SGLT2 Inhibitors and Adverse Events: A Closer Look
Sodium-glucose cotransporter 2 inhibitors (SGLT2i) are an emerging class of medications that have become increasingly popular in medication regimens for diabetic patients with other comorbidities, such as heart failure. The recent DAPA-HF, EMPEROR and CANVAS trials showed a reduction in morbidity and mortality for patients with HFpEF, and HFrEF patients on dapagliflozin, empagliflozin, and canagliflozin. Since the main mechanism of action of SGLT2i is at the kidney tubules, it is important to investigate how modulation at this level can affect kidney status in patients on these drugs. The CREDENCE trial was designed to address this particular question of kidney related adverse events in patients with type 2 diabetes mellitus.
Before discussing the risk-benefit profile of these medications, it is important to understand what they do at the kidney level. As shown in the figure below, there are two main functions of SGLT2 on the kidney tubules: natriuresis and glucosuria. The SGLT2 receptor is responsible for resorption of one sodium ion for every glucose molecule in the early proximal tubule. In diabetes, kidney hypertrophy leads to increased SGLT2 expression and activity. Tubuloglomerular feedback from decreased sodium concentration at the macula densa leads to vasoconstriction of arterioles. This results in a mismatch between kidney tubular oxygen requirements and delivery. SGLT2i can help prevent hypoxia related kidney injury by blocking SGLT2 at the proximal tubules. The kidney protective effects of these medications have been outlined in the NephMadness 2020 region dedicated to these agents.

Figure 1 from Tuttle et al, AJKD © National Kidney Foundation and American Diabetes Association
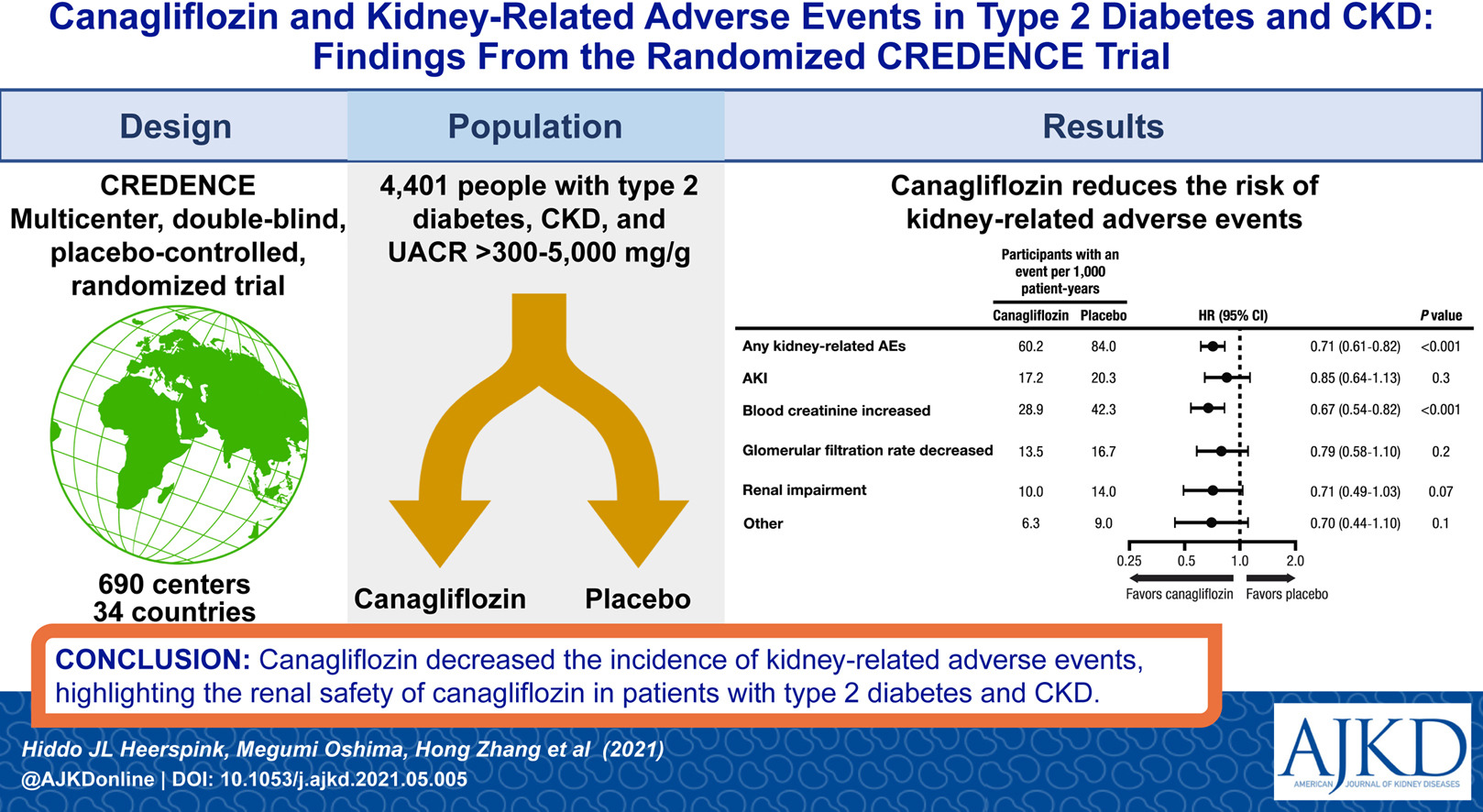
In a recent AJKD article, Heerspink et al analyze the CREDENCE trial with a specific look at kidney-related adverse events. The original CREDENCE study was a randomized, double bind, placebo controlled, multicenter trial with 4401 participants who had type 2 diabetes, chronic kidney disease (CKD), and a protein-creatinine ratio of 300-500 mg/g. Patients were assigned to daily treatment with canagliflozin 100 mg or matching placebo. The treatment was continued until meeting one of the pre-defined endpoints (dialysis, transplant, diabetic keoacidosis, pregnancy, starting unapproved therapy or completion of study). Data was obtained by individual investigators reporting kidney related adverse events (AE).
The results of this specific data analysis revealed the following:
- Kidney related serious AEs occurred in 2.8% of patients on canagliflozin compared to 3.7% placebo [HR 0.72 (95% CI 0.5-1)].
- Acute kidney injury (AKI) occurred in 3.9% on canagliflozin compared to 4.5% on placebo [HR 0.85 (95% CI 0.64-1.13)].
- For the initial months, the proportion of serious AE and AKI was similar, then began to favor canagliflozin for less adverse outcomes over time.
These findings are graphically represented in the plots below:

