#NephMadness 2022: Inequities Region
Submit your picks! | NephMadness 2022 | #NephMadness | #Inequities
Selection Committee Member: Dinushika Mohottige @DMohottige
Dinushika Mohottige is a Nephrologist and Assistant Professor in the Division of Nephrology at Duke University Hospitals. She works under the mentorship of Dr. Ebony Boulware and Dr. Clarissa Diamantidis to engage in patient- and community-centered, inequity-focused research around the impact of socio-structural factors on kidney health and kidney transplantation.
Writer: Katie Rizzolo @katierizzolo
Katie Rizzolo is a second-year nephrology research fellow at the University of Colorado. She is a graduate of Tufts University School of Medicine in Boston, MA, and completed her residency and chief year at Maine Medical Center in Portland, ME. Her clinical and research interests include immigrant health, social justice, and disparities in kidney care. She is a 2021 graduate of NSMC.
Competitors for the Inequities Region
Governmental Policies vs Institutional Initiatives
Environmental Determinants of Health vs Social Determinants of Health

Copyright: Iscotlanda Photography / Shutterstock
Introduction
The social determinants of health are the non-medical factors that influence health outcomes. They are the conditions in which people are born, grow, work, live, and age, and the wider set of forces and systems shaping the conditions of daily life. These forces and systems include economic policies and systems, development agendas, social norms, social policies and political systems. The social determinants of health have an important influence on health inequities – the unfair and avoidable differences in health status seen within and between countries. In countries at all levels of income, health and illness follow a social gradient: the lower the socioeconomic position, the worse the health. – World Health Organization
Kidney disease is strongly affected by social determinants of health, including economic, environmental, and living conditions. While individual factors such as demographics, health behaviors, and biological responses affect kidney health, their effects are attenuated by a complex interplay of social conditions.

Social determinants of health: key domains. Based on information available from the Healthy People 2020 initiative. Figure 2 from Hall, AJKD, © National Kidney Foundation.
Social categories, including race and immigration status, affect the health of the individual due to multiple factors such as access to education and health care, living conditions, and socioeconomic status. Reduced access to these opportunities, investments, and resources leads to greater burden of poverty and food insecurity, greater exposure to environmental stressors and pollutants, etc. These issues may be further exacerbated by discriminatory policies and treatment.

Fundamental influences on health (on the basis of Warnecke et al). Figure 1 from Boulware and Mohottige, CJASN, © American Society of Nephrology. Reproduced with permission.
Health disparities are differences in health that disproportionately affect vulnerable and disadvantaged groups, leading to adverse outcomes. Disparities in kidney outcomes and access to care—including pre-dialysis care, dialysis, and transplantation—exist around the world. In the United States, Black and Latinx/Hispanic people are 3.4 and 1.3 times more likely to develop end stage kidney disease (ESKD), respectively. Racial minorities are less likely to have a timely pre-dialysis nephrology referral, have an arteriovenous fistula for vascular access, or be treated with home hemodialysis (HD) or peritoneal dialysis (PD). Black patients are less likely to be referred for transplant evaluation, have longer wait list times, and are less likely to receive a pre-emptive transplant or living donor kidney transplant. These outcomes are a few of the many examples illustrating the complex interplay of biology and social conditions that leads to disparities in kidney health.
In the last few years there has been a growing interest in health equity research with the motive of identifying underlying systemic injustices contributing to health disparities and key strategies that are needed to narrow inequities. It is important to clearly define the meaning of “health equity” as it applies to research. First, we use the term “equity” rather than “equality” throughout this text, as, “equality means giving everyone the exact same resources, whereas equity involves distributing resources based on the needs of the recipients.” Second, it is also important to be clear on the definition of “health inequity” in order to ensure the target solutions align with equitable outcomes. Braveman and colleagues define “health inequity” as follows:
Health equity means that everyone has a fair and just opportunity to be as healthy as possible. This requires removing obstacles to health such as poverty, discrimination, and their consequences, including powerlessness and lack of access to good jobs with fair pay, quality education and housing, safe environments, and health care. For the purposes of measurement, health equity means reducing and ultimately eliminating disparities in health and its determinants that adversely affect excluded or marginalized groups.
In this region, we discuss kidney health inequities through examples illustrating the effects of policies, institutional initiatives, and physical and social determinants on equitable kidney health.
Governmental Policies vs Institutional Initiatives
Governmental Policies

Copyright: David A Litman / Shutterstock
Government policies and laws affect almost every aspect of healthcare, and nephrology care is no exception. These policies, for the sake of this discussion, are defined as the laws that guide access to healthcare, education, criminal justice, housing etc. Many key inequities are caused and driven by past and current public policy. Governmental decisions on health care access and resources play a huge role in the disparities we see across nephrology care in every nation. These policies, without an equity lens that ensures that we achieve health justice, often exert a differentially harmful effect on the outcomes of marginalized individuals and groups. This NephMadness team examines policies and initiatives around the globe and discusses how policy change may allow for more equitable care.

