#NephMadness 2021: Anemia Region
Submit your picks! | NephMadness 2021 | #NephMadness | #AnemiaRegion
Selection Committee Member: Nupur Gupta @nupur_nephron
Nupur Gupta is an Assistant Professor of Medicine at Indiana University, where she did her Nephrology training. She is the medical director of the home dialysis program at her university. She currently serves on the ASN Quality Committee and ASN Home Dialysis focus group. Her area of research is practical application of Peritoneal Dialysis and its complication.
Writer: Arun Rajasekaran @Nephronpower
Arun Rajasekaran is a third-year nephrology fellow at the University of Alabama at Birmingham. He is passionate about academic nephrology and is involved in collaborative and online medical education. His clinical and research interests lie in glomerular diseases. He is a current AJKD Editorial Intern.
Competitors for the Anemia Region
Oral Iron vs IV Iron
ESAs vs HIF Stabilizers

Copyright: Vladimir Mucibabic / Shutterstock
“By far the most remarkable character of the blood in the advanced stage of the Bright’s disease (nephritis) is a gradual and rapid reduction of its colouring. No other natural disease came as close to hemorrhage for impoverishing the red particles of the blood.”
Sir Robert Christison, 1839
Ever since the 1800s, anemia has been recognized as a significant complication of chronic kidney disease (CKD), typically developing with even moderate reductions in kidney function. A statistically significant decrease in hemoglobin (Hb) becomes apparent among men starting at a creatinine clearance less than 70 mL/min and among women starting at creatinine clearance less than 50 mL/min. Anemia severity increases as CKD worsens, though the relationship is not linear and the rate of decline differs between patients. Around 50% of predialysis patients with CKD have anemia.
Anemia in CKD is associated with several adverse events. Anemia (from any cause) results in impaired oxygen delivery resulting in fatigue, shortness of breath, and limited activity. However, it must be noted that these are also symptoms associated with uremia. Therefore it is not surprising that anemia in patients receiving dialysis is associated with worse quality of life. Several studies suggest that anemia in CKD is associated with left ventricular hypertrophy, suggesting a direct link to cardiovascular disease progression. Other studies have associated anemia with increased mortality in patients with kidney disease. However, it should be emphasized that while the association of anemia with left ventricular hypertrophy and mortality exists, randomized clinical trials have failed to show an improvement in either of these parameters after anemia is corrected. This suggests that other factors are contributing to excess cardiovascular and mortality risk in patients with CKD, and anemia is confounding these results.
Several mechanisms underlie the pathogenesis of anemia in CKD. These include relative deficiency of erythropoietin (EPO), absolute and functional iron deficiency, increased blood loss, and reduced erythrocyte lifespan.

Common Causes of Anemia in CKD. Box 1 from Fishbane et al, AJKD © National Kidney Foundation.
The NephMadness anemia region is dedicated to the treatment of anemia in kidney disease (both for those receiving dialysis and those not). We will discuss the treatment of iron deficiency anemia (oral vs intravenous iron repletion), the use of erythropoietin stimulating agents (ESAs), and the novel hypoxia inducible factor-prolyl hydroxylase inhibitors (HIF-PHIs) in patients with kidney disease.
Oral Iron vs IV Iron
Oral Iron

Copyright: Repina Valeriya / Shutterstock
Oral iron formulations are cheap, easy to administer, and sometimes well-tolerated. Additionally, they show good efficacy in increasing hemoglobin levels in patients with CKD. With all these positive attributes, you would be well-advised to pick team Oral Iron for the win!
Iron deficiency can be defined as either absolute or functional. Absolute iron deficiency is due to significantly reduced or absent iron stores in the bone marrow, liver, and spleen. Among patients with CKD, absolute iron deficiency is defined arbitrarily using serum transferrin saturation or ferritin (see below). Absolute iron deficiency can be caused by blood loss from apparent or occult gastrointestinal sources and procedures (surgeries or hemodialysis itself), or very rarely from reduced iron intake in the diet.
Functional iron deficiency is characterized by sufficient iron stores (demonstrated by stainable iron in the bone marrow) but inadequate incorporation of iron into erythroid precursors, even with the exogenous administration of ESAs. Functional iron deficiency is also characterized using serum transferrin saturation or ferritin.

Diagnosis of absolute and functional iron deficiency in kidney disease.
*Neither TSAT nor ferritin are very good predictors of bone marrow iron or response to iron supplementation.
The pathophysiology of functional iron deficiency is thought to occur secondary to increased levels of hepcidin, a key regulator of iron homeostasis. The elevated hepcidin levels in patients with CKD are a consequence of both a chronic inflammatory state and decreased kidney clearance of hepcidin itself. Hepcidin binds to ferroportin, a transmembrane conduit for the transfer of cellular iron to plasma, resulting in its internalization and degradation. Consequently, there is decreased intestinal iron release from duodenal cells, the liver, and macrophages (Figure 2). These all conspire towards reduced available iron for use in erythropoiesis. Importantly, this limits the response of ESA therapy towards correcting anemia.

