#NephMadness 2022: Hemodialysis Region
Submit your picks! | NephMadness 2022 | #NephMadness | #Hemodialysis
Selection Committee Member: Daniel Weiner @DanTheKidneyMan
Daniel Weiner is a nephrologist at Tufts Medical Center, interested in CKD, dialysis, and just about everything else. He also really likes basketball, and (deep sigh) is a Duke fan. As a former AJKD Deputy Editor, he was there at the beginning of NephMadness, when Dick Vitale was yelling for home dialysis to be the diaper dandy. (OK, that didn’t really happen, but it should have.) Dr Weiner is Editor-in-Chief of Kidney Medicine.
Writer: Bourne Auguste @bourneauguste
Bourne Auguste is an Assistant Professor in at the University of Toronto and a staff nephrologist at Sunnybrook Health Sciences Centre. His area of focus includes home dialysis and quality improvement. He is a graduate of 2021 class of the Nephrology Social Media Collective (NSMC).
Competitors for the Hemodialysis Region
Intradialytic Hypotension vs Intradialytic Hypertension
Small Molecules vs Middle Molecules

Copyright: Narubas Bangpasert / Shutterstock
Intradialytic Hypotension vs Intradialytic Hypertension
Blood pressure (BP) management during hemodialysis treatments can have a significant impact on patient symptoms and clinical outcomes, with emerging observational data suggesting that suboptimal BP control during hemodialysis is associated with lower self-reported patient quality of life. In this matchup, we discuss risk factors for dialysis-associated hypotension and hypertension while highlighting some important preventative and management strategies for hypotension and hypertension among patients receiving hemodialysis treatment.
Intradialytic Hypotension
Hypotension during hemodialysis is common and can lead to significant complications for patients. Nephrologists are often faced with important questions such as, “When should I intervene?”, “How do I manage?”, and “What BP is too low?”. This scouting report summarizes the latest evidence to better prepare you for your next encounter with intradialytic hypotension.

Copyright: Robyn Mackenzie / Shutterstock
Intradialytic hypotension is a serious complication of hemodialysis treatment and may lead to vascular access thrombosis, suboptimal dialysis with volume overload, and increased mortality. Additionally, more frequent episodes of intradialytic hypotension are associated with brain white matter changes, reduced residual kidney function, and higher rates of peripheral arterial disease. In 2005, the KDOQI Guidelines defined intradialytic hypotension as a decrease in systolic blood pressure (SBP) ≥20 mm Hg or mean arterial pressure ≥10 mm Hg associated with symptoms (including cramping, headache, lightheadedness, vomiting, or chest pain) or the need for intervention, including reduction in ultrafiltration (UF) rate or administration of IV fluids.
A more recent analysis of data involving participants in the HEMO study revealed that intradialytic hypotension definitions that included symptoms, interventions, or reductions in BP during dialysis were not associated with mortality outcomes. However, nadir-based definitions demonstrated a better association between intradialytic hypotension and mortality. As a result, the 2019 KDIGO controversies conference on blood pressure and volume management in dialysis suggested, “any symptomatic decrease in SBP or a nadir in intradialytic SBP <90 mm Hg should prompt reassessment of BP and management.”

