#NephMadness 2022: Lupus Region
Submit your picks! | NephMadness 2022 | #NephMadness | #Lupus
Selection Committee Member: Keisha Gibson @Dr_KeishaGibson
Keisha Gibson is Chief of Pediatric Nephrology at the University of North Carolina at Chapel Hill and Vice Chair of Diversity and Inclusion for the Department of Medicine. Her research and clinical interests focus on glomerular diseases.
Writer: Jason Kidd @jkidd03
Jason Kidd is an Associate Professor of Medicine in the Division of Nephrology at Virginia Commonwealth University. He serves as the adult nephrology fellowship Program Director at VCU Health. His clinical and research interests are in glomerular kidney diseases.
Competitors for the Lupus Region
Belimumab vs Voclosporin
Family Planning in LN vs Pregnancy in LN

Copyright: CHAINFOTO24 / Shutterstock
Belimumab vs Voclosporin
Systemic Lupus Erythematosus (SLE) is a complex, multisystem autoimmune disease that can cause damage to any organ, including the kidneys. Lupus Nephritis (LN) is a serious, organ threatening manifestation of SLE and should be considered in any patient with SLE who develops hematuria, proteinuria, and/or kidney insufficiency. LN is difficult to treat and is associated with a significant decrease in survival in patients with SLE.
The gold standard for diagnosis of LN is a kidney biopsy. LN has a wide array of histopathological abnormalities, most commonly manifesting as an immune complex-mediated glomerulonephritis. Immune complex deposition leads to inflammation and activation of complement mediated pathways. Glomerular patterns of immune complex mediated injury are in many cases related to the site of accumulation of immunoglobulins in the mesangium, subendothelial space, and subepithelial space. Injury in these spaces can lead to kidney failure if left untreated. It is common for multiple patterns to co-exist. The International Society of Nephrology/Renal Pathology Society developed a classification system in order to standardize the description of clinically relevant lesions.
Table I ISN/RPS LN Classifications
|
Category |
Description |
Light Microscopy |
Capillary Loop |
|
Normal |
|
|
|
|
Class I |
Normal glomeruli on light microscopy, mesangial immune complexes on immunofluorescence and/or electron microscopy |
|
|
|
Class II |
Mesangial hypercellularity with mesangial immune deposits seen on electron microscopy |
|
|
|
Class III |
Focal segmental or global disease involving <50% of glomeruli. Active and chronic lesions may be seen. Crescents are possible. |
|
|
|
Class IV |
Diffuse segmental (S) or global (G) disease involving ≥50% of glomeruli. Crescents can be present. |
|
|
|
Class V |
Membranous pattern, subepithelial immune deposits with glomerular basement membrane thickening. Can occur in combination with class III or IV. |
|
|
|
Class VI |
Advanced glomerulosclerosis involving > 90% of glomeruli |
*Images courtesy of Dr. J. Charles Jennette
The variability of histopathologic manifestations leads to a diverse pattern of clinical presentations. Patients may present with subclinical disease, nephrotic syndrome with mostly preserved kidney function, or rapidly progressive glomerulonephritis. Without therapy, patients with active Class III and IV LN are at high risk of kidney loss.
Immunosuppressive therapies are used in an attempt to achieve remission. The standard of care for treatment of LN includes glucocorticoids, cyclophosphamide, and mycophenolate. Antimalarial drugs and renin-angiotensin-aldosterone system (RAAS) blockade should also be employed as standard of care in patients with lupus nephritis. Despite these intensive regimens, the likelihood of attaining complete remission in patients with lupus nephritis is low and 10%-30% of patients with LN develop end stage kidney disease (ESKD).
In highly resourced countries, 40% of patients with class IV LN and 20% of patients with class V LN will develop ESKD within 15 years of diagnosis. Black and Hispanic patients with SLE develop LN at a younger age and are more likely to develop ESKD and die than White patients with SLE. To further complicate matters, patients who appear to have good clinical responses to therapy may continue to have active disease on repeat kidney biopsy, and patients with histologic remission on biopsy may still have active disease. Histologic transformation is a frequent occurrence on repeat biopsy.
The goal of treatment in LN is to preserve kidney function and reduce proteinuria all while limiting medication-related side effects as well as complications related to chronic kidney disease (CKD). Simple, right?
Patients that reach a complete remission of LN are unlikely to progress to ESKD. So, the search for more effective treatment regimens in lupus nephritis is an urgent problem.
The addition of calcineurin inhibitors to mycophenolate mofetil (MMF) has shown promise in some populations with lupus nephritis. Biologic drugs are used across the continuum of glomerular kidney diseases and have been studied extensively in LN. In the last year, two industry funded studies have demonstrated efficacy of novel add-on therapies for the treatment of lupus nephritis. We give you, Belimumab versus Voclosporin.