Regulation of iron availability by hepcidin. Created with BioRender.com.
The KDIGO guidelines to managing iron deficiency anemia:
| 2.1.2: For adult CKD patients with anemia not on iron or ESA therapy we suggest a trial of IV iron (or in CKD not on dialysis patients alternatively a 1–3 month trial of oral iron therapy) if (2C): • an increase in Hb concentration without starting ESA treatment is desired* and • TSAT is ≤ 30% and ferritin is ≤500 ng/ml (≤500 mg/l) |
| 2.1.3: For adult CKD patients on ESA therapy who are not receiving iron supplementation, we suggest a trial of IV iron (or in CKD not on dialysis patients alternatively a 1–3 month trial of oral iron therapy) if (2C): • an increase in Hb concentration** or a decrease in ESA dose is desired*** and • TSAT is ≤ 30% and ferritin is ≤ 500 ng/ml (≤ 500 μg/l) |
| 2.1.4: For CKD not on dialysis patients who require iron supplementation, select the route of iron administration based on the severity of iron deficiency, availability of venous access, response to prior oral iron therapy, side effects with prior oral or IV iron therapy, patient compliance, and cost. (Not Graded) |
| 2.1.5: Guide subsequent iron administration in CKD patients based on Hb responses to recent iron therapy, as well as ongoing blood losses, iron status tests (TSAT and ferritin), Hb concentration, ESA responsiveness and ESA dose in ESA treated patients, trends in each parameter, and the patient’s clinical status. (Not Graded) |
KDIGO recommendations for iron therapy in CKD
*Based on patient symptoms and overall clinical goals, including avoidance of transfusion, improvement in anemia-related symptoms, and after exclusion of active infection.
**Consistent with Recommendations #3.4.2 and 3.4.3.
***Based on patient symptoms and overall clinical goals including avoidance of transfusion and improvement in anemia-related symptoms, and after exclusion of active infection and other causes of ESA hyporesponsiveness.
Reprinted from the 2012 Kidney Disease: Improving Global Outcomes (KDIGO) Clinical Practice Guideline for Anemia in Chronic Kidney Disease. Used with permission.
However, traditional iron formulations were used in most of the studies that guided the KDIGO recommendations. With the advent of newer iron formulations, are these recommendations still valid? What are the benefits of one iron formulation over the other? And, can we use newer oral iron agents for iron repletion in patients with CKD and patients with kidney failure?
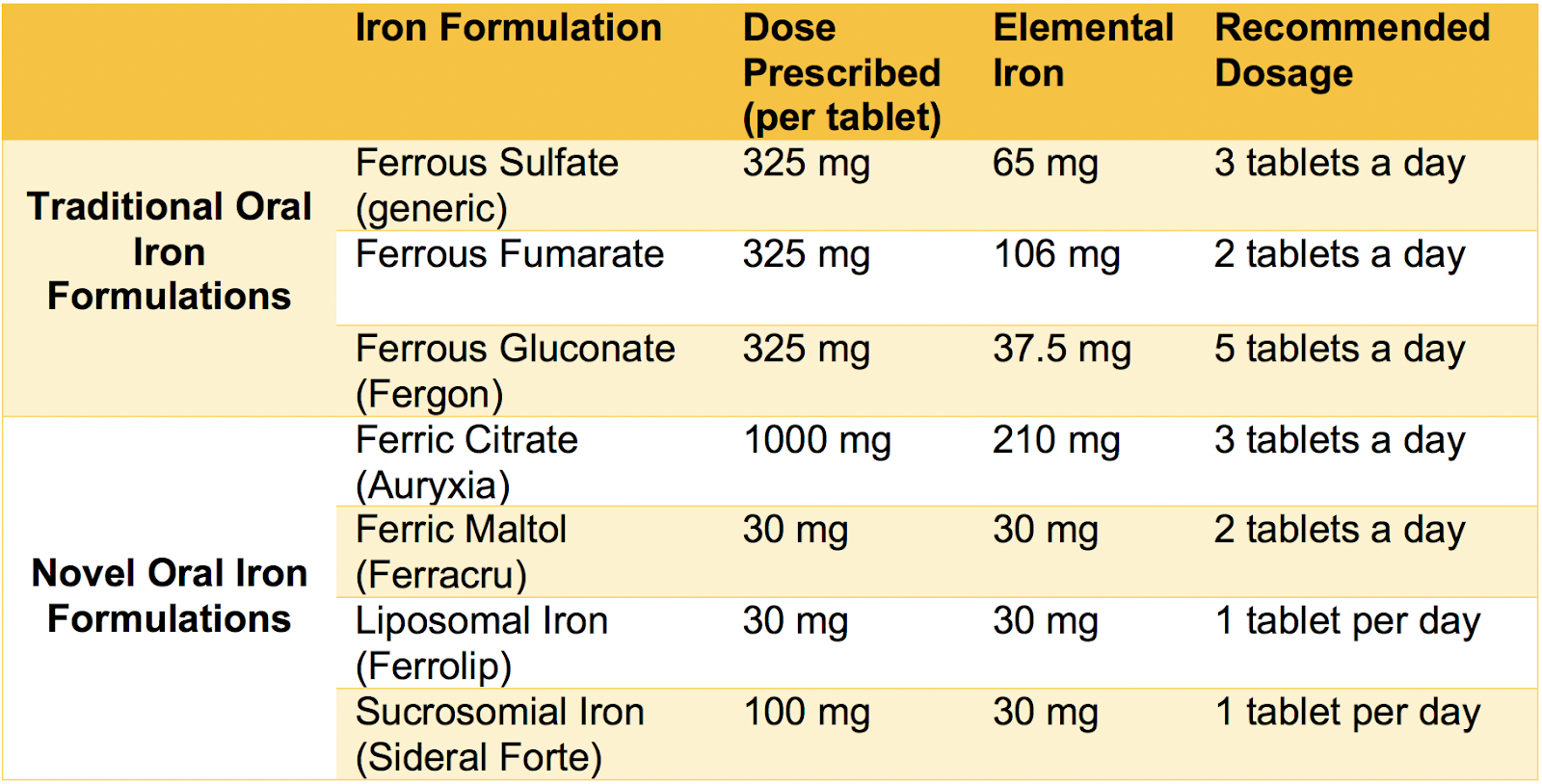
Oral iron agents used for treating anemia in patients with kidney disease. Based on Table 1 from Pergola et al, ACKD © National Kidney Foundation.
Ferrous (Fe2+) sulfate is a commonly used oral iron formulation to treat iron deficiency anemia in patients with CKD. It contains 65 mg of elemental iron per 325 mg tablet. Ferrous sulfate is given three times a day between meals. The oral bioavailability of ferrous iron formulations is typically low with some variability depending on the preparations. Gastrointestinal adverse events (nausea, diarrhea, constipation) limit ferrous preparations’ tolerability, leading to lower delivery of elemental iron and poor patient compliance.