Figure 1 from Heerspink et al, AJKD © National Kidney Foundation
This study showed that patients with type 2 diabetes and CKD who were on canagliflozin had fewer adverse kidney side effects as compared to the placebo group. The graphs above reflect that in the first few months of the study, there was no significant difference between canagliflozin and placebo. It is interesting to note this, because there is often an initial decrease in estimate glomerular filtration rate (GFR) from tubuloglomerular feedback associated with medications such as SGLT2i and RAAS inhibitors. In rare instances where patients developed AKI, patients on canagliflozin demonstrated partial to full recovery. Since the majority of AKI occurred within the first four weeks, it may be that these dips in GFR are a reflection of the mechanism of action rather than a true adverse event affecting the kidneys.
The CREDENCE trial once again demonstrated a clear benefit of using an SGLT2i to reduce renal AEs in diabetic patients with CKD. With ongoing data now supporting benefit of SGLT2i in non-diabetic kidney disease, and with benefits extending to other organs such as the heart, the future looks bright for this class of medications.
From Heerspink et al, AJKD © National Kidney Foundation
– Post prepared by Baani Singh (AJKDBlog Guest Contributor) and Edgar V. Lerma @edgarvlermamd, AJKD Social Media Advisory Board member.
To view Heerspink et al please visit AJKD.org.
Title: Canagliflozin and Kidney-Related Adverse Events in Type 2 Diabetes and CKD: Findings From the Randomized CREDENCE Trial
Authors: Hiddo J.L. Heerspink, Megumi Oshima, Hong Zhang, Jingwei Li, Rajiv Agarwal, George Capuano, David M. Charytan, Jagriti Craig, Dick de Zeeuw, Gian Luca Di Tanna, Adeera Levin, Bruce Neal, Vlado Perkovic, David C. Wheeler, Yshai Yavin, Meg J. Jardine
DOI: 10.1053/j.ajkd.2021.05.005

Leave a Reply