Belimumab
B cells are sources of autoantibody production and contribute to inflammation. Biologic agents that affect B cells are commonly used for the treatment of a wide range of rheumatologic conditions.
Copyright: Kateryna Kon / Shutterstock
Rituximab, a chimeric monoclonal antibody that eliminates CD20-positive B cells, is the Duke Basketball of glomerular kidney disease. Omnipresent. A winner. (note: the author of this region is undeniably biased as a graduate of Duke University.) However, when it comes to management of LN, rituximab, like Duke in 2020, does not always come out on top.
In 2012, the LUNAR trial sought to evaluate the safety and efficacy of rituximab in patients with lupus nephritis. Patients were treated along with MMF and steroids. More patients receiving rituximab had a complete or partial response when compared to placebo, but this difference was not statistically significant. There was a signal for greater reduction of proteinuria in the group of patients receiving rituximab. However, more patients in that group also required cyclophosphamide as rescue therapy. As a result, use of rituximab for LN is considered an off-label use.
B cell dysfunction leads to the production of autoantibodies and contributes to an abnormal immune response in patients with SLE. B lymphocyte stimulator (BLyS), which promotes survival of B cells allowing them to differentiate towards autoantibody production, is elevated in patients with SLE, and correlates with disease activity. Belimumab is a monoclonal antibody that binds to soluble BLyS thereby preventing binding and stimulation of B cells. It is available in both intravenous and subcutaneous forms.
The BLISS trials studied the addition of belimumab to standard of care in patients with active SLE and led to reduced disease activity and flares. The BLISS trials were not designed to study patients with active LN, but a pooled post hoc analysis of patients in these trials with active kidney disease indicated that those who received belimumab were more likely to achieve a kidney remission. Patients with severe lupus nephritis were excluded from the initial BLISS trials including those with more than 6 grams of proteinuria or serum creatinine >2.5 mg/dL, and those who required hemodialysis or high-dose prednisone within 90 days of the study. Based on these results, further investigation was pursued.
BLISS-LN was an international phase III, randomized, double-blind, placebo-controlled study to evaluate the efficacy and safety of belimumab plus standard of care in patients with biopsy-proven active class III, IV, and/or V LN. The primary end point at week 104 was a primary efficacy kidney response, as defined by a urinary protein to creatinine ratio of ≤0.7 and an estimated eGFR no worse than 20% below the value before the flare, or >60 mL/min/1.73 m2. The secondary end point was a complete kidney response: a ratio of urinary protein to creatinine of <0.5, an eGFR no worse than 10% or >90 mL/min/1.73 m2. Time to kidney-related events or death was assessed.

Visual abstract by @whatsthegfr on Furie et al.
The trial randomized 448 patients. Cyclophosphamide followed by azathioprine was administered in 119 patients and MMF was given to 329 patients. About 90% of patients were women and 14% were Black. Almost 60% of patients had Class III or IV nephritis. Use of hydroxychloroquine was not reported in the study.
At the end of the trial, 43% of patients in the group receiving belimumab reached a primary efficacy kidney response compared to 32% of patients not receiving the drug. Fewer patients in the belimumab arm experienced treatment failure, and they had a lower risk of kidney related event or death.
The benefit of adding belimumab appeared more prominent in the group receiving MMF when compared to those receiving cyclophosphamide/azathioprine, though the study was not powered to show this difference.
Of note, the initial primary end point in the study was a complete, partial, or lack of response to therapy. The study end point was changed to a primary efficacy renal response (PERR) several years into the study, which has garnered controversy. The authors have noted that the decision to change the primary end point was made prior to the unblinding of data. Furthermore, this change was based on emerging evidence of short-term predictors of long-term outcomes in patients with lupus nephritis. Reduction in proteinuria in the first year of treatment is an important prognostic marker in LN and is being described across the literature in glomerular kidney diseases, including in patients with focal segmental glomerulosclerosis (FSGS).
When the results of the study were analyzed using the original end points of the trial, there was no significant difference between patients receiving placebo versus belimumab.
There are other limitations to this study. Patients were not randomized to receive different induction therapies and this may have contributed to some bias in the initial management of patients. There was also limited enrollment of Black patients (13.7%), so it is unclear if the results can be generalized to all patients with LN.
Another concerning aspect of this study was the relatively low number of patients on RAAS blockade; only 67% of patients in this trial were on ACEi or ARB therapy. Unfortunately, this is also seen across the continuum of clinical trials in glomerular kidney diseases.
No conclusions can be made about the use of belimumab in the treatment of patients with severely reduced kidney function resulting from active lupus nephritis, as patients with an eGFR of <30 mL/min/1.73 m2 BSA were excluded from BLISS LN.