How does oral ferrous sulfate compare to intravenous (IV) iron? The REVOKE trial randomized 136 participants with CKD stage 3 and 4 to oral ferrous sulfate (325 mg three times daily for 8 weeks) versus IV iron sucrose (200 mg every 2 weeks, total 1 gram). The trial found that Hb levels improved in both groups and were not statistically different. Ferritin levels were significantly higher in the IV iron group as compared to the oral iron group. Nevertheless, the trial was terminated early due to a high risk of serious adverse events in the group that received IV iron, in particular cardiovascular events (adjusted incidence rate ratio for IV/PO 2.51 [1.56-4.04], P < 0.001). A potential hypothesis for the latter is that IV iron can induce oxidative stress, which in turn can result in endothelial dysfunction, atherogenesis, and plaque instability. This hypothesis, however, has not yet been definitively proven. Nevertheless, this observation should encourage us to adopt an oral iron first strategy as we await more studies on IV iron’s potential cardiovascular consequences.
Newer oral formulations include ferric citrate, sucrosomial iron, and ferric maltol. We will discuss them and examine how they compare with IV formulations.
Ferric (Fe3+) citrate was initially approved as an oral phosphate binder to treat hyperphosphatemia in patients with kidney failure. It was later approved by the United States Food and Drug Administration (FDA) to treat iron deficiency anemia in patients with CKD not on dialysis. This was based upon data showing that ferric citrate increased circulating iron concentrations and Hb levels in patients with CKD with iron deficiency anemia, as compared with placebo. The absorption of ferric iron formulations can be 3-4 times lower than ferrous sulfate preparations, given the physiology of enteral iron absorption and the low solubility of ferric iron in the alkaline milieu of the duodenum. However, each 1 g tablet of ferric citrate contains 210 mg of elemental iron, potentially allowing for a greater total quantity of iron delivery than ferrous sulfate without resulting in more adverse effects. Ferric citrate is generally well tolerated and associated with gastrointestinal adverse effects in 10-20% of patients. In a randomized trial, treatment with ferric citrate (2 g three times a day with meals) as compared to ferrous sulfate (325 mg three times a day) for 12 weeks resulted in a more significant mean increase in transferrin saturation (TSAT) and ferritin concentrations in individuals with moderate to severe CKD and iron deficiency anemia without any differences in adverse event profile. There were no between-group differences in the 12-week change in Hb.