Infographic by @sophia_kidney and @katierizzolo
A Case
Hilda was a young mother and an undocumented immigrant with kidney failure living in Colorado (“undocumented immigrant” in the United States refers to a person without US citizenship who does not have current permission, such as a visa, to live and work within the country). In Colorado, undocumented immigrants with kidney failure requiring dialysis had very few options for treatment. Three-times-a-week maintenance in-center HD was not covered in Colorado by federal insurance options (eg, Emergency Medicaid, Medicaid) and private insurance is often prohibitively expensive. Without insurance, maintenance HD is expensive out-of-pocket (around $55,000 per year in 2002); thus, most undocumented immigrants rely upon “emergency-only HD”. The criteria for emergency-only HD vary widely throughout the country. For Hilda to be eligible to receive emergency-only HD in Colorado, she needed to demonstrate complications of kidney failure (eg, uremia, volume overload, hyperkalemia, metabolic acidosis, or mental status changes) in order to be admitted.
Without scheduled maintenance HD, Hilda would often show up to the hospital with nausea, vomiting, and shortness of breath requiring HD in the intensive care unit. After two years, she was too sick to work and became homeless. Her sons watched her collapse multiple times and be resuscitated over and over. Without other options, Hilda found an adoptive family for her sons, elected to stop dialysis, and eventually passed away. Her legacy lives on in the work of Dr. Lilia Cervantes, whose work is inspired by Hilda’s story, which she shared in 2020.
US Example: The Perils of Emergency-Only HD in the United States
Poor access to HD has broad implications for patients, families, medical providers, and the healthcare system. Patients who receive regular maintenance HD experience 5 fewer emergency department visits per month and 1.6 fewer hospitalized days per 6 months than those receiving emergency-only HD.
Emergency-only HD has a 14 times higher mortality rate compared to those receiving maintenance HD at 5 years, and emergency-only HD is associated with a higher symptom burden (nausea and severe psychosocial distress), higher burden on family, and increased fear of dying weekly. Caregivers of patients receiving emergency-only HD report distress and burnout, likely due to the moral injury of knowingly providing substandard care. Providers report feeling exhausted at betraying the patient’s trust and witnessing needless suffering and mortality. Emergency-only HD is costly; in the United States, one year of emergency-only HD is more than 4 times the cost of maintenance HD – $212K compared with $55K.
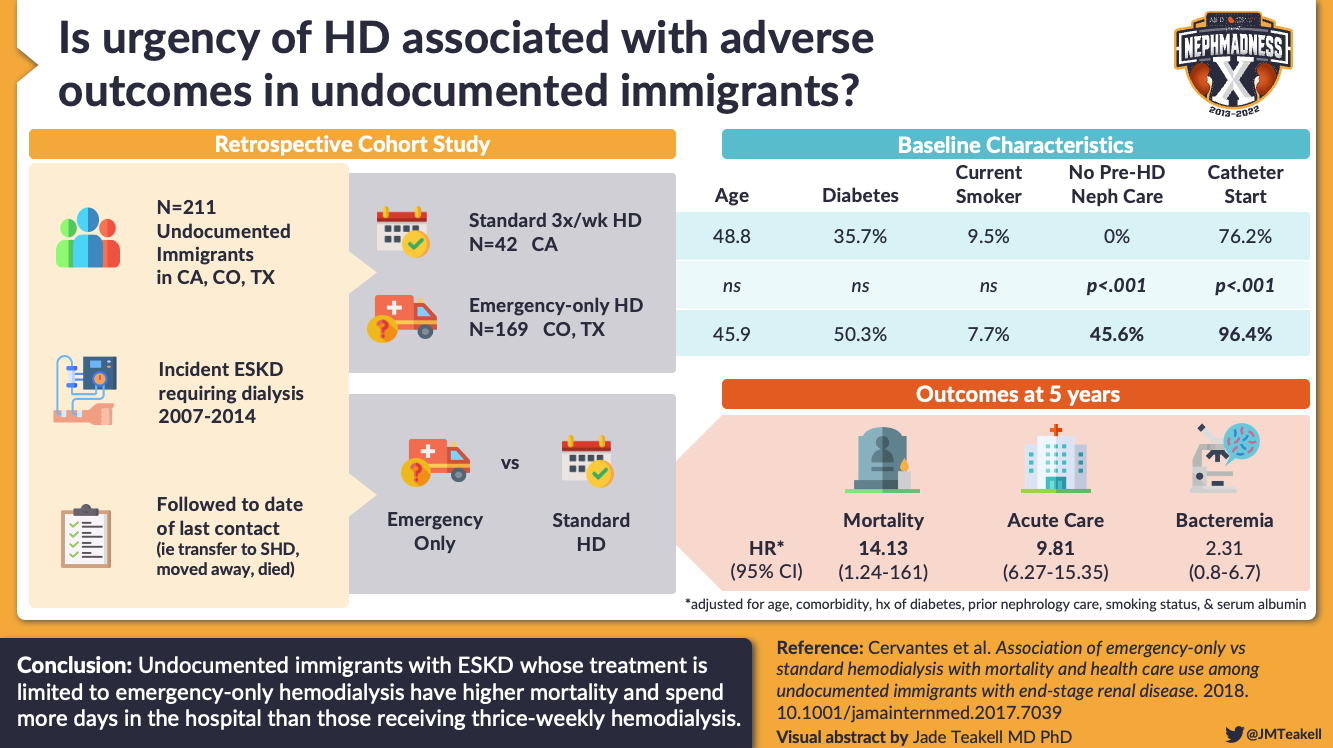
Visual Abstract by @JMTeakell on Cervantes et al.
Dialysis Policy for Undocumented Immigrants in the United States: What is an “Emergency”?
There are approximately 6,000-8,000 undocumented immigrants with kidney failure in the United States. The definitions of the main immigration categories and a summary of healthcare coverage of dialysis by immigration status are summarized below. While undocumented individuals are excluded from federal insurance policy, they are allowed emergency care under Emergency Medicaid. Emergency Medicaid was passed under the Emergency Medical Treatment and Labor Act of 1986, which prohibits hospitals from turning away anyone requiring emergency care, regardless of their ability to pay. An emergency is defined as:
“[…] a medical condition (including emergency labor and delivery) manifesting itself by acute symptoms of sufficient severity (including severe pain) such that the absence of immediate medical attention could reasonably be expected to result in:
- placing the patient’s health in serious jeopardy
- serious impairment to bodily functions
- serious dysfunction of any bodily organ or part.”
However, what is actually covered under Emergency Medicaid depends largely on the state’s definition of what exactly an emergency is, resulting in patchwork variability of maintenance dialysis care by state.
Notably, kidney transplantation eligibility for undocumented immigrants with kidney failure also varies largely by state. Citizenship status is not a barrier per the Organ Procurement and Transplantation Network Policy. Though Emergency Medicaid can be used for dialysis, it explicitly excludes transplantation “unless the state plan provides for written standards”. Barriers to transplantation for undocumented immigrants includes insurance access, language and culture concordant care, and ethical concerns. Several states such as California and Illinois do cover transplantation for undocumented immigrants with kidney failure via state insurance policies.

Table 1 from Cervantes et al, AJKD, © National Kidney Foundation.
International Examples: Dialysis Policy for Immigrants Outside the United States
Europe
In Europe, immigrants make up 1.5% of those requiring dialysis and their dialysis care varies widely. In France, emergency-only HD is used for the duration of a tourist visa (90 days). In Italy, dialysis and transplant care is provided without costs to all undocumented immigrants.
With a large number of refugees entering Europe, it is important to know what options will be available. Please refer to the table above defining different immigration categories. In one study surveying European physicians regarding refugee dialysis, 25% did not know how dialysis was reimbursed, and many were unclear on how refugees with kidney failure should be managed. Regardless, most physicians reported having a moral duty to provide dialysis to refugee patients who need it.
Africa
Data on kidney failure prevalence are limited in Africa. Kidney replacement therapy (KRT) resources are scarce: in 2015, 20 out of 100,000 people were treated with HD in Sudan, despite an estimated kidney failure prevalence of 159 per 100,000. Where KRT may be a scarce resource, immigrants may be denied maintenance dialysis. In 2019, a 36-year-old Ethiopian woman with kidney failure seeking asylum in South Africa was denied maintenance dialysis treatment in South Africa due to her lack of South African citizenship status. Notably, KRT access is limited in some areas of Africa. Given scarcity of KRT resources, this dialysis policy for immigrants may arise from an inability to supply adequate dialysis therapy for all patients with kidney failure.
Dialysis Policy in Eras of Forced Migration for Refugees
The impact of forced migration around the world has called current practices of dialysis for immigrants into question. The Syrian Civil War offers one such example. The overload of Syrian refugees in Lebanon after the Syrian Civil War exceeded both financial and staffing resources in many areas. This resulted in many private Lebanese hospitals refusing to treat patients if the hospitals did not have the financial or staffing resources. Emergency dialysis strained existing resources, causing many dialysis facilities to refuse patients. Prioritizing citizens over refugees in the setting of tight resources led to ethical dilemmas for nephrology providers. Syrian refugees underwent dialysis for free in public hospitals from 2011-2014. For the first two years, the government was unable to reimburse hospitals and doctors taking care of these patients; over time, fees were paid for by several charitable organizations. These issues are not unique to one country; other studied experiences include Afghanistan and Venezuela. In most countries receiving large numbers of immigrants, emergency-only hemodialysis was used almost exclusively. In most countries, there was no national policy for the treatment of refugees with kidney failure. In this case, the lack of government policy regarding dialysis treatment for immigrants resulted in poor care and diminished resources.
How to Change Policy
In 2019, Colorado elected to include “kidney failure” as an emergency condition, becoming the 12th state to do so. This action was due to work by a number of stakeholders, data gathering, policy analysis, and implementation of a mechanism for monitoring outcomes. For example, Colorado Medicaid concluded that emergency-only dialysis cost ten times more than maintenance HD (emergency: $20,291/person/month, maintenance in-center: $2,413/person/month). Patients previously on emergency-only dialysis, now receiving maintenance in-center HD, report improvement in quality of life and symptoms, though they still report apprehension.
Outside the United States, the impact of forced migration on dialysis access and care offers several lessons on policy. A national policy, engaging all stakeholders including providers, nurses, and dialysis companies is necessary to prepare for the potential influx of dialysis needs. An organized approach, in addition to universal health care access and availability of dialysis services in the accepting country, may greatly mitigate potential chaos and avoid exacerbating inequitable outcomes. Lastly, readily available immigrant/refugee chronic kidney disease (CKD) care may lessen the burden of dialysis patients on existing resources if kidney failure progression is slowed.
Call to Action
For those wishing to implement policy, the Colorado example offers some insights into how to go about it:
- Gather a coalition: Involve key stakeholders involved with the population in question. This may include patients, providers, non profit groups, dialysis organizations, and policy makers.
- Gather the data: Changing policy necessitates identifying the financial, as well as qualitative benefits
- Provide policy analysis from other states or areas and engage policy makers: Engaging those with memberships or established relationships within the governing body may help to demonstrate a clear path to policy change.
- Ensure a mechanism to monitor outcomes: For continuation of policy it is important to monitor clinical, financial, and operational outcomes, which may be useful for similar states or governments looking to employ similar policies.
Other ways to get involved in policy change for kidney patients:

Box 3 from Mendu and Weiner, AJKD, © National Kidney Foundation.
Institutional Initiatives
Institutions exist in healthcare, law, media, religion, financial, and political systems. An institutional initiative may thus be considered a practice that mandates or constrains actions and enforces compliance with applicable laws, ethical norms, accepted best practices, promotion of operational efficiencies, and reduction of institutional risks (adapted from Smith College). While these may vary across contexts even under the same government and laws (eg, nations, states), institutional initiatives can employ policies leading to inequitable care. Alternatively, institutions can employ laws which help reduce the impact of national and other governmental policies. When institutions develop and employ discriminatory policies, the institutions themselves contribute to inequities. In this section, we discuss the implications of initiatives both in the United States and globally in the context of 2 institutions: the prison system in the United States, and delivery of home dialysis around the world.

Copyright: Irene_A / Shutterstock
United States Example: Incarceration and Kidney Care
Incarcerated populations in the United States include multiple groups over-represented in CKD, including racial and ethnic minorities, patients with substance use disorders, and the elderly. While the exact burden of kidney disease amongst the incarcerated remains unknown, differences in kidney care access at the institutional level contribute to disparities in outcomes in patients with kidney failure who are incarcerated.
Kidney Disease Among Incarcerated Individuals
CKD is prevalent among incarcerated individuals, though the exact number is unknown. In 2015, the Bureau of Justice reported “kidney-related problems” in 6% of incarcerated individuals. Overall, Black individuals are 6 times more likely to be imprisoned than non-Black individuals, and Latinx/Hispanic individuals are three times as likely. Many investigators have described that Black people have worse outcomes in police encounters, bail and sentencing, and capital punishment in comparison to white people. In addition to implicit/explicit bias, inequitable practices in policing and criminalization stemming from existing or former policies have contributed to these racial inequalities in incarceration and police violence.
Racial minorities are also at high risk for CKD; compared to non-Hispanic whites, Black people are 3.4 times more likely to develop kidney disease, and Hispanic people are 1.3 times as likely to develop kidney disease. Comorbidities are common among incarcerated individuals and higher than that of the general population. Forty-three percent of inmates in state prisons and 36% of those in federal prisons are diagnosed with at least one chronic illness. The most common among these are obesity, diabetes, and hypertension. Substance use is also common among incarcerated individuals compared with the non-incarcerated population, most commonly smoking and alcohol use. The incarcerated population is aging; 12% of those carrying a >1 year sentence are over 55 years old. Age, substance use, and diabetes/hypertension are all risk factors associated with development of CKD, making this population particularly susceptible to developing kidney failure.
People who are incarcerated have a constitutional right to health care per the Supreme Court 1976 ruling Estelle vs Gamble. However, there is very little data on access, quantity, and quality of health care available for people who are incarcerated. Access to lab testing, screening services, and medical providers is quite variable depending on the institution. Notably, almost all US states suspend Medicaid benefits upon incarceration, which may make insurance difficult to obtain upon release from prison.
Kidney Disease Management for the Incarcerated Individual
There is scant data on the management of CKD and kidney failure in the incarcerated population. Twenty-four states report dialysis services on site, 10 states report dialysis availability off-site, and the remaining 16 states report a mix of dialysis availability (Murphy et al 2021). While data on PD availability is not well studied, one pilot program at the University of Connecticut indicate that many incarcerated people are able to perform PD well.
Kidney transplantation is not readily available for the majority of incarcerated individuals with advanced kidney disease. Though the Organ and Procurement Transplantation Network states “one’s status as a prisoner should not preclude them from consideration for transplant”, most inmates requiring KRT receive HD, despite HD being more costly and inferior for survival in comparison to transplantation. Only 19% of kidney transplant programs report at least one incarcerated individual on the kidney transplant waitlist. Many transplant programs preclude inmates from consideration for transplantation, though these listing decisions are made in the absence of uniform criteria. Barriers may include the logistical hurdle of bringing inmates to hospitals on short notice, issues with getting lab results rapidly, and barriers to follow up, especially if the inmate is released from prison post transplant.
Individual institutional/prison policy changes demonstrate how improvements can be made in incarcerated individuals’ access to transplantation. The University of Buffalo offers one such example. Their transplant program worked with the local prison administration to improve access to transplantation for individuals with advanced kidney disease. Referral was made on condition of good behavior and adherence to HD and medications. These efforts resulted in 45 referred, 27 listed, and 18 successful transplants between 2003 and 2018. Results showed a 94% kidney graft survival within the first year, and 72% kidney graft survival at 3 years. The financial benefits per patient showed $80,941 in the first three years, and $60,749 per year thereafter compared with HD. The biggest issue was loss-to-follow up. The authors note over 50% of incarcerated individuals who received a transplant over the last 15 years were unable to be located. Given that many individuals move after being released from prison, and insurance access may vary by state, there is room for improvement regarding transplant follow up.
Next Steps
Murphy et al outline multiple areas for improvement in institutional policy to allow for equitable access to kidney care in incarcerated individuals. Steps may include the following:
- Implement health care screening, including medical provider and lab testing.
- Promote healthy living habits (for example, limiting smoking, healthy food).
- Link individuals with CKD to care in the community on release from prison.
- Build institutional frameworks as those mentioned above (medical provider and testing) which will allow better access to home dialysis, transplant.
- Coordinate transplant programs with multi state prison authorities to improve transplant follow up.
International Example: Home Dialysis Initiatives
Institutional policies have powerfully shaped home dialysis uptake in a few areas around the world, and provide an example of how individuals institutions can deploy policies that advance equity.
Home dialysis (home HD and PD) is widely viewed as an equivalent method for KRT compared with in-center dialysis, yet in-center HD remains more widely used worldwide. Home dialysis is superior to in-center dialysis in multiple areas, including higher quality of life and cost savings, and is comparable in clinical outcomes. In-center dialysis’ popularity is not driven by patient preference or quality of life, cost, or clinical measures.
PD in low and middle income countries (LMICs) has been hailed as a more accessible, sustainable, and cheaper option, especially as it can be utilized in areas without electricity. PD has reduced needs for technical support, medical staff, and electricity, and greater flexibility in natural disasters. In some LMICs, urgent start PD is preferred to HD given the high rate of vascular access infections associated with central venous catheters. In areas such as sub-Saharan Africa, KRT access is extremely limited and mainly concentrated in urban areas. PD is used in several LMICs across Sub-Saharan Africa as it can be used in a low-technology setting, though many barriers to access still remain. In some areas of the world, PD use is on the rise, such as in China and Thailand. In other areas, home dialysis use has stagnated or even fallen. The reasons for lack of uptake in home dialysis is multifactorial: barriers to home dialysis exist at the patient, provider, and system level.
Barriers to home dialysis (adapted from Weiner et al)
| Level | Barrier |
| Patient | |
| Environmental: limited space for supplies at home | |
| Social: lack of social support, fear of performing dialysis independently | |
| Health literacy: insufficient home dialysis education, socioeconomic status, pre-dialysis nephrology care | |
| Provider | |
| Limited CKD pre-dialysis education | |
| Provider enthusiasm (or lack thereof) | |
| Inadequate home dialysis education for fellows | |
| System | |
| Lack of pre-dialysis insurance | |
| Urgent-start standard modality is hemodialysis | |
| Home dialysis clinics often not available in rural communities | |
Nine countries boast >20% home dialysis use overall: Canada, Netherlands, Iceland, Finland, Denmark, Australia, New Zealand, Mexico, and Hong Kong. While some countries have instituted policy at the government level (for example, “PD First” policies in Hong Kong or Thailand), many changes to home dialysis uptake have been instituted at the institutional level. Institutional policy changes addressing barriers described in the table include patient education infrastructure, availability of home dialysis related care, access to urgent-start PD programs, and provider experience and training.
In this section we examine three examples of grass-roots policy change at the institutional level that aimed to reduce inequities in home dialysis use around the world stemming from patient, provider, and health system-level interventions and activation.
Patient Level: New Zealand
In many areas of the world, space for dialysis remains a barrier to home dialysis, inequitably disenfranchising those of lower socioeconomic status. One institutional policy change in New Zealand helped to increase home dialysis uptake for those who may have social, economic, or cultural barriers. Community house HD programs allow those with unstable or unsuitable housing for home dialysis to dialyze in an independent community setting. This initiative was created by dialysis providers and patient support groups. The first home HD community house patient was funded in 2001 by a nephrology charitable association, but funding quickly increased with interest. Due to growing demand, the Kidney Society of Auckland bought a community house, and the initiative grew from there. Qualitative studies illustrated that local Maori and Pacific people found community house dialysis much more acceptable. Community house HD allowed those with socioeconomic or cultural barriers—often from marginalized backgrounds—to gain access home dialysis, allowing for more equitable care.
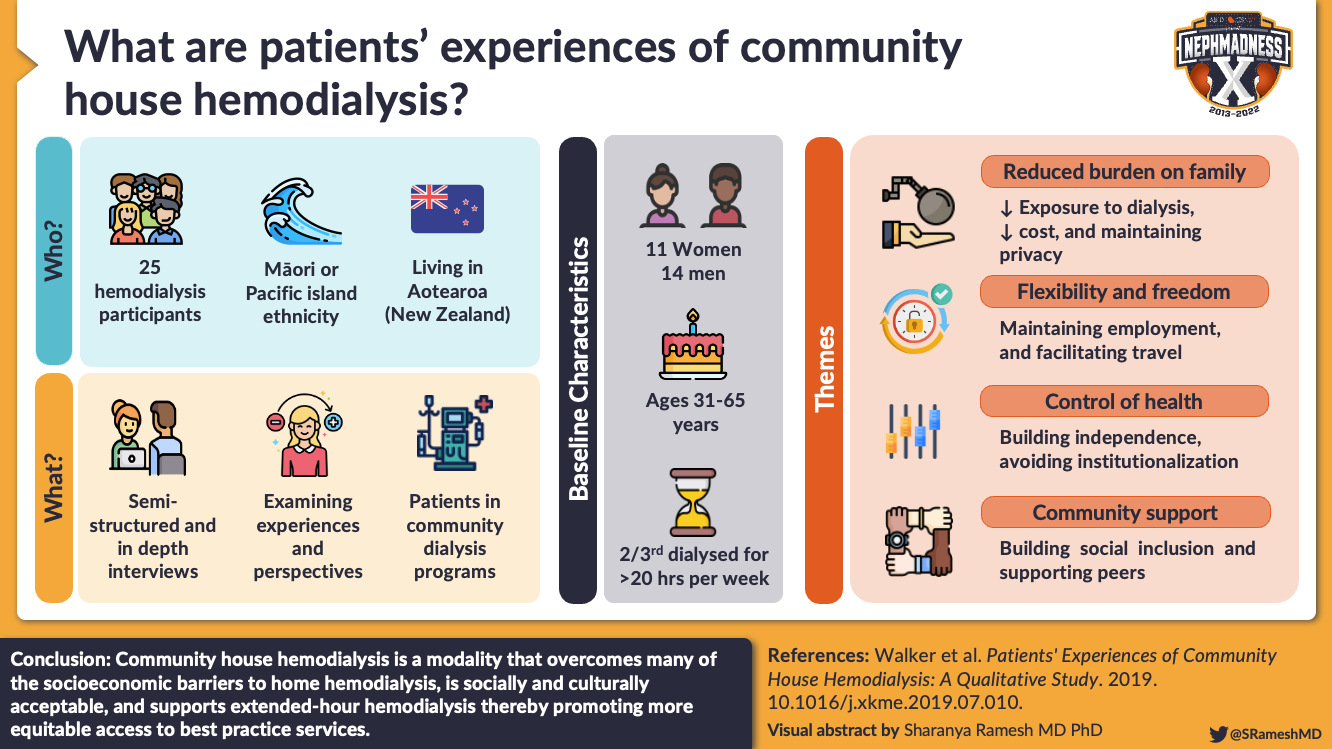
Visual Abstract by @SRameshMD on Walker et al.
Provider Level: Mexico
A key determinant of success in institutional policy change is provider enthusiasm. In many areas of the world, lack of professional expertise in home dialysis techniques remains a barrier to its use. Mexico is a powerful example of provider enthusiasm shaping the institutional infrastructure driving home dialysis uptake. In the 1940s-1960s, PD was used to treat acute kidney failure in Mexico. The introduction of continuous ambulatory PD in the late 1970s allowed for widespread use of continuous ambulatory PD for maintenance dialysis for patients with kidney failure. Today, Mexico’s PD population makes up ¼ of the entire world’s PD population (incredible!), and around 65% of all dialysis patients in Mexico are on PD. There are several reasons for this—provider enthusiasm, established acute start programs, and successful training programs—all championed by an avid home dialysis advocate.
PD first became widely used in Mexico City, where the first nephrology training program was established, allowing for the wide dissemination of expertise in Latin America. In the early 1970s, Dr. Alejandro Treviño Berrecera, a Mexican nephrologist, spent a year studying catheter placement, PD patient management, and methods of home PD use in the United Kingdom. Along with his tutor, he brought skills back to Mexico via a training course at the National Medical Center. A year later he organized an International PD symposium attended by nephrology physicians throughout Latin America. He also wrote a PD book in Spanish (Peritoneal Dialysis in the Management of Chronic Renal Failure) that is still in use today. Along with his nephrology colleagues he advocated for Baxter to start producing PD fluid in Mexico, making it easier for nephrologists to obtain supplies. With improved training and supplies, PD uptake escalated greatly, to over 10,000 patients by the 1990s.
System Level: Canada
One such example of institutional policy change at the system level is urgent start PD programs. HD is the predominant dialysis modality for urgent dialysis initiation worldwide. The majority of patients who initiate dialysis suboptimally (eg, inpatient, via central venous catheter, and/or not on their chosen modality) then remain on maintenance in-center HD after their initiation. Urgent-start PD programs offer an alternative to urgent-start HD. One urgent-start PD program in Canada used fluoroscopic and laparoscopic insertion methods for PD catheters. In their study, six patients started PD right away after the catheter was placed, and all patients commenced PD within two weeks of catheter insertion. The program experienced nearly a 50% growth in their PD program over three years, from 90 to 133 patients.
A similar program in Singapore noted several essential elements for an urgent PD start program:
- Capacity for outpatient care of low-volume PD exchanges
- Adequate nursing staff to supervise low-volume PD exchanges
- Process in place for urgent PD catheter insertion
- Dedicated outpatient home dialysis clinic
- Dedicated PD coordinator for the above processes
Next Steps
Creative institutional initiatives observed worldwide offer insight into effective institutional policies that improve home dialysis access. These demonstrate how patient, provider, and system-level inputs may help develop institutional policy and practice solutions that tackle inequities, even when government policies fail to do so.
Potential solutions may include the following:
- Investigating creative ways to expand home dialysis access
- Improving reimbursement
- Increasing learning opportunities for trainees
- Creating user-innovative models to expand access for those with potential barriers
Check out this podcast episode of The Drs. Washington featuring Katie Rizzolo @katierizzolo and Dinushika Mohottige @DMohottige:
Inequities in Nephrology
Environmental Determinants of Health vs Social Determinants of Health
Environmental Determinants of Health

Copyright: travelview / Shutterstock
Disparities in physical contexts including access to care, environmental exposures, access to healthy food, and housing stability are important drivers of kidney disease. Marginalized communities (characterized by a greater proportion of minority individuals and lower socioeconomic status) are often exposed to high levels of pollutants and other environmental toxins, and have lower access to health care, and healthy foods, to name a few. We review two examples demonstrating implications of physical contexts and determinants on kidney health disparities in the United States and worldwide.

Infographic by Denisse Arellano and Jade Teakell.
US Example: Food Insecurity and CKD in the United States
Food security is defined as the perceived ability to obtain nutritious and healthy foods with essential nutrients, per the US Department of Agriculture. In areas where healthy food is scarce or expensive, healthy foods—especially fresh vegetables and fruits—are replaced by cheap, energy-dense foods which are often low in nutritional value. 10.5% of American households are considered food insecure as of 2020.
Food Insecurity and Outcomes in CKD
Lack of access to healthy foods increases the risk of diet-related diseases, including heart disease, kidney disease, diabetes, and hypertension. A diet high in sodium, often found in prepackaged foods, can worsen blood pressure, volume management, and cardiovascular events. Dietary acid load, higher in “unhealthy” diets due to the lack of fresh fruits and vegetables, is associated with CKD progression. Nutrition has been linked to blood pressure control, hyperlipidemia, anemia, metabolic acidosis, and bone/mineral disorders.
Food insecurity is associated with progression to kidney failure independent of income, clinical risk factors, and kidney markers (eGFR and albuminuria). In an earlier study, high food insecurity was associated with greater odds of CKD only in patients with diabetes or hypertension. Higher dietary acid load is more likely in groups with lower socioeconomic status (SES) and in racial minorities.

Visual Abstract by @deniise_am on Banerjee et al.
Relationship of nutrition, physical context, and CKD
The availability of healthy foods for patients with CKD relies upon the availability of grocery stores, decreased density of fast food options, transportation, and affordability of healthy foods. In particular, diets that are high in fat, sodium, and phosphates are more common in low-income areas, and more likely to be linked to chronic disease:
- High fat diets– These are more common in lower income neighborhoods where low nutrient energy dense items are more available. High intake of energy dense foods can lead to obesity and CKD.
- High sodium diets– In the United States and the United Kingdom, the greatest source of sodium is additives in processed foods. Those with lower neighborhood supermarket density are more likely to consume processed foods.
- High phosphate– Phosphates are used as an additive in meat and baking products, and processed food is more likely to have a higher phosphorus load. Lower annual family income is associated with higher serum phosphorus levels.
- High acid loads– Western diets include a higher acid load due to high concentration of animal and grain products. High dietary acid load is associated with albuminuria and progression to kidney failure.

Both individual and contextual socioeconomic factors affect the quality of nutrition in individuals with CKD. As depicted in the figure, these factors are often interconnected, such that any one factor can accentuate the adverse impact of the others, compounding the overall adverse effects of poverty on the ability of individuals with CKD to follow nutritional recommendations for a healthy diet. Figure 1 from Gutiérrez, ACKD, © National Kidney Foundation.
Disparities in access to healthy foods
The term “food deserts” was widely popularized in the 1970s and 1980s to describe areas where people have “limited access to a variety of healthy and affordable foods”. One US analysis found 20% of rural counties are considered food deserts, meaning people live more than 10 miles from a supermarket. In these areas, there are often few transportation options, both in urban and rural environments. The concentration of food outlets supplying healthy foods influences the ability to comply with dietary recommendations in kidney diseases and other chronic illnesses.
The racial make-up of these areas labeled as “food deserts” are often predominantly Latinx and Black populations. In one study, 8% of African Americans lived in a census tract with a supermarket, compared with 31% of non-Latinx White people. Even when comparing to areas of similar socioeconomic status, Black and Latinx neighborhoods have a higher number of convenience and liquor stores and fewer supermarkets, leading to a scarcity of healthy foods such as fruits and vegetables. Contributing to this disparity is the discriminatory practice of redlining, a US Housing policy in the 1930s restricting housing loans and other financial opportunities for racial minorities. This policy led to racial disparities across US neighborhoods, manifesting racially segregated healthy food outlets (among others). It should be noted in this light that the term “food deserts” does not completely encapsulate how systemic racism and subsequent policy contribute to today’s inequities of food security.
Lower SES is correlated with food insecurity; almost half of US residents living two times below the poverty line are food insecure. Many studies have shown lower SES is linked with higher intake of fats, sugars, salt, and grains linked to adverse outcomes as described above. Lower access to supermarkets and higher proximity to fast food restaurants is correlated with lower household income. In addition, lower income zip codes have 30% more convenience/liquor stores. Supermarkets in lower SES neighborhoods, when they exist, are less likely to stock healthy options. The lack of fresh, healthy foods likely contributes to higher dietary acid load observed in patients who are food insecure. Those of lower SES are also more likely to develop kidney disease. This may in part be due to diet quality for those experiencing food insecurity, though this causal relationship is difficult to determine.
Overall, contextual determinants of health such as neighborhood, availability of healthy food outlets like supermarkets, transportation access, and density of fast food restaurants/convenience and liquor stores hinders healthy eating. Healthy eating is linked to lower risks of kidney disease, and those most at risk of food insecurity and kidney disease are racial minorities and those experiencing poverty.
International Example: Environmental Exposures and Kidney Disease Outside the United States
Inequitable pollution and environmental exposures have been associated with poor kidney disease outcomes. There are clear environmental factors driving the risk of kidney disease that affect racial minorities and those of lower SES. These groups are more likely to be exposed to environmental toxins that negatively affect the kidney. We provide three examples of environmental exposures linked with CKD, demonstrating the impact on vulnerable communities.
Air Pollution
While pollution is a global issue, those most affected are racial minorities and people residing in low SES areas. Multiple studies show fine particulate matter <2.5 um (PM2.5) is associated with increased risk of CKD, kidney failure, and increased mortality risk from kidney disease. In one study of 10,997 participants, exposure to higher annual fine particulate matter was associated with higher levels of albuminuria and increased risk of kidney disease.
However, the distribution of who is affected by air pollution is not equitable; 12% of global premature deaths are related to pollutants that are from outside the region where the death occurred. Individuals who are Black and socioeconomically disadvantaged are more likely to be exposed to air pollution. It is estimated that fine particulate air pollution may contribute to up to 20% of CKD worldwide, with highest burden in Mesoamerica, Northern Africa, Afghanistan, Pakistan, India, and a few countries in the Eastern Mediterranean regions.
Occupational Exposures
CKD prevalence has been steadily increasing in Central America, accounting for 20,000 deaths in the last ten years. In particular, many young men aged 20-40 who work in sugarcane cultivation in Central America have been noted to develop CKD of Unknown Etiology (CKDu). For some areas, kidney disease has become truly endemic, giving rise to the name Mesoamerican Nephropathy (often termed Chronic Interstitial Nephritis in Agricultural Communities [CINAC]). CINAC is one such example of the physical environment affecting kidney disease, leading to disparities in incidence, prevalence, and treatment of CKD.
Sugar cane is a profitable and reliable industry, as many regions rely upon Central American sugarcane production. Workers harvest sugarcane 6-7 days per week for up to 12 hours a day, and are often paid on the amount of sugarcane produced. Most workers do not receive personal protective equipment despite harsh working conditions, which can lead to extreme heat exposure and dehydration.
While the pathophysiology is not completely worked out, these patients are found to have tubulointerstitial nephritis. The proposed etiology is likely multifactorial due to direct toxins in agrochemicals, pollutants, and heavy metals, in addition to volume depletion, heat/rhabdomyolysis, and endemic infections such as leptospirosis and hantavirus. These patients are often young, and do not have any of the typical conditions known to cause kidney disease, such as hypertension or diabetes.

Possible causes for Mesoamerican nephropathy. Figure 2 from Correa-Rotter et al, AJKD, © National Kidney Foundation.
CINAC is also found in other members of poor agricultural communities, including cotton, corn, shrimp farm workers. Similar kidney diseases have been found in Sri Lanka (possibly due to exposure to hazardous water and other agricultural exposures) and amongst rural farmers in India (often in warm rural areas where farming coconuts, cashews, or rice is common).
Sugar cane farmers are often migrants, and sugar cane production is lucrative as it offers financial stability and decent wages. In Costa Rica, for example, migrants are not offered healthcare access, and are much less likely to receive health insurance, leading to delays in presentation for kidney disease with considerable out of pocket costs. Moreover, many countries in Central America lack kidney disease registries, making patient outcomes difficult to track. Unfortunately, given limited health records, inadequate healthcare, and low amount of research funding, research into CINAC was difficult for many years.
Next steps
Recognition of the contextual determinants of health—including but not limited to food access, environmental toxins, pollution, green spaces, and access to healthcare—is critical to addressing disparities in kidney health. Once recognized, early education of those at risk, and prevention of deleterious environmental factors are necessary. For example, modification of work environments and personal protective equipment for those at high risk for CKDu may prevent progression to kidney failure. Community-based solutions may be able to provide healthy options for food and outdoor space. In preventing kidney disease, policy change (in both local and national governments) is necessary to ensure equitable access to healthy living conditions/health-promoting resources.
COMMENTARY BY PATRICK GEE:
Whom Do You See When You See Me?
Please, My Humanity Over My Race
Social Determinants of Health
The social environment is a powerful element contributing to and influenced by disparities in kidney disease. The environment in which a person lives, plays, and works is associated with their well-being, and exposure to social environmental stressors or influences can influence risk of kidney disease. Many factors associated with well-being are shared within a community, including exposure to stress, discrimination, incarceration, and social cohesion. Socio-environmental contexts may shape or influence psychosocial, behavioral, and medical risk factors mitigating kidney outcomes.

Copyright: James Mercier / Shutterstock
US Example: Discrimination and Kidney Transplantation
While kidney transplant is considered the gold standard for treatment of kidney failure, there exist numerous racial inequities along every step of the path to transplant. Even when accounting for social determinants of health, Black patients in the United States are less likely to be referred for transplant evaluation, have longer wait list times, and are less likely to receive a pre-emptive transplant or living donor kidney transplant.
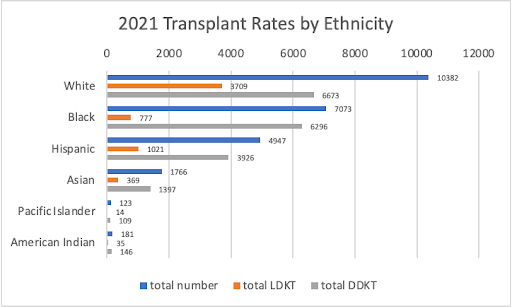
Figure created from UNOS data.
Socioeconomic status and race are correlated and each is important. This is because sociopolitical systems in many contexts are inherently racialized, such that economic advantages and other resources are differentially distributed across individuals based on race/ethnicity and other social domains. For example, fewer financial resources, lower education levels, and residence in lower SES neighborhoods are associated with a lower likelihood of transplant listing.
Multiple studies have demonstrated that racial discrimination plays a key role in the racial disparities observed in kidney transplantation. Perceived discrimination—in addition to transplant knowledge and lower income—has been independently associated with longer time to waitlist acceptance. In addition, Black and other minoritized patients reported more discrimination in healthcare, more perceptions of racism in healthcare, and a higher degree of medical mistrust. Similar findings are notable regarding the role of discrimination, medical mistrust, and perceived racism, which were associated with lower initiation of transplant evaluation among individuals with kidney failure in the Southeastern United States.
Another study also found the experience of racial discrimination reduced the likelihood of waitlist listing for Black patients, especially when factoring in lifetime discrimination experiences outside of those experienced in medicine. The authors note one reason for this finding may be that “victims of discrimination learn to anticipate poorer outcomes than advantaged members of society and become reluctant to enter into situations in which they expect to be treated unfairly.”
Latinx people with kidney failure also experience discrimination in the setting of transplantation, while working within a culturally and linguistically discordant system. Latinx patients are much less likely to receive a living donor kidney transplant. Latinx patients with kidney failure in some studies demonstrate low understanding of transplant processes and the benefits of transplant. In part, this may be due to lack of language-concordant information. A national survey of Latinx/Hispanic individuals showed language concordance is associated with less confusion, frustration, and perceptions of poor quality health care. In addition, many Latinx patients report perceptions of discrimination in health care, which may be mitigated through interventions that address provider biases, both explicit and implicit. Many report mistrust of the medical system due to fear of deportation amongst undocumented immigrants, and fear their organs will be sold.
Next Steps
A culturally and structurally equitable health care system acknowledges and incorporates a patient’s culture into shared and informed clinical decision-making and patient care through an understanding of cultural differences, cultural knowledge, and individualized care to meet culturally unique needs. Language- and culture-concordant education can increase favorability of transplant and organ donation rates amongst Latinx and Black patients. Further, the health care system must demonstrate trust and a commitment to equity. Engagement with the community and adapting practices and policies centering on the needs of marginalized communities are key.
Some examples of culturally and structurally concordant education interventions for racial and ethnic minorities include the following:
- Mass media interventions for living donors
- Culturally- and linguistically-competent websites
- Transplant education via house calls
- Live culturally sensitive education sessions for living donors and recipients
- Peer navigators for transplant process
- Social worker and donor financial assistance programs for living donor kidney transplant
- Culturally and linguistically competent transplant centers
- Culturally sensitive immunosuppression adherence education
International Example: Traditional Medicines and Kidney Disease
Social environmental factors shape perceptions of health care and disease. One such example is the role of traditional medicines. Traditional medicines broadly refer to health practices utilizing animal, plant, or mineral based medicines or therapies. The WHO defines traditional medicines as the “sum total of the knowledge, skill, and practices based on the theories, beliefs, and experiences indigenous to different cultures, whether explicable or not, used in the maintenance of health as well as in the prevention, diagnosis, improvement or treatment of physical and mental illness.” In comparison, biomedicines, for the sake of this article, are medicines or practices that have gone through rigorous scientific study and regulation and are prescribed by a licensed profession. While biomedicines are often referred to as Western or conventional medicines, this term is not necessarily synonymous.
In many parts of the world, in particular low and middle income countries, traditional medicines represent the predominant form of healthcare access. Traditional medicines contribute to ensuring access to health care in these regions—care that is affordable, accessible, close to hold, and culturally accepted and trusted. Elucidation of how traditional medicines may be incorporated into the care of kidney disease is thus vital to develop safe and effective public health strategies across the world. We evaluate the different uses and harms of traditional medicines in Africa, China, and India and examine their role in kidney disease.
Africa
CKD represents a significant health burden in Sub-Saharan Africa due to the risks of non-communicable and communicable disease. The prevalence of CKD and AKI in Africa has increased quickly, especially in dense cities where etiology may be multifactorial in the setting of environmental toxins, infectious disease, and non communicable diseases. As such, the role of traditional medicines in treatment and prevention of kidney disease is critical.
Traditional medicines are widely used throughout Africa. Aloe vera is one such example, used in Southern Africa for a variety of ailments, including arthritis, burns, skin conditions, hypertension, dyspepsia, inflammation, and infertility. Traditional medicines vary by region and represent distinct cultures, history, and traditions, and are used in both urban and rural areas by all socioeconomic classes. They may treat an array of non-communicable diseases including kidney disease, hypertension, and diabetes. In some areas, 70% of people with CKD—many of whom are unaware that they have CKD—are using traditional medicines for a variety of purposes, but may be causing harm to their kidneys unknowingly.
Traditional medicines are often used as “cleansing agents”, with the understanding that the kidneys are a filtering agent that cleans the blood. While there are treatments for kidney disease, most fit into a culture’s understanding of how the kidneys affect overall health. Many traditional medicines are known to be nephrotoxic. For example, Aloe vera has been associated with volume depletion, electrolyte imbalance, acute tubular necrosis, and acute interstitial nephritis.
The safety of traditional medicines in Sub-Saharan Africa necessitates more investigation. A few studies have demonstrated that up to 35% of AKIs are associated with nephrotoxic traditional medicines. Harms include the following:
- Direct Nephrotoxicity– Some traditional medicines are directly nephrotoxic, while others may worsen pre-existing kidney disease. It is difficult to establish safe doses, especially with little to no rigorous scientific studies.
- Polypharmacy– Often, traditional medicines are taken side by side with biomedicines, and the interactions of these medications has not been well studied. For example, some traditional medicines interact with HIV treatment.
- Access to kidney care– Given the silent nature of kidney disease until late stages, there can often be a delay in presentation. Delayed presentation is highly morbid in sub Saharan Africa.
China
Traditional Chinese medicine is over 5,000 years old, and is well institutionalized and integrated within the Chinese health care system, making up around 20% of all health care services. There are 120 million people in China with CKD. Many traditional medicines focus on restoring balance, body nourishment, and eliminating heat and dampness.
Several traditional Chinese medicines have been associated with positive kidney outcomes, including a reduction in proteinuria, kidney fibrosis, and prevention of oxidative injury:
- Sairei-to has been shown to reduce urinary protein and hematuria in children with IgA nephropathy
- Tangsen Formula has shown reduction of macroalbuminuria and improvement in eGFR in diabetic nephropathy
- A mixture of traditional Chinese medicines and an ACE inhibitor showed more reduction in albuminuria and improvement in eGFR compared with ACE inhibitor alone
However, some medicines can cause harm. For example, aristolochic acid has been used for edema or kidney/urinary ailments. It is known to be an anti-inflammatory with bactericidal and diuretic effects. Unfortunately, aristolochic acid is also associated with Balkan endemic nephropathy, chronic interstitial nephritis, and urothelial carcinomas.
There are several harms for patients with CKD who are also taking traditional Chinese medicines, including the following:
- Polypharmacy– Patients are often prescribed both biomedicines and traditional Chinese medicines, which may exacerbate potential nephrotoxic side effects.
- Heterogeneity of ingredients and preparations– Traditional Chinese medicines are often modified for the individual, and individuals may modify the medicines themselves depending on their needs. The multitudinous combinations and dosing adjustments for patients with kidney disease are important to consider. Traditional Chinese medicines are prepared in a variety of ways, which alter the toxicity of individual ingredients.
- Availability– Though traditional Chinese medicines should be prescribed and used following a pathway of evaluation, diagnosis, and treatment, they are widely available to practitioners who may not be trained in this pathway. Further, many foods and dietary supplements are widely available for individuals, who may not know the harms of the medications.
India
Multiple forms of traditional medicines are widely present in India, including Ayurveda, yoga, naturopathy, Unani, and Siddha. These practices are institutionalized into the healthcare system, co-existing with today’s biomedical practices. Over 100,000 people enter KRT programs in India, so evaluating the role of traditional medicine in treatment of kidney disease in India is necessary for prevention, management, and patient education.
Ayurveda is the most complex and ubiquitously used traditional medicine in India, often existing in rural settings (home to 70% of India’s population). In Ayurvedic medicine, kidney disease is seen as an imbalance amongst the three body humors (doshas). Treatment focuses on restoring balance via diuresis, anti-inflammatories, glucose lowering, and erythropoiesis. Some traditional medicines within Ayurveda have shown positive effects on kidney outcomes, such as proteinuria reduction in diabetic nephropathy. Some others have mixed benefits and harms. For example, Dolichos biflorus (Doob grass) is used for edema, nephrolithiasis, weight loss, oligomenorrhea, and constipation. It may cause diuresis, reduced calcium oxalate excretion, and inhibition of adipogenesis or increased lipolysis.
Safety evidence for ayurvedic medicines is limited. Much like traditional Chinese medicines, polypharmacy with biomedicines and the heterogeneity of ingredients and preparation may exacerbate any potential nephrotoxicity. This is especially important as nephrotoxic drugs remain a leading cause of AKI in India (along with infectious disease). In addition, Ayurvedic medicines have been associated with heavy metal toxicity. Some traditional medicine formulations include heavy metals such as lead, arsenic, cadmium, and mercury due to preparation and processing. Cases of heavy metal toxicity arising from these medications have been reported.
Next Steps
Recognition of the role of traditional medicines in different social contexts is crucial for CKD treatment around the world. Traditional medicine has an important cultural role in care, and is often the most readily available healthcare in low and middle income countries. Traditional medicine practices are reflective of the culture and health belief models of the area. An understanding of these practices is imperative in treating and preventing kidney disease. Traditional medicines may ameliorate, worsen, or cause CKD depending on the practice (the table below illustrates additional examples worldwide). Public health initiatives, and collaboration with traditional medicine practitioners and community members may help elucidate and address how cultural practices can be used to optimize the surveillance, treatment, and prevention of chronic kidney disease.

Table 2 from Luyckx, ACKD, © National Kidney Foundation.
COMMENTARY BY EBELE UMEUKEJE:
Social Determinants of Health:
The Biggest Elephant in the Room
Conclusion
Kidney disease is a result of a complex interplay between biologic and socio-environmental factors. For instance, government policies can drive inequities through discriminatory limitations in access to health care. Institutional policies or initiatives can worsen inequitable access to care through unequal treatment dependent on social categories such as immigration status, incarceration, or race. Yet both government and institutional policies and actors play an important role in the development and implementation of interventions that could address and undo inequities.
Physical determinants include possible inequitable exposures to stressors or potentiators of kidney disease, such as pollution and health harming food landscapes. Social determinants, including exposure to racism and the harms of racialized policies and practices, also drives inequitable access to care, resource availability, and kidney outcomes. It is important for nephrology providers to be aware of how social conditions may impact kidney health. Further research and work in this arena necessitate a holistic lens addressing social, biological, and behavioral forces affecting kidney care.
– Executive Team Members for this region: Matthew Sparks @Nephro_Sparks and Samira Farouk @ssfarouk
How to Claim CME and MOC
US-based physicians can earn 1.0 CME credit and 1.0 MOC per region through NKF PERC (detailed instructions here). The CME and MOC activity will expire on June 1, 2022.
Click to read more NephMadness 2022 Regions


Leave a Reply