Despite the encouraging results in PERR in patients receiving belimumab along with standard of care, questions remain regarding the optimal therapy for patients with LN. One of the major difficulties in studying as well as treating patients with LN is the heterogeneity of disease presentations. Patients across histologic classes may respond to treatment differently. A post hoc analysis of the study suggested that belimumab was less likely to be efficacious in patients with Class V LN. Patients receiving belimumab were also less likely to have a flare of their LN, suggesting a potential role in maintenance of remission.
There is interest in investigating the efficacy of belimumab when combined with other biologics, including old faithful, rituximab. A recent study looked at the combination of rituximab therapy with belimumab in patients with SLE. The authors hypothesized that high B cell activating factor activity after rituximab infusion leads to more flares of SLE. Patients who were resistant to conventional treatment were treated with rituximab infusions and 4 to 8 weeks later were started on belimumab or placebo for 52 weeks. The primary end point of the study was anti-dsDNA levels at 52 weeks and patients receiving belimumab achieved lower levels of this antibody, but this study was not powered to detect differences in outcomes. A phase II trial, CALIBRATE, studied the use of cyclophosphamide plus rituximab followed by belimumab in patients with refractory LN, and this combination was found to be safe and effective in depleting B cells. Eleven of 21 patients receiving belimumab reached a partial or complete renal response compared to 9 of 22 patients who were not treated with the drug.
Belimumab has also been studied in children with SLE. The PLUTO trial was a phase II trial looking at safety and efficacy of intravenous belimumab in a pediatric population (ages 5-17 years). Children who received belimumab plus standard of care were more likely to reach the study end point as measured by SLE responder index score. Children who received belimumab were more likely to show improvement in kidney indices and less likely to develop new kidney involvement, although these outcomes did not reach significance.
The trial was funded by GlaxoSmithKline, the makers of belimumab. Based on the results of BLISS-LN, belimumab is now FDA approved for the treatment of LN in combination with standard therapy (IV and subcutaneous belimumab in adults and IV only for children under 18 years of age).
Belimumab costs approximately $35,000 per year.
Voclosporin
Calcineurin inhibitors have been combined with MMF in several trials of LN and this is often referred to as multitarget therapy. In 2 trials, the combination of MMF and tacrolimus along with glucocorticoids was compared to patients receiving IV cyclophosphamide.
In 2008, Bao et al prospectively studied 40 patients with class IV and V lupus nephritis and compared treatment with multitarget therapy (MMF, tacrolimus and steroids) to treatment with intravenous cyclophosphamide. More patients who received multitarget therapy achieved complete remission at 6 and 9 months.
A much larger study published 7 years later showed similar results at 24 weeks. Calcineurin inhibitors are thought to decrease proteinuria in lupus nephritis due to both their immunosuppressive effects and their direct effect on podocytes. Calcineurin inhibitors have a direct effect on the actin cytoskeleton of podocytes, increase the expression of proteins associated with the slit diaphragm, and inhibit podocyte apoptosis.

Copyright: Dayonara / Shutterstock
Tacrolimus and cyclosporine have well known side effects. Patients receiving these medications are at risk of developing diabetes mellitus, hypertension, gout, and hypomagnesemia, and have an increased susceptibility to infections. Frequent monitoring is necessary to ensure therapeutic levels.
Voclosporin is a calcineurin inhibitor that is structurally similar to cyclosporine with stronger calcineurin binding. Like tacrolimus and cyclosporine, Voclosporin blocks T cell-mediated immune responses and has a direct stabilizing effect on the podocyte. Voclosporin is more potent and drug monitoring is not required. Steady state concentrations are reached after 6 days of twice daily dosing.
Voclosporin was initially developed for use in patients with kidney transplants. In a study comparing voclosporin to tacrolimus in patients with kidney transplants, voclosoporin did not lead to more rejection and caused less new onset diabetes when compared to patients receiving tacrolimus. Cyclosporine may decrease levels of MMF in the blood, but this effect is not seen in voclosporin.
New onset diabetes after kidney transplant (NODAT) is commonly seen in patients treated with tacrolimus and may be related to direct inhibition of insulin secretion from islet cells. Patients treated with voclosporin are potentially less likely to develop NODAT. On the heels of promising data from the multitarget studies showing decreased adverse effects related to calcineurin inhibition and no need for therapeutic dose monitoring, focus was turned to the use of voclosporin in patients with LN.
AURA LV was a phase II trial comparing 2 separate doses of voclosporin plus standard of care to placebo and standard care in patients with active class III, IV, or V LN. The primary outcome of the study was a complete kidney remission (CRR) at 24 weeks. Of the patients receiving a higher dose of voclosporin, 27.3% reached CRR, while patients receiving the lower dose of 23.7 mg twice daily of voclosporin reached CRR in 32.6% of cases, a significant improvement when compared to those receiving placebo.
AURORA 1 was a multicenter, double-blind, randomized phase III trial comparing treatment of patients with active LN with voclosporin 23.7 mg twice a day or placebo in combination with MMF 1000 mg twice a day and rapidly tapered prednisone for 52 weeks. Patients with an estimated eGFR of <45 mL/min/1.73 m2 BSA were excluded from the study.

Visual abstract by @priti899 on Rovin et al.
The primary end point of the study was a complete kidney response at 1 year. A complete kidney response was defined as a combination of urine protein creatinine ratio of less than or equal to 0.5 mg/mg, stable or no decrease in kidney function, no need for rescue therapy, and no need for increase in prednisone dose.
The primary end point of complete renal response at week 52 was achieved in significantly more patients in the voclosporin group (73 of 179 patients, or 41%) than in the placebo group (40 of 178 patients, or 23%). Patients receiving voclosporin also reached a 50% reduction in UPCR and <0.5 g proteinuria more rapidly. The significant reductions were maintained for 52 weeks. Adverse events were similar in the groups.
As expected in trials involving calcineurin inhibitors, a reduction in eGFR was seen in 26% of patients receiving voclosporin, but there was no overall difference in eGFR change between the voclosporin and placebo arms. The reduction in eGFR occurred soon after voclosporin was initiated and was mostly stable over time. Severe adverse events were uncommon and not significantly different between groups, although hypertension was a more frequent complication in the group receiving voclosporin.
The study population was diverse and its effects can likely be generalized to the diverse populations we see with LN. No significant changes were noted in lupus activity biomarkers in the group receiving voclosporin. However, this has not been shown to be a reliable indicator of disease activity in patients with LN.
Most patients had kidney biopsies within 6 months of screening. However, the study did not differentiate response between patients with new onset versus a relapse of LN. Non-kidney manifestations of SLE were not studied.
The trial was funded by Aurinia Pharmaceuticals, the maker of Voclosporin. This drug has not been studied in children with LN. The cost of the drug is $92,000 per year. Voclosporin has not been studied in a head-to-head fashion against tacrolimus. Although voclosporin may be less likely to cause nephrotoxicity and new onset diabetes mellitus, and may obviate the need for drug monitoring, it is unclear whether the astounding sticker price would lead to a more significant change in outcome than the use of other calcineurin inhibitors.
Based on the results of AURORA 1, voclosporin is FDA-approved for the treatment of lupus nephritis in combination with MMF and glucocorticoids.
Family Planning in LN vs Pregnancy in LN
SLE predominantly affects women of child-bearing age. Treatments for SLE can lead to infertility and can have an adverse effect on fetal development. The presence of active LN during pregnancy increases the risk of developing pre-eclampsia, preterm birth, intrauterine growth restriction, and fetal loss.
Treating LN is hard enough. However, it is imperative for nephrologists to play a role in educating our patients on the potential complications they may face related both to their disease and to the treatments we prescribe. Nephrologists may feel unprepared to offer counsel in this area—I know I do—but I would advocate that we play a pivotal role in supporting our patients as they embark on building their families in whatever way they desire. Family planning should be discussed from the first meeting with the patient and should be continually reinforced throughout their care.
COMMENTARY BY SWAPNIL HIREMATH:
Lupus Nephritis – Progress, Far from Perfection
Family Planning in LN
Careful planning is key to making pregnancy as successful as possible in women with LN, and it is important to start thinking about the implications of treatments for pregnancy at the outset of caring for patients with LN. When a patient is started on treatment for LN, nephrologists should make it a priority to discuss the need for contraception, the teratogenic effects of their medications, the importance of quiescent disease prior to becoming pregnant, and the potential implications for fertility from the treatments they receive. Nephrologists should make a habit of addressing this regularly with their patients.
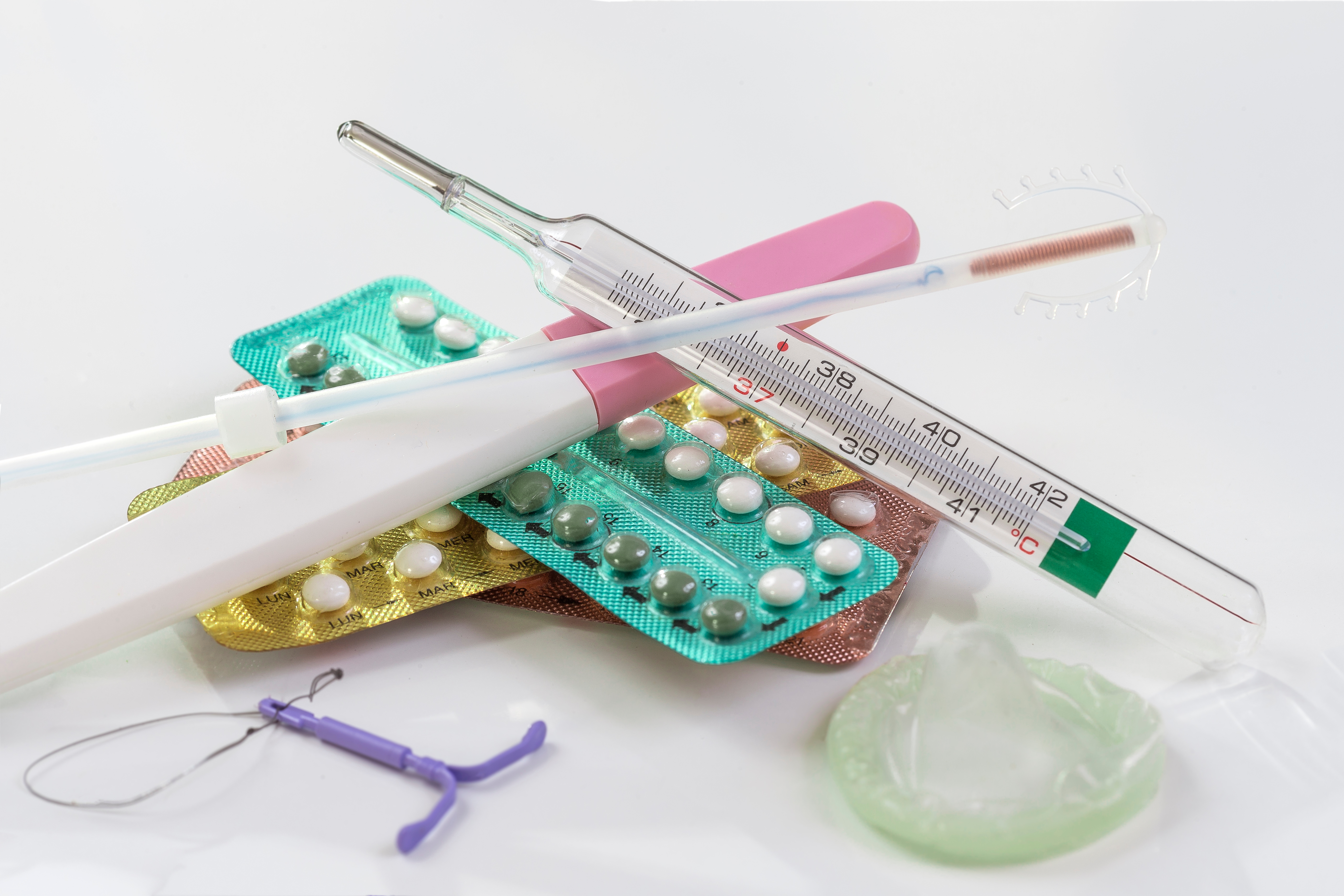
Copyright: JPC-Prod / Shutterstock
Women benefit from a multidisciplinary approach prior to conception along with their rheumatologist, nephrologist, and a maternal-fetal medicine specialist. Due to their integral role in the care of patients with LN, the nephrologist plays a key role in this approach.
Pregnancy risks in SLE are associated with disease activity at the time of conception. Women should be counseled on the importance of contraception. Many women with SLE are at risk of unintended pregnancy and they want to discuss contraception with their providers. Women with LN should be counseled on the importance of quiescent disease prior to planning to become pregnant. Active LN increases the risk of fetal loss during pregnancy and of maternal complications. These risks decrease if disease activity in SLE is low at the time of pregnancy. Expert guidance suggests that pregnancy should be delayed until patients have quiescent disease for at least 6 months with minimal proteinuria. The American College of Rheumatology strongly recommends that patients with connective tissue diseases are in a quiescent or low activity state prior to becoming pregnant, and advocate for maintaining concurrent care with specialists in obstetrics-gynecology, maternal fetal medicine, and other specialists as appropriate.
Contraceptive choice in women with SLE is complicated due to the potential risk of flare of their disease from hormones, their increased risk of thrombosis, increased risk of bleeding from thrombocytopenia, and potential need for treatment with glucocorticoids. Physicians who care for women with SLE would benefit from having knowledge of contraceptive options for their patients. The table below describes the different contraceptive methods available as well as the risks and benefits of each type in both patients with SLE and those with antiphospholipid antibodies (APS).
| Contraceptive Method | Failure Rate | SLE | APS* |
| Levongestrol IUD | 0.1-0.4% | Ease of use, no increased risk of SLE flare. May be helpful in women with menorrhagia from thrombocytopenia. | Low risk of thrombosis |
| Copper IUD | 0.8% | Effective, may increase risk of bleeding/dysmenorrhea | Low risk of thrombosis |
| DMPA* | 4% | Injections every 3 months. Could worsen bone density if also treated with steroids. Return to fertility may be delayed. | Uncertain risk of thrombi |
| Progestin implant | 0.1% | Inserted by MD every 3 years. Rapid return to fertility | Risk of thrombi not known |
| Estrogen/Progesterone combination | Pill 7%
Patch 7% Ring 7% |
Multiple options to fit a patient’s preferences (pill, patch, vaginal ring)
No increased risk of flare with stable disease Increased risk of thrombotic events. Avoid in patients with prior thrombosis. Drug-drug interactions are common Avoid in active disease/flare |
Avoid due to thrombotic risk |
| Progestin only pill | 7% | No increased risk of flare. | Low risk of thrombosis |
| Emergency contraception | Can be used | No disease specific risk | |
| Barrier methods | Varies e.g.
Diaphragm 17% Male condom 13% |
Can be used
Male condoms only option aside from abstinence that would also decrease risk of sexually transmitted illnesses |
No disease specific risk |
| Sterilization (tubal ligation or vasectomy) | Could be performed in partner. Highly effective. Considered permanent. | Can be used | No disease specific risk |
*APS: antiphospholipid syndrome
*DMPA: depot medroxyprogesterone acetate
**Adapted from Sammaritano LR; Lupus 2014
Women with APS are at high risk for thrombotic events and they should not receive estrogen containing contraception.
To decrease the risk of sexually transmitted diseases, women should also be encouraged to use barrier protection, though this is less effective at contraception prevention than other methods and should be used in conjunction with other methods if possible.
In addition to contraception, women should be counseled on the potential teratogenicity of medications they may be taking including MMF, warfarin, and ACEi/ARBs. Unintended pregnancies do occur. If women become pregnant while on these medications, the drugs should be stopped immediately and they should alert their nephrologist.
The use of MMF during pregnancy increases the risk of miscarriage and birth defects. Congenital abnormalities of the ear and palate may occur. Anomalies of the kidneys, heart, and distal limbs may also result from use in pregnancy. All providers can undergo training on the embryo-fetal consequences of the drug.
RAAS inhibitors can cause severe teratogenic effects. Angiotensin converting enzyme inhibitors (ACEi) have been associated with oligohydramnios, kidney tubular dysplasia, and hypocalvaria, and carry a high mortality rate. First trimester exposure does increase risk to the fetus, and ACEi and ARBs should be discontinued prior to conception. Women who did take these medications while pregnant should undergo a detailed fetal ultrasound.
Women on warfarin for management of APS should be transitioned to low molecular weight heparin. The use of anti-Xa inhibitors is not recommended during pregnancy.
When LN is in a stable remission, the patient can be transitioned to medications that are safe in pregnancy. Cyclophosphamide and MMF are contraindicated. If continued immunosuppression is indicated, azathioprine can be used. Hydroxychloroquine should be continued during pregnancy and data from the Johns Hopkins Lupus cohort show that withdrawal increases the risk of a kidney flare. Rituximab should be avoided for about 1 year prior to conception, as it can cross the placenta and may lead to fetal B cell depletion.

Visual abstract by @priti899 on Clowse et al.
Women with APS are at high risk for blood clots. Women with prior history of thrombosis should be anticoagulated during pregnancy. Anticoagulation can also be considered in women without a history of clot. All women with APS should receive aspirin. Presence of antiphospholipid antibodies also portend to a higher risk of fetal loss. Careful history regarding prior miscarraige in patients with SLE can inform further evaluation of APS.
But what about the implications of LN on fertility?
SLE itself does not generally lead to decreased fertility. However, patients with CKD are less likely to become pregnant. At diagnosis, patients should be counseled on the potential implications of their disease as well as their treatments on fertility.
Cyclophosphamide, an alkylating agent, may lead to premature ovarian failure in women. The cumulative dose of cyclophosphamide as well as advancing maternal age increase the risk of infertility. This is difficult to study in a controlled fashion and as a result, much of the data available are retrospective. A meta-analysis of 1,388 women receiving cyclophosphamide demonstrated that almost 20% developed sustained amenorrhea. Ovarian failure was correlated with increasing age at time of dosing and cumulative dose of cyclophosphamide >5 grams.
Fertility preservation with GnRH agonists should be considered in women treated with cyclophosphamide who wish to preserve their fertility. Nephrologists may refer these patients to fertility specialists prior to initiating cyclophosphamide. However, in some cases, this therapy cannot be delayed. Leuprolide is a gonadotropin-releasing hormone (GnRH) agonist used for the management of endometriosis and treatment of precocious puberty. Initially, the drug stimulates luteinizing hormone (LH) and follicle-stimulating hormones (FSH). As the drug is continuously given, leuprolide inhibits LH and FSH and leads to decreased levels of testosterone and estrogen.
In a small study, women with severe SLE treated with intravenous cyclophosphamide and leuprolide were less likely to develop premature ovarian failure than matched controls. Women in this study received an average of 12.9 grams of cyclophosphamide. GnRH-a is most effective if administered about 3 weeks prior to receiving cyclophosphamide. While this is difficult to plan due to the nature of the presentation of LN, delay of treatment of disease can have severe long term consequences.
SLE is much less common in men. This imbalance increases in the childbearing years and has been attributed to a hormonal effect. Men are not commonly enrolled in trials of LN. In the BLISS-LN trial, 13.5% of patients were men. In Aurora 1, 12.3% were men. A study from Taiwan demonstrated worse kidney outcomes in men with SLE. A multiethnic cohort of patients with SLE in the United States has described a higher rate of kidney disease in men with SLE and a poorer long term prognosis, likely related to accelerated damage earlier in their disease course.
How should we counsel men on the risks of LN and treatments and the effect on their fertility?
Similar to female patients, both the underlying disease and its treatment may affect fertility. Disease activity in SLE may have a significant effect on testes, and cases of vasculitis have been described. CKD is a common cause of erectile dysfunction as well as a decrease in spermatogenesis. Klinefelter Syndrome, a genetic condition which predisposes men to infertility is more common in men with SLE than the general population.
Cyclophosphamide is gonadotoxic and impairs spermatogenesis. It has been associated with reduced levels of testosterone. As in women, infertility is linked both to cumulative dose of cyclophosphamide and the age of dosing. Sperm quantity and quality have been shown to be significantly decreased in boys treated with cyclophosphamide after puberty. When compared to controls, 35 males treated for lupus had lower teste volume, lower sperm count, and decreased sperm motility. Glucocorticoid use has been associated with decreased testosterone in patients with chronic obstructive pulmonary disease, so they may also contribute to decreased androgen levels in men with SLE.
Women and men should be counseled on cryopreservation options available to them, and referral to reproductive endocrinologists is beneficial. Unfortunately, due to the rapidity and severity of disease onset in some patients, urgent initiation of therapy is needed. Also, these therapies are often cost-prohibitive. Assisted reproduction techniques including in vitro fertilization are generally safe in women with SLE.
COMMENTARY BY SILVI SHAH:
Family Planning with Lupus Nephritis
Pregnancy in LN
Pregnant women with SLE are at higher risk for the development of maternal and fetal complications. Pregnancy is associated with a higher risk of SLE flare. Pregnant women with SLE are also at higher risk for the development of diabetes mellitus, infections, thrombosis, thrombocytopenia, and need for transfusion.

Copyright: Paul Velgos / Shutterstock
During pregnancy, women with a history of LN should be closely monitored by their multi-specialty team. Regular monitoring of kidney function, urinalysis, 24-hour urine collections, and blood pressure monitoring should be performed. Hypertension during pregnancy leads to an increased risk of preterm birth. However, aggressive management of high blood pressure could lead to a reduction in placental blood flow and complications. The optimal blood pressure in pregnant women with a history of LN is unknown, and will need to be individualized for each patient. Based upon the ACOG guidelines, treatment should be initiated to maintain a systolic blood pressure below 160 mm Hg and diastolic blood pressure of 110 mm Hg.
In a prospective study of 71 pregnancies with mostly quiescent LN, flares of SLE occurred in almost 20% of patients, 8.4% of patients developed pre-eclampsia, and 2.8% developed HELLP syndrome. Low C3 and high anti-DNA antibodies were predictive of kidney flares and obesity was associated with worse outcomes. Preterm birth occurred in 30.8% of patients, a much higher rate than the general population. Although these statistics may feel daunting, the vast majority of women who become pregnant with quiescent LN or stable extra-kidney SLE have unremarkable pregnancies. Prompt recognition and intervention can stabilize patients and protect the fetus.

Visual abstract by @krithicism on Moroni et al.
Preeclampsia (Pre-E) is a common complication of pregnancy and is characterized by new onset hypertension and proteinuria after 20 weeks of gestation. A history of LN increases the risk of developing Pre-E, and this risk is further increased if active LN is present. Severe Pre-E may lead to liver injury, seizures, thrombocytopenia, microangiopathic hemolytic anemia, and acute kidney injury. Pre-E may lead to intrauterine growth restriction and often necessitates early delivery.
Differentiating between LN and Pre-E can be challenging. Patients with LN generally have an active urine sediment with hematuria, proteinuria, and cellular casts. In Pre-E, the predominant urinary finding is proteinuria and a benign urinary sediment. However, in severe Pre-E the presence of an active sediment has been reported. Checking complement levels and anti-dsDNA may be helpful in differentiating the diseases. It should be noted that C3 and C4 levels gradually increase during pregnancy, so this should be taken into account when evaluating complement levels. Timing during pregnancy can also be helpful in differentiation (as Pre-E is unlikely to develop prior to 20 weeks of gestation) as can measurement of the sFlt-1/PlGF ratio.
In a pregnant patient with a known history of LN, it can be clinically difficult to determine if the development of hypertension, kidney insufficiency, and an active urine sediment represents an LN flare or Pre-E. Rarely, a woman’s first presentation of LN may occur during pregnancy. If a woman develops hypertension, an active urine sediment, nephrotic syndrome, or rapid loss of GFR prior to 20 weeks, LN is a distinct possibility. In these cases, definitive diagnosis may be important as the initiation of immunosuppressive medications may prolong pregnancy and allow for a successful pregnancy to occur.
In some cases, it may be necessary to perform kidney biopsy during pregnancy, and this can be technically challenging. If needed, an experienced operator should perform biopsy, and ultrasound-guided percutaneous biopsy could be performed in women sitting upright or in the lateral recumbent position later in pregnancy. Bleeding complications are more common in pregnant patients undergoing biopsy, but most of these complications have been noted to occur later in pregnancy (between 23 and 26 weeks). Kidney biopsies during pregnancy do lead to a change in treatment plan in the majority of patients. The balance between the risks and benefits of pre-emptive treatment and a kidney biopsy should be weighed with the patient and the multidisciplinary team.
Patients with active LN during pregnancy benefit from therapy. However, some medications commonly used in LN can be harmful to the fetus. Specifically, cyclophosphamide and MMF are associated with the development of congenital malformations. Both azathioprine and tacrolimus can be used in pregnant women. Women should be counseled to continue hydroxychloroquine during pregnancy. If a woman has a severe flare of LN during pregnancy, high-dose steroids, intravenous immunoglobulin (IVIG), and plasmapheresis have been utilized.
Of course, active LN during pregnancy can cause complications not only for the mother, but the developing child as well. Patients with active LN during pregnancy are at higher risk of fetal loss. Women with antiphospholipid antibodies are also at high risk for spontaneous abortion and fetal loss along with thrombosis. A woman with a past history of thrombosis should be anticoagulated with low molecular weight heparin during pregnancy and would benefit from being treated with low dose aspirin. Women with CKD are at high-risk for preterm delivery and developing Pre-E.
Women with LN and APS should undergo extra fetal surveillance—especially in the third trimester—to screen for placental insufficiency and gestational size.
Other antibodies associated with lupus can cross the placenta and have deleterious effects on the fetus, including SSA and SSB antibodies. These antibodies are associated with the development of congenital heart block and fetal demise. Patients with these antibodies should be followed closely with fetal echocardiograms to screen for congenital heart block, other dysrhythmia, or myocarditis. Treatment with hydroxychloroquine decreases the risk of congenital heart block in women with anti-Ro antibodies.
Breastfeeding is encouraged in women with LN and can improve the wellbeing of both the infant and mother. Many medications taken for SLE are safe in breastfeeding including prednisone, azathioprine, tacrolimus, and hydroxychloroquine. There are no data on the use of belimumab or voclosporin in breastfeeding.
Summary of the safety of medications used in SLE in pregnancy and breastfeeding
| Safe in Pregnancy | Breastfeeding | |
| Glucocorticoids | Yes | Compatible |
| Hydroxychloroquine | Yes | Compatible |
| Cyclosporine | Yes | Compatible |
| Tacrolimus | Yes | Compatible |
| Voclosporin | No | Data limited, avoid use |
| Mycophenolate Mofetil | No | Data limited, avoid use |
| Azathioprine | Yes | Compatible |
| Cyclophosphamide | No, if organ threatening disease in 2nd or 3rd trimester may need to be considered | Contraindicated |
| Leflunomide | No | Contraindicated |
| Rituximab | No | Compatible |
| Belimumab | No | Data limited, avoid use |
Table adapted from Sammartino et al.
– Executive Team Members for this region: Anna Burgner @anna_burgner and Tim Yau @Maximal_Change
How to Claim CME and MOC
US-based physicians can earn 1.0 CME credit and 1.0 MOC per region through NKF PERC (detailed instructions here). The CME and MOC activity will expire on June 1, 2022.
Click to read more NephMadness 2022 Regions


Leave a Reply