Visual abstract by Eric Au on Womack et al.
One study that evaluated the efficacy of oral ferric citrate as a phosphorus binder in patients with kidney failure on dialysis also examined iron and anemia parameters. This was a phase 3 randomized clinical trial involving 441 patients on hemodialysis who were randomized to receive oral ferric citrate versus control (calcium acetate or sevelamer alone or in combination) for phosphorus control. Ferric citrate-treated patients required less cumulative IV iron than those receiving active control over the 52-week randomized period (median dose 12.9 vs 26.88 mg/week; P < 0.001). At week 52, 85% of patients in the ferric citrate group received no IV iron as compared to 69% in the control group. Likewise, the cumulative ESA use was lower in the ferric citrate group (median dose 5303 vs 6954 U/wk for control; P = 0.04). Hb levels were statistically higher on ferric citrate. Based on this, ferric citrate looks to be a two-way player; it is a safe phosphate binder and iron supplement that increases iron stores and reduces IV iron and ESA use while maintaining target Hb in patients on hemodialysis.

Visual abstract by Sinead Stoneman on Lewis et al.
Sucrosomial iron is an oral iron formulation that contains ferric pyrophosphate with a phospholipid bilayer membrane coating. A randomized trial of patients with iron deficiency anemia and CKD stage 3-5 not on dialysis showed that a short course of oral sucrosomial iron was as sufficient as IV ferrous gluconate in correcting anemia. Oral sucrosomial iron also had a favorable tolerability profile. While Hb levels increased faster in the IV iron group, at the end of the 3 months of follow-up Hb levels in both groups were comparable. The IV iron group had an increase in TSAT and ferritin which was not seen in the sucrosomial iron group, and this may have contributed to Hb levels dropping faster in the oral iron group following discontinuation of iron supplementation. Of note, sucrosomial iron was associated with gastrointestinal side effects in 12% of participants but compliance remained high, at 96%. In the IV iron group, 18% experienced gastrointestinal side effects, 12% had infusion site reactions, 18% experienced headache, and 12% developed hypotension.
Ferric maltol is a novel oral iron therapy that is approved for the treatment of iron deficiency anemia in adults in Europe and the United States. It is thought to have better absorption owing to its composition, which has both hydrophilic and lipophilic properties that can be more efficiently taken up by enterocytes. Ferric maltol was assessed in a randomized, double-blind, placebo-controlled phase 3 trial in patients with iron-deficient anemia and CKD (N = 168; AEGIS-CKD study; NCT02968368). The study has been completed but results have not been published yet. However, in patients with inflammatory bowel disease, ferric maltol significantly increased Hb as compared to placebo and had a similar safety profile.
In summary, oral iron formulations are generally cheaper and easier to administer than IV formulations. However, they have historically had limited efficacy due to limited absorption and are associated with gastrointestinal side effects. Newer formulations are well-tolerated and have better efficacy as compared to the older formulations. But are they a match to IV iron therapy? In the next section, we take you through the pros and cons of IV iron so that you can choose the winner of this first matchup.
IV Iron

Copyright: yongyut rukkachatsuwa / Shutterstock
Cardiovascular disease, bacterial infections, hypophosphatemia… IV iron may have a bad reputation, but when it comes to fast and efficient iron delivery, it simply can’t be beat. Read all about what makes IV iron a contender in this year’s NephMadness competition.
IV iron formulations differ in their physicochemical properties, biologic contents, and amount of iron that can be administered during a single session. Older IV iron formulations—including iron sucrose, iron gluconate, and iron dextran—rapidly release iron from an iron-carbohydrate moiety (Table 5). Consequently, the dose of iron that can be administered per session is limited. Patients receiving maintenance hemodialysis who regularly attend a dialysis center and have vascular access are able to receive IV iron formulations several times a week if needed. However, this need for several sessions to administer older IV iron can be problematic for patients who are not on dialysis. Patients with CKD not on dialysis may benefit from the newer IV iron formulations (ferric carboxymaltose, iron isomaltoside, ferumoxytol) (Table 5), which permit a relatively large quantity of iron to be given during a short time period, and in a single session.
The FERWON-NEPHRO trial, a randomized open-label multicenter trial in the United States, compared the safety and efficacy of the newer IV formulation (iron isomaltoside 1000/ferric derisomaltose [IIM]) to the older IV formulation (iron sucrose) in 1,538 iron-deficient patients with CKD not on dialysis. Patients were randomized to receive either a single dose of 1000 mg IV IIM or IV iron sucrose administered as 200 mg injections up to 5 times within a 2 week period. IV IIM was associated with a more robust elevation in Hb during the first 4 weeks (P ≤ 0.021) of treatment. However, the change in Hb at week 8 was not different between groups. There was no significant difference in the incidence of serious or severe hypersensitivity reactions between groups . Composite cardiovascular adverse events were significantly reduced in the IV IIM infusions versus the IV iron sucrose group (4.1% vs 6.9%; P = 0.025). Hypophosphatemia was rare in both treatment groups, with no individual having a serum phosphate < 1 mg/dL. Thus, when compared with multiple doses of IV iron sucrose, a single dose of IV IIM induced a non-inferior 8 week hematological response, comparably low rates of hypersensitivity reactions, and a significantly lower incidence of composite cardiovascular adverse events. However, long-term data are needed to confirm the above findings, as this study was only 8 weeks in duration.

Visual abstract by Michelle Lim on Bhandari et al.
The most common side effects of IV iron formulations include transient hypotension, nausea, vomiting, and abdominal discomfort. These side effects are generally self-limited and do not require any specific intervention other than slowing the iron infusion rate. Hypersensitivity reactions to IV iron, including anaphylaxis, were more prevalent when iron dextran was being used and are rare nowadays. Longer-term concerns regarding IV iron encompasses elevated oxidative stress, progressive atherogenesis, cardiovascular adverse effects, and immune dysregulation.
Infectious risk is a concern with the use of IV iron. This risk is two-fold; iron is required for bacterial growth and excess iron has been associated with neutrophil and T cell dysfunction. Several studies have been published on the independent association between iron administration and higher rate of infections in dialysis patients. However, a prespecified secondary analysis of the randomized controlled PIVOTAL trial showed that rates of infection did not differ between the high- or low-dose iron group. The higher infection risk described in older studies may then theoretically be circumvented by giving maintenance doses of iron as opposed to bolus doses of iron, regardless of the dose of iron administered. Further studies are needed to confirm this.
Hypophosphatemia has been a reason for concern associated with the use of specific iron formulations (notably with ferric carboxymaltose).The underlying pathophysiology involves a rapid activation of fibroblast growth factor 23 (FGF23), which results in kidney phosphate wasting, decreased levels of vitamin D and calcium, and increased levels of parathyroid hormone. Therefore, for this pathway to work, one needs a functioning kidney, making hypophosphatemia a rare occurrence in patients with kidney failure. Hypophosphatemia typically becomes detectable a week after administration with lowest levels occurring by the second week following administration, and in some cases can be prolonged for several months.
Nevertheless, there are many advantages to using IV iron, including greater potential of iron delivery to the bone marrow, mitigated concerns regarding reduced adherence to oral iron formulations, and mitigated concerns regarding reduced iron absorption from the duodenum, especially with declining glomerular filtration rate (GFR) and increasing hepcidin levels. Further, the recently published AFFIRM-AHF study in patients with iron deficiency and heart failure did not show an increased risk of cardiovascular disease with IV iron as compared to placebo, an important finding that mitigates concerns about cardiovascular safety of IV iron preparations.
The FIND-CKD trial (Ferinject assessment in patients with Iron deficiency anemia and Non-Dialysis-dependent Chronic Kidney Disease) was a multicenter, prospective, randomized, three-arm study which randomized patients with CKD (GFR < 60 mL/min/1.72m2) at a 1:1:2 ratio to receive IV ferric carboxymaltose targeting a ferritin of 400–600 µg/L, IV ferric carboxymaltose targeting a ferritin of 100-200 µg/L, and oral ferrous sulfate at a dose of 100 mg iron twice daily, respectively. After a 56 week follow-up period, 32% of those receiving oral iron as compared to only 24% of those receiving high ferritin IV iron (HR 0.65 [95% CI, 0.44–0.95], P = 0.026) required a change in anemia management. All groups had an increase in Hb. However, the high ferritin IV group had higher Hb as compared to the oral group, showed a faster increase in Hb levels after drug initiation, and was the group most likely to have an increase in Hb ≥1 g/dL. Adverse events were reported by participants in all 3 groups. Of those reported, 14%, 15% and 29% were thought to be related to the high ferritin IV, low ferritin IV, and oral iron, respectively. Unlike the REVOKE trial (discussed above) in which IV iron therapy was associated with higher risks of cardiac events, cardiac events were similar between groups in this study. Up to 8% of participants who received oral iron but only 2% of participants who received IV iron discontinued the drug due to intolerance.

Visual abstract by Krithika Mohan on Macdougall et al.
Finally, we have to talk about The PIVOTAL study. Even though this trial did not compare IV to PO iron, it is an essential study that guides how we administer supplement IV iron. PIVOTAL randomized 2,141 patients on hemodialysis to either proactive high dose IV iron supplementation (400 mg monthly, unless the ferritin concentration was > 700 μg per liter or the transferrin saturation was ≥40%), or reactive low-dose IV iron supplementation (0 to 400 mg monthly, with a ferritin concentration of < 200 μg per liter or a transferrin saturation of <20% being a trigger for iron administration). The high-dose regimen resulted in a significantly lower risk of death or major nonfatal cardiovascular events as compared with that observed with the reactive, low-dose regimen. This dosing strategy also resulted in significantly lower doses of ESAs used and a lower incidence of blood transfusions, whereas the incidence of infection and hospitalization for any cause did not differ significantly between the two treatment groups.

Visual abstract by Dominique Tomacruz on Macdougall et al.
This region and the comprehensive evidence it brings in can create a paradigm shift in how we treat anemia of CKD. Are we converts who will use the newer oral iron formulations to treat our patients or will we persist in using IV iron until new evidence emerges?
ESAs vs HIF Stabilizers
ESAs
Why would I enter into a sport and then dope myself up and risk my life again? That’s crazy. I would never do that. No way.
– Lance Armstrong, 2005
We have observed, with Mlle. Deflandre, that regeneration of blood after bloodletting is under the influence of a humoral process… we give this substance the generic name Hemopoietine.
– P. Carnot and C. Deflandre, French investigators who reported in 1906 that plasma from anemic rabbits injected into normal animals caused an increase in red blood cell production.

Copyright: Dewald Kirsten / Shutterstock
In 2012, we found out that Lance Armstrong had in fact been doping all along, and erythropoietin (EPO) was a key element in helping him to win 7 Tour de France Championships. But Erythropoiesis-stimulating agents (ESAs) haven’t only helped Armstrong, they’ve also helped countless patients with kidney disease avoid blood transfusions and improve their quality of life. Consequently, ESAs have the opportunity to win this championship fair and square!
Throughout the 1940s and the 1950s, experiments by Krumdieck et al pointed to a protein in the plasma that increases red blood cell production. In 1957, Jacobson et al demonstrated that the kidney produces what became known as EPO. EPO is an amino acid glycoprotein hormone formed and secreted by kidney interstitial cells to stimulate bone marrow red blood cell production in response to tissue hypoxia. Human EPO was extracted from the urine of patients with anemia in 1977, and the human EPO gene was successfully cloned in 1983. In 1989, recombinant EPO (formed by cloning the human EPO gene) was approved by the FDA for use. Today, recombinant EPO drugs are known as ESAs.
| Available ESAs in the US | Marketed Name |
| Epoetin alfa | Procrit, Epogen |
| Darbepoietin alfa | Aranesp |
| Epoietin alfa-epbx | Retacrit |
Available ESAs in the US
ESAs are effective in treating anemia in most patients with kidney disease. The resultant increase in Hb level alleviates many anemia-related symptoms, and improves health-related quality of life measures including depression, neurocognitive function, and fatigue. Furthermore, among patients with severe anemia receiving hemodialysis, ESAs reduce transfusion need (Figure 3) and left ventricular hypertrophy, factors independently associated with increased morbidity and mortality in this population.
Nancy Spaeth was one of the first dialysis patients to receive EPO in 1987. She shared her experience with EPO as a testament to the impressive effects of this drug.
Before dialysis, when I was 15, in 1963, I took first place skiing in a coed slalom race. I beat the boys. Two years later, I couldn’t walk from the lodge to the lift.
I started on dialysis in 1966. I don’t remember how many transfusions I had. It was many, at least one per month. I had to be desperate before the doctor would order blood.
I was part of the phase III clinical trial in 1987. I was on home dialysis, raising two children, alone. My hematocrit (HCT) was 11-15 percent; I couldn’t walk up the stairs. I had to crawl on my hands and knees because I was so out of breath. Once I started on the drug, I could tell there was a change within two weeks. Within 30 days, my HCT was 40! My daughter said, ‘All of a sudden you were up and going.’ Eventually, I could run up the stairs.
I used to be asked, ‘What are you doing?’ I answered, ‘What can I do? My body has me in prison.’ Epo changed all of that. What a difference it made in my life!

Reductions in blood transfusions in dialysis coincided with introduction of EPO.
Data from USRDS Annual Data Report, chapter on Clinical Indicators & Preventive Health.
The use of ESAs to treat anemia in patients with CKD has been a tumultuous journey. This was highlighted by the Normalization of Hemoglobin region team in NephMadness 2016. While the use of ESAs was immediately embraced and the reduction in red blood cell transfusions quick, results of randomized clinical trials put a halt to this enthusiasm. The Normal Hematocrit Cardiac Trial published in 1998 (NHCT), the Correction of Hemoglobin and Outcomes in Renal Insufficiency published in 2006 (CHOIR) study, and the Cardiovascular Risk Reduction by Early Anemia Treatment with Epoetin Beta published in 2006 (CREATE) study were important studies that showed that ESAs contribute to adverse cardiovascular, cerebrovascular, and cancer outcomes.
Briefly, the NHCT trial (a randomized controlled trial of 1223 patients on dialysis randomized to hematocrit of 42% versus 30%) found an increase in vascular access thrombosis and a trend toward increased death and myocardial infarctions among patients in the higher hematocrit group. The CHOIR trial confirmed an increased risk of a composite endpoint (encompassing death, myocardial infarction, hospitalization for congestive heart failure, and all cause-hospitalization) among patients with CKD not on dialysis in the higher Hb target (13-13.5 g/dL as compared to 10.5-11 g/dL). The CREATE study did not demonstrate differences in cardiovascular events between patients with CKD not on dialysis assigned to either full or partial correction of anemia. Still, a prespecified secondary analysis revealed a higher risk of kidney failure necessitating dialysis among those in the higher Hb target group. The revised guidelines have tempered the use of ESAs because of these risks.
ESA resistance or hyporesponsiveness is used to characterize patients who do not achieve the desired Hb concentration despite obtaining higher than regular doses of ESAs or who require increasingly higher doses to maintain a target Hb concentration. The Kidney Disease Outcomes Quality Initiative (KDOQI) criteria for ESA hyporesponsiveness is the inability to achieve or maintain a desired Hb concentration using a maximum dose of 450 units/kg per week intravenous EPO or 300 units/kg per week subcutaneous EPO. This is important as most of the adverse effects described with ESAs, including increased mortality, cardiovascular events including strokes, increased risk of hemodialysis vascular access thrombosis, and hypertension are associated with higher ESA doses in patients hyporesponsive to ESA. Some of the common causes for ESA resistance include absolute and functional iron deficiency (contributes to 10-20% of ESA hyporesponsiveness), infection or inflammation, inadequate dialysis, severe hyperparathyroidism, and malignancy or hematologic disorders. Most importantly though, is that ESA resistance is a surrogate to the underlying causes of ESA resistance, which themselves are thought to be the main driver of mortality and other complications.
Pure red cell aplasia is a rare adverse effect of ESAs observed with older subcutaneous formulations (especially with a particular epoetin alfa product, Eprex) but can occur with intravenous formulations as well. The underlying etiology is likely related to organic compounds in the ESA acting as adjuvants, that lead to the formation of anti-EPO antibodies. Given that this phenomenon is extremely rare, dialysis patients should not be routinely screened for the presence of anti-EPO antibodies.
Another big concern is the use of ESA in patients with current or past malignancies. However, there is paucity of data to allow us to make any definitive conclusions. Most studies demonstrate that the association between ESA and mortality in patients with cancer occurs at higher Hb targets (> 12 g/dL) and is not associated with cancer progression. Therefore, as in CKD patients without malignancy, the mortality risk seems to be conferred secondary to high Hb targets and high doses of ESA contributing to higher thromboembolic risk. In our patients with CKD and cancer, the decision to start or continue ESA has to be a risk-benefit conversation between the physician and the patient.
KDIGO guidelines suggest considering ESA use only if Hb is less than 10 g/dL and addressing all potentially correctable etiologies of anemia before ESA use, including repleting iron stores if iron deficient. Generally, Hb levels are maintained between 10 and 11.5 g/dL in patients on dialysis treated with ESA. Secondary to excessive cardiovascular and stroke risk of ESAs, it is important to use the lowest dose possible to achieve the desired Hb level. Moreover, when ESA hyporesponsiveness develops, reducing the cumulative dose of ESA is paramount.
Therapy is individualized in some patients who may have improvements in quality of life at Hb greater than 11.5 g/dL, particularly in healthy young individuals willing to accept the risks associated with higher Hb targets. Significant exceptions include patients with active malignancy, particularly those in whom cure is anticipated or those who have had a stroke, since such patients may be at higher risk for adverse effects from ESAs. Management in these patients should be individualized after careful consideration and discussion of ESA therapy’s risks and benefits.

Infographic by Priti Meena.
In conclusion, several studies have demonstrated adverse events associated with high ESA doses or high Hb targets. Nevertheless, we cannot overlook the role of ESAs in the treatment of anemia in kidney disease. These drugs have withstood the test of time and continue to be a mainstay of treatment of anemia in kidney disease.
COMMENTARY BY VOLKER H. HAASE:
HIF-PHIs – The new kids on the block in anemia therapy and much more
HIF Stabilizers

Copyright: TP71 / Shutterstock
The discovery of the Hypoxia-Inducible Factor (HIF) pathway represents a seminal moment in the field of medicine, culminating in a Nobel Prize in Physiology or Medicine in 2019. This pathway has been harnessed for the development of HIF stabilizers, new drugs that may become major players in the treatment of anemia of CKD. Roxadustat, vadadustat, daprodustat… Read this scouting report to learn about these drugs and why they are here to win NephMadness 2021.
The Nobel prize for Physiology/Medicine – 2019 was awarded to William G. Kaelin Jr, Peter J. Ratcliffe, and Gregg L. Semenza for their seminal work on the HIF pathway. The Academy stated that, “Through the combined work of these three laureates it was thus demonstrated that the response by gene expression to changes in oxygen is directly coupled to oxygen levels in the animal cell, allowing immediate cellular responses to occur to oxygenation through the action of the HIF transcription factor.”
In this section, we take you through a distilled discussion of the significance of HIF stabilizers, also known as hypoxia-inducible factor prolyl hydroxylase inhibitors (HIF-PHIs), and why they are contenders in this competition.
HIF is present in nearly all tissues, and is a heterodimer consisting of an α and β subunit. While HIF-β is constitutively expressed, HIF-α is regulated by oxygen tension. This occurs via HIF-prolyl hydroxylases which rapidly degrade the HIF-α subunit utilizing a proteasome complex in normal oxygen conditions, thereby impeding the heterodimerization with the HIF-β subunit and its transcriptional effects on ≥ 4000 genes. Gene activation results in enhanced red blood cell synthesis mediated via enhanced production of endogenous EPO and its receptor, and increased synthesis of various proteins associated with iron handling that includes transferrin and its receptor, duodenal cytochrome-B, divalent metal transporter-1, and ceruloplasmin (Figure 4). HIF-PHIs are small-molecule oral formulations that result in the enhancement of HIF-regulated gene expression (Figure 5). The overall effect is enhanced erythropoiesis stimulation beyond what can be accomplished by ESA use alone. Additionally, HIF-PHIs stimulate increased endogenous EPO production which is sustained and dose-dependent. The efficiency of iron utilization by reducing ferritin and hepcidin levels is also observed in patients with CKD and kidney failure. Importantly, HIF-PHIs also activate a variety of genes that do not affect erythropoiesis including those responsible for cellular proliferation, extracellular matrix production, glucose metabolism, and angiogenesis.

Hypoxia-inducible factor (HIF) pathway. Abbreviations: DcytB, duodenal cytochrome B; DMT1, divalent metal transporter 1; EPO, erythropoietin; PH, prolyl hydroxylase. Figure 1 from Gupta et al, AJKD © National Kidney Foundation.

HIF-prolyl hydroxylase inhibitors activate HIF signaling. Figure 1 from Sanghani et al, ACKD © National Kidney Foundation.
Based on this physiology, HIF-PHIs were developed for the treatment of anemia in patients with CKD and kidney failure. Currently, 3 HIF-PHIs are under development in the United States: roxadustat, vadadustat, and daprodustat. However, none have been approved for use in the United States as of January 2021.
The FDA accepted the new drug application of roxadustat in February 2020, but as of January 2021 has extended the review period by 3 months for additional clinical data analysis. Roxadustat has a half-life of 12–13 hours and is dosed orally three times weekly. In patients on dialysis randomized to roxadustat vs. EPO and followed for 26 weeks, roxadustat was non-inferior to EPO, demonstrating comparable effectiveness in raising Hb levels (roxadustat 0.7±1.1 g/dL vs epoetin alfa 0.5±1.0 g/dL). As compared with EPO, roxadustat resulted in a more significant increase in transferrin levels, decrease in transferrin saturation, maintenance of iron levels, and a reduction in hepcidin levels. Another striking difference is that roxadustat increased Hb levels to a similar degree in patients with high vs low C-reactive protein levels, whereas patients with high C-reactive protein levels who received EPO had lower Hb levels as compared to those with normal C-reactive protein levels. C-reactive protein levels are a surrogate for the inflammatory conditions that typically lead to “ESA resistance.” This alludes to the fact that perhaps HIF-PHIs can be useful in raising Hb levels independent of the baseline inflammatory milieu commonly seen in kidney failure.
Roxadustat increases Hb in patients with CKD not on dialysis as well. Patients with CKD randomized to roxadustat had a 1.9±1.2 g/dL increase in Hb levels while patients randomized to placebo had a 0.4±0.8 g/dL decrease in hemoglobin levels (P < 0.001). Hyperkalemia and upper respiratory tract infections occurred more frequently in those receiving roxadustat. Roxadustat was again compared to placebo in a recent multicenter study conducted in 2781 patients with CKD not on dialysis. Patients randomized to Roxadustat experienced increased mean hemoglobin levels of 1.75 g/dL while those randomized to placebo experienced a 0.4 g/dL increase in hemoglobin levels (P < 0.0001). The increase in hemoglobin seen in the Roxadustat group was significantly higher than that seen in the placebo group even in patients who had elevated C-reactive protein levels, and transfusion requirements were down by 63% in the Roxadustat group. Side-effect profile was comparable between both groups.

Visual abstract by Dhwanil Patel on Chen et al.
Daprodustat has a half-life of around 4 hours and is administered daily. In a randomized, double-blind, active-control, phase 3 study in Japanese patients with kidney failure receiving hemodialysis, oral daprodustat was non-inferior to darbepoetin alfa. Daprodustat-treated patients had lower hepcidin levels and improved iron binding capacity as compared with darbepoetin-treated patients. Further, daprodustat maintained target hemoglobin levels even in patients with ESA hyporesponsiveness.
An essential practical benefit of HIF-PHIs over ESAs is their oral administration, which circumvents the discomfort and pain from injections. When compared to ESA therapy, hemoglobin targets were attained with reduced plasma EPO levels. Given the elevated cardiovascular mortality and morbidity burden in ESA-treated patients which has been associated with supraphysiological EPO regimens and plasma EPO levels, HIF-PHI treatment has the ability to improve cardiovascular outcomes in CKD. Nonetheless, safety data to support this are not currently available. Another significant advantage is the suppression of hepatic hepcidin production and its adverse effects on iron mobilization, with the potential to decrease the need for iron therapy in patients with advanced CKD. These agents can be used to treat anemia independent of baseline inflammation and could be a viable alternative in ESA hyporesponsive patients. Roxadustat and daprodustat have been shown to lower total cholesterol and triglyceride levels as well. HIF-PHIs are also less expensive to manufacture compared to ESAs, as the manufacturing technique pertains to the application of synthetic chemistry as opposed to recombinant DNA technology, and their transport requires less challenging logistics. HIF-PHIs are also more cost-effective than placebo in patients with CKD not on dialysis. Since these agents lack a protein structure, this removes concerns regarding immunogenicity, a problem encountered with ESA use.
Nevertheless, since HIF pathways regulate or interact with many biologic processes, there are long-term safety concerns related to oncogenic risk in theory, effects on glucose metabolism, metabolic alterations, effects on cartilage and bone growth, and potential detrimental effects on CKD and progression of kidney cyst expansion. Hyperkalemia and metabolic acidosis have also been reported with roxadustat use. Certain cardiovascular risks such as predilection towards pulmonary arterial hypertension, thromboembolic disease promotion, and increased rates of angiogenesis are a big concern and have recently been a concern in the PRO2TECT study.
The Global Phase 3 Clinical Trials of Vadadustat vs Darbepoetin Alfa for Treatment of Anemia in Patients with Non-Dialysis-Dependent Chronic Kidney Disease (PRO2TECT) results were announced in September 2020 and presented at the late-breaking session of Kidney Week 2020. PRO2TECT included 2 randomized, global, multicenter, non-inferiority trials (Correction and Conversion) comparing vadadustat to darbepoetin alfa in patients with CKD not on dialysis for 52 weeks. Vadadustat achieved the efficacy endpoints demonstrating non-inferiority to darbepoetin alfa as measured by a mean change in Hb between baseline and 52 weeks. However, vadadustat was inferior to darbepoetin alfa in time to first occurrence of major adverse cardiovascular events (MACE), which was defined as the composite of all-cause mortality, non-fatal myocardial infarction, and non-fatal stroke. This is a major finding.
We are awaiting the publication of HIF-PHI clinical trials in order to fully understand their risks and benefits in the CKD population. Nevertheless, despite being a new therapy, HIF-PHIs are far from being the underdog of this matchup. Following 33 years of stagnation in the field of anemia in kidney disease, it is time for promising new therapies that could revolutionize the field. Impact on the field aside, will HIF-PHIs make their mark against ESAs on the court?
Check out this podcast episode of The Curbsiders featuring Joel Topf, Matt Sparks, and Pascale Khairallah:
#263 Iron Deficiency and Anemia of CKD
– Executive Team Members for this region: Pascale Khairallah @Khairallah_P, AJKD Editorial Board Member, and Matthew Sparks @Nephro_Sparks, AJKD Social Media Advisory Board member.
How to Claim CME and MOC
US-based physicians can earn 1.0 CME credit and 1.0 MOC point for reading this region.
- Register/log in to the NKF’s Professional Education Resource Center (PERC). If you select “Physician” in the drop-down menu during registration, the ABIM ID will pop up – make sure to complete this during registration to receive MOC points after course completion.
- Review the activity, disclosure, and accreditation information.
- Click “Continue” and review Course Instructions.
- Complete Post-Test. Please note: By selecting “Yes” to the participation questions for each region, the corresponding Post-Test questions will appear. Click “Save Draft” to save your responses and finish later. When you are ready to submit your answers, click “Preview” to review all responses, then click “Submit.”
- Click “Next” to complete the Evaluation form, then click“Submit.”
- Claim 1.0 CME credit and 1.0 MOC point per region (up to 8.0 total for 8 regions of NephMadness).
- Save/print your certificate.
The CME and MOC activity will expire on June 14th, 2021.



Leave a Reply