Visual Abstract by @whatsthegfr on Flythe et al.
The factors leading to intradialytic hypotension are multifactorial but primarily include insufficient refilling of the vascular compartment from the interstitial compartment in the presence of UF. Autonomic dysfunction along with an inadequate cardiovascular compensatory response in the setting of intravascular volume contraction also leads to intradialytic hypotension. High interdialytic weight gain is a risk factor, likely due to subsequent high UF rate requirements. This understanding has been supported by large retrospective data showing an increased incidence of intradialytic hypotension with both higher absolute and relative interdialytic weight gain.
Additionally, pre-dialysis osmolality may be a factor in BP stability; observational data has highlighted that episodes of intradialytic hypotension are associated with higher predialysis calculated osmolarity followed by rapid shifts of osmolality during dialysis sessions. Arginine vasopressin (AVP) impacts BP control in dialysis patients, and it is postulated that impaired AVP secretion can lead to intradialytic hypotension. In 2008, a small nonrandomized study revealed that very low volumes of hypertonic saline and hypertonic glucose solutions as well as a physiologic dose of AVP all raised BP and may in turn prevent intradialytic hypotension. Because the volume of the hypertonic solutions was insufficient to impact blood volume, this suggests that effects of these solutions are mediated by AVP.
Management
Several management strategies can be utilized for patients with intradialytic hypotension and are further categorized into non-pharmacological and pharmacological approaches. Non-pharmacological strategies may include reducing the UF rate and continually reassessing the target-weight for patients. Higher UF rates are associated with increased mortality risk along with end-organ ischemic events; however, there have been no large studies demonstrating that a lower UF rate in hemodialysis patients leads to better clinical outcomes. Additionally, most studies examining UF rate have been observational in nature, limited by confounders and lack of data on residual kidney function. As a result, there is no consensus on an ideal UF rate, and clinicians must consider individualized treatment depending on patient comorbid conditions while balancing the risk of volume overload.
Dialysis treatment modifications may include using higher dialysate sodium concentrations. Higher dialysate sodium likely impacts BP through several mechanisms, including maintaining intravascular volume and stimulating neurohormonal effects that are associated with vasoconstriction to decrease the likelihood of intradialytic hypotension. Higher dialysate sodium concentrations can be used in a fixed or profiled manner, whereby the dialysate concentration is gradually reduced to isotonic or even hypotonic levels as the dialysis treatment progresses.
This has the significant drawback that patients often end up with less sodium removal during dialysis and potentially even positive sodium balance, with resultant increased interdialytic weight gain due to increased thirst and fluid intake. A recent analysis of Dialysis Outcomes and Practice Patterns Study (DOPPS) data involving 10,250 patients from numerous international sites revealed that routine sodium profiling for intradialytic hypotension was associated with higher all-cause mortality [HR 1.36 (95% CI 1.14-1.63)]. As a result of these safety concerns and lack of strong evidence, sodium profiling should not be routinely used.
The risk of intradialytic hypotension can be reduced by lowering the dialysate temperature below the core body temperature. In fact, this is recommended as a first line prevention strategy by the European Best Practice Guidelines. The cooler dialysate leads to increased vasoconstriction and in turn activation of the sympathetic nervous system which increases BP during dialysis. This presumed mechanism is supported by a meta-analysis and systematic review that have shown a significant increase in intradialytic mean arterial pressure readings. The results of a recently completed, large, pragmatic Canadian clinical trial (MyTEMP) examining patient outcomes of personalized dialysate temperature may provide further insight on the effect of cooling dialysate on cardiovascular outcomes.
Practitioners may consider the use of pharmacologic interventions as a next step in conjunction with non-pharmacological approaches. The most commonly used pharmacological agent for dialysis-associated hypotension is midodrine. Midodrine is an 𝝰-1-adrenergic receptor agonist with a half-life of approximately 3 hours and is readily cleared with hemodialysis. Midodrine improves symptoms associated with intradialytic hypotension and increases nadir SBP by an average of 13.3 mm Hg (95% CI, 8.6 -18.0); however data on overall mortality and cardiovascular outcomes are lacking. Other agents that may be useful in the treatment of intradialytic hypotension include AVP, fludrocortisone, and sertraline, but strong evidence for their use is lacking.
Given the prevalence of intradialytic hypotension, future large randomized studies examining the effectiveness and overall patient outcomes of pharmacological and non-pharmacological approaches in treating intradialytic hypotension are needed. The prevalence of and huge clinical impact associated with intradialytic hypotension make it a worthy NephMadness contender.
COMMENTARY BY ETIENNE MACEDO:
Intradialytic Hypotension – The Bad and the Ugly
Intradialytic Hypertension
Patients with hypertension associated with hemodialysis are at an increased risk of negative outcomes. The pathophysiology is complex, multifactorial, and poorly understood, presenting numerous challenges in implementing effective and safe interventions. This report reviews the current evidence on the causes and management of intradialytic hypertension and why this competitor is poised to win NephMadness.

Copyright: SAJE / Shutterstock
The prevalence of hypertension during dialysis varies depending on the definition but ranges between 5% and 15%. The 2019 KDIGO controversies conference on blood pressure and volume management in dialysis suggested that intradialytic hypertension be defined as, “An SBP rise >10 mm Hg from pre- to post-dialysis in the hypertensive range in at least 4 of 6 consecutive dialysis treatments,” and noted that this, “should prompt a more extensive evaluation of BP and volume management, including home and/or ABPM.” This definition is important, as intradialytic hypertension may be an important predictor of interdialytic hypertension as well as overall cardiovascular outcomes.
The causes of intradialytic hypertension appear multifactorial and complex, but likely involve volume overload along with activation of both the sympathetic nervous system as well as the renin angiotensin aldosterone system. Additionally, small studies have shown that endothelial dysfunction and arterial stiffness may play a role in intradialytic hypertension. Lastly, the dialyzability of various antihypertensives, particularly angiotensin-converting enzyme (ACE) inhibitors and some 𝝱-blockers, could lead to increased incidence of intradialytic hypertension.
Volume overload as a contributory mechanism to intradialytic hypotension is supported by findings from a post hoc analysis of the Dry-weight reduction in hypertensive hemodialysis patients (DRIP) trial where 100 patients were randomized to intensive UF treatment for a period of 8 weeks, and 50 patients were assigned to a control group that did not undergo dry-weight probing. Patients in the UF group who underwent dry-weight probing had a more significant drop in the intradialytic BP compared to the control group. Each percent per hour steepening of the intradialytic BP was associated with a 0.71 mm Hg reduction [95% CI, 0.01-1.42, P = 0.05] in interdialytic ambulatory systolic BP. This study highlights that dry-weight probing may be highly effective in addressing intradialytic hypertension and may also improve BP during the interdialytic period.

Visual Abstract by @krishnadoctor1 on Agarwal et al
The activation of the renin-angiotensin system in the setting of intravascular volume contraction due to rapid UF can also lead to intradialytic hypertension. An older study, albeit small, showed that 50 mg of captopril given to patients prior to hemodialysis treatment resulted in better BP control with reduced episodes of intradialytic and post-dialytic hypertension. Additionally, sympathetic nervous system overactivity may also play a significant role in the underlying pathophysiology of intradialytic hypertension. Sympathetic overactivity has been documented, with a microneurographic study demonstrating that sympathetic-nerve discharging was 2.5 times higher among hemodialysis patients who had not undergone nephrectomy as compared to healthy individuals (58±3 vs. 23±3 bursts per minute, P < 0.01). The rate of sympathetic-nerve discharge was similar in the patients receiving hemodