
Evaluation for coinfection: Individuals from areas endemic for loiasis or onchocerciasis should be screened for coinfection. Treatment with DEC can trigger severe inflammatory reactions in such patients.
Treatment
Microfilaria are more responsive to treatment than adult worms, which presents a challenge. Drug of choice is diethylcarbamazine (DEC). Doxycycline has both micro- and macrofilaricidal activity (including cases of coinfection with loiasis or onchocerciasis), and also acts against Wolbachia. Coinfections with loiasis and onchocerciasis are an excellent indication for Ivermectin.
Control and Prevention
Mass drug administration strategy is used to decrease the microfilaria levels in the blood below that required for transmission by mosquitoes.Vector control and personal protection measures are important. Vaccine development is underway, but has proven difficult due to biological hurdles and poor funding.
In the News: Anti-Wolbachia Treatment
Wolbachia, an indispensable endosymbiont for filaria, is an exciting and promising target for treatment and control of filarial disease. A first phase I human trial has recently begun with a novel anti-Wolbachia compound.
– Executive Team Members for this region: Joel Topf @kidney_boy and Anna Vinnikova @KidneyWars
How to Claim CME and MOC
US-based physicians can earn 1.0 CME credit and 1.0 MOC per region through NKF PERC (detailed instructions here). The CME and MOC activity will expire on June 1, 2022.
Click to read more NephMadness 2022 Regions


Excellent overview of parasitic infections and the kidney . Very interesting to know about the history of these infections dating back to the Before Christian (BC) era . The infographics are also very informative and eye-catching. In these times of easy travel across continents, the conditions need not be really limited to the endemic areas alone and it is important for all physicians and nephrologists to be aware of the different modes of presentation of these diseases.
If I may point out certain factual errors here,
1. Alphonse Laveran got the Nobel Prize in Physiology or Medicine in the year 1907 for his work in protozoa causing diseases(https://www.nobelprize.org/prizes/medicine/1907/laveran/biographical/)
2.Nobel Prize was instituted in 1901 .
3. In 1902, Sir Ronald Ross got the Nobel prize in Physiology or Medicine for his work on plasmodium parasites in malaria causation (https://www.nobelprize.org/prizes/medicine/1902/summary accessed on 21 March 2022). Incidentally he was the first India-born individual to win the Nobel , though he was a British Origin Doctor and the research where he demonstrated the malarial parasites in the gastrointestinal tract of female anopheline mosquitoes was carried out while he was posted as the British army medical officer in Secunderabad (https://en.wikipedia.org/wiki/List_of_Indian_Nobel_laureates#Indian_Nobel_Laureates_of_foreign_origin accesssed on 21 march 2022)
4. MCGN and MPGN were terms used interchangeably to describe the picture of MPGN in the past , and I believe that it should have been
actually written as Mesangioproliferative instead of MCGN in the first visual abstract
Thank you for the comments regarding the Parasites region. The points 1-3 are well taken. However, our statement that Alphonse Laveran received Nobel prize for the discovery of plasmodium, work done in 1880, remains correct. Unfortunately, space and focus limitations do not allow us to give full credit to all pioneers of malaria research. We appreciate the point #4, and will make appropriate changes to the visual abstract in question.