Peritoneal Dialysis in Patients With Ascites: Is it an Option?
Many of my personal patient encounters over the past years are near mirror images of the vignette discussed in a recent AJKD In Practice article from Rajora et al. A patient with known Hepatitis C cirrhosis, ascites, and CKD3 presents with spontaneous bacterial peritonitis and a decreasing GFR due to either CKD progression or hepatorenal syndrome. As the patient is waiting for liver (or potentially liver-kidney) transplant, hemodialysis is initiated, but ultrafiltration is difficult due to hypotension. As this patient still requires large volume paracenteses, can we kill two birds with one stone by considering peritoneal dialysis (PD)?
Patients with liver disease, portal hypertension, and ascites have several challenges that make traditional hemodialysis a challenge. In addition to the hemodynamics that make volume management difficult, we are often also dealing with coagulopathy, malnutrition, and encephalopathy. PD can offer both hemodynamic stability and control of ascites volume, but brings its own set of potential problems. Infection risk, albumin loss and subsequent malnutrition, and poor clearance are commonly cited as reasons PD is contraindicated in these patients, but are these theoretical or actually tied to poor outcomes?

Box 1 from Rajora et al, AJKD © National Kidney Foundation
It’s worth noting that of all patients initiated on dialysis with ascites, only 1% of them are initiated on PD. The primary drivers for this low rate are concerns about nutrition and infection. Many patients with advanced cirrhosis, ascites, and kidney failure are already malnourished and hypoalbuminemic due to decreased synthesis, inadequate dietary intake, and uremia. The obvious concern regarding peritoneal dialysis is that PD effluent will simply lead to more protein loss. Small cohort studies have yielded slightly conflicting results; initial PD effluent may be higher in protein but this decrease gradually. Additionally, the increase in intra-abdominal pressure generated by PD fluid may counter portal pressure and reduce the formation of ascites. Despite the theoretical risk, overall there were no significant changes in albumin from baseline values in these small patient groups.
How about infections? Patients with cirrhosis are already at risk of infection due to bacterial translocation. Does placement of an indwelling catheter create another pathway for bugs to get into ascetic fluid? Again, we are dealing with several small cohort studies, but the vast majority show that placement of an indwelling PD catheter does not confer a higher peritonitis rate when compared to non-cirrhotic controls. As always, strict sterile technique is the most important factor in preventing peritonitis.
Despite this encouraging data that does not preclude PD with regards to infection or malnutrition, few studies have compared mortality between HD and PD among patients with kidney failure and ascites. No major differences in mortality have been observed in comparison studies.
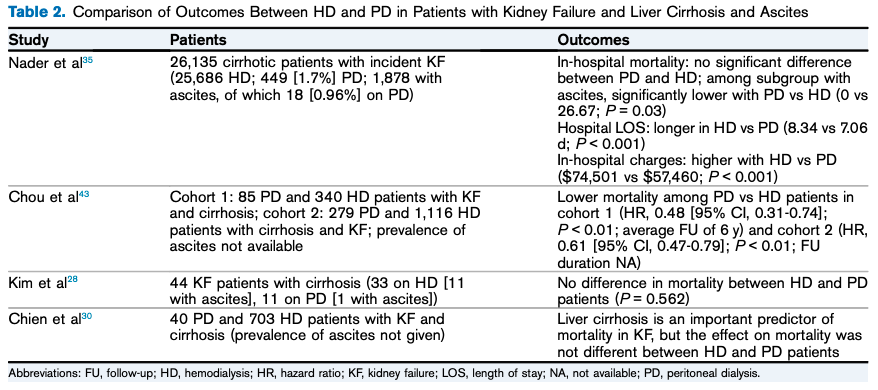
Table 2 from Rajora et al, AJKD © National Kidney Foundation
If the decision is made to start PD in a patient with ascites, there isn’t much else to guide you. Essentially no consensus exists regarding placement of the catheter, initial fill volumes, and the amount of ascites/effluent to drain. However, for a small minority of patients, PD may be a better dialysis modality. It provides hemodynamic stability and may facilitate better volume management compared to HD. The concerns of higher peritonitis rates and worsening malnutrition in PD have not been seen in small observational studies. And lastly, PD does not affect transplant candidacy and these patients can still receive liver, or simultaneous liver-kidney transplant. With all this in mind, perhaps PD is more viable in this population than most of us think.
– Post prepared by Timothy Yau @Maximal_Change, AJKD Social Media Editor
To view Rajora et al (subscription required), please visit AJKD.org.
Title: Peritoneal Dialysis Use in Patients With Ascites: A Review
Author: Nilum Rajora, Lucia De Gregorio, Ramesh Saxena
DOI: 10.1053/j.ajkd.2021.04.010

Leave a Reply