Anemia in Patients Approaching Dialysis: Moving From When to TREAT to Whether to TREAT
In late 2009, the primary results of the placebo-controlled Trial to Reduce Cardiovascular Events with Aranesp Therapy (TREAT) were published. TREAT randomized 4,038 patients with anemia and type 2 diabetes with estimated GFR (eGFR) of 20 to 60 ml/min/1.73 m2 to injections of placebo or Aranesp, an erythropoiesis-stimulating agents (ESA), and tracked cardiovascular (CV) events and deaths.

Copyright: ktsdesign / Shutterstock.com
Previous nephrology outcomes trials compared higher versus lower hemoglobin targets, and consequently higher versus lower ESA doses. As Dr. Marc Pfeffer, the lead investigator of TREAT has said, in such trials the drug (and treatment) always wins. Not so in TREAT, which showed that ESAs had serious risks and limited benefits. This led to a major shift in use of ESA in the anemia of chronic kidney disease (CKD).
But doubts about not treating anemia in CKD with ESA therapy linger. Anemia strongly associates with worse outcomes, and the severity of anemia accelerates as patients approach and transition on to dialysis. Also, CV events and mortality are particularly high during the first 3 months of dialysis. Surely, in these very high-risk patients with rapidly worsening anemia, ESA therapy might be beneficial.
In a recent study published in AJKD, Mc Causland et al examine whether randomization to ESA versus placebo in a subgroup of TREAT patients led to better outcomes the first 180 days on dialysis. This is a marvelously well-written study, and well worth a thorough read.
The patients in this analysis had exited TREAT by reaching the end-stage renal disease (ESRD) endpoint. Normally, I say there are lies, damn lies, and subgroup analyses. Unless a subgroup is predefined and the study powered to examine outcomes in the subgroup, results that deviate from the overall group outcome should be viewed as “exploratory,” which is a nice way of saying “misleading.” However, because ESA therapy did not delay or accelerate progression to ESRD, this comparison may be reasonable.
The ESA vs placebo-treated groups should be similar in characteristics, and Table 1 in the article demonstrates they are comparable. While there are some differences between groups, they do not invariably favor one arm over the other. The eGFR “at the time of dialysis initiation” seems shockingly high (median 15-16 ml/min/1.73 m2), but may reflect the last on-study labs weeks to months before actual dialysis initiation. Compared to the overall TREAT subjects, patients in this subgroup analysis are more likely to be men (~53% vs ~43%), black race (~29% vs ~20%), and have heart failure (~45% vs ~33%).

Participant Characteristics According to Randomized Treatment Assignment at the Time of Maintenance Dialysis Initiation. Table 1 from Mc Causland et al, AJKD, © National Kidney Foundation.
Another reason to accept this subgroup analysis is that the results parallel the overall TREAT results. The investigators found randomization to ESA achieved a higher hemoglobin (mean hemoglobin 11.3 vs 9.5 g/dL) at the time of dialysis start, but did not reduce deaths (10.4% vs 9.6%), death from CV causes (5.0% vs 4.5%), nonfatal myocardial infarction, or heart failure.
As in the primary TREAT trial, the authors here found that strokes were significantly higher in the ESA group (2.8% vs 0.3%; hazard ratio, 8.6; 95% confidence interval, 1.1-68.7). I would not run out and tell CKD patients nearing dialysis that ESA therapy will increase their risk of stroke 8.6-fold, as the small number of strokes (8 vs 1) leaves great uncertainty about the magnitude of the risk. Do tell them that use of ESAs may increase stroke risk ~2-fold, and ~3-fold if patients have suffered prior strokes.
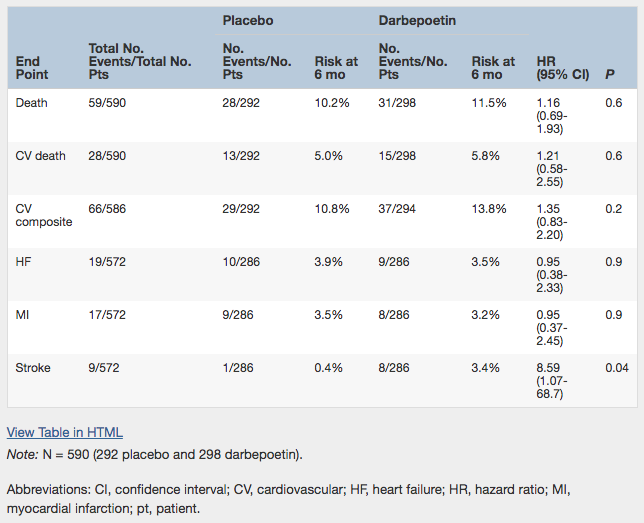
Association of Randomized Treatment Assignment With Death, CV Death, Heart Failure, Myocardial Infarction, and Stroke During the First 6 Months After Maintenance Dialysis Initiation. Table 2 from Mc Causland et al, AJKD, © National Kidney Foundation.
If giving ESA to patients as they approach dialysis doesn’t improve any of the above outcomes, should we treat to reduce fatigue? In Table 1, the EuroQol 5-dimension visual analogue score and Functional Assessment of Cancer Therapy fatigue score were identical between groups despite a 1.8 g/dL difference in hemoglobin. Even in the primary TREAT results, the improvement in fatigue reported was quite small (placebo-subtracted difference of 1.4 points on a 100-point scale) and could reflect unblinding errors.
ESA therapy significantly reduced transfusions in the primary TREAT results, while Mc Causland’s analysis did not examine this potential benefit. In the primary TREAT results, the placebo arm’s transfusion rate was 29% in North America compared to 16-19% in other major regions. In the ESA arm, transfusion rates were similar (~16%) in all regions. Thus, only in North America (and overwhelmingly in the US) did ESA therapy significantly reduce transfusions.
Avoidance of transfusions is most important in transplant candidates, who are generally healthier and less anemic than the TREAT population. To reduce transfusions, we should be more generous in prescribing iron, more conservative in ordering transfusions, and maybe use ESA therapy when the hemoglobin falls below 9.0 g/dL. The placebo arm in TREAT was rescued with ESA whenever the hemoglobin fell below 9.0 g/dL, and reverted to placebo once hemoglobin was above 9.0g/dL. Overall, 46% of placebo arm patients received at least 1 dose of ESA.
Use of any ESA in US patients prior to initiating dialysis has fallen to ~13%, after peaking at ~33% in 2005. This analysis by the TREAT investigators reinforces the original suggestion that low utilization of ESA in non-dialysis CKD patients is prudent.
– Post prepared by Daniel W. Coyne, AJKDBlog Guest Contributor. Follow him @DrDanMO.
To view Mc Causland et al (subscription required), please visit AJKD.org.
Title: Treatment of Anemia With Darbepoetin Prior to Dialysis Initiation and Clinical Outcomes: Analyses From the Trial to Reduce Cardiovascular Events With Aranesp Therapy (TREAT)
Authors: F.R. Mc Causland , B. Claggett, E.A. Burdmann, G.M. Chertow, M.E. Cooper, K.-U. Eckardt, P. Ivanovich, A.S. Levey, E.F. Lewis, J.B. McGill, J.J.V. McMurray, P. Parfrey, H.-H. Parving, G. Remuzzi, A.K. Singh, S.D. Solomon, R.D. Toto, and M.A. Pfeffer
DOI: 10.1053/j.ajkd.2018.10.006

Leave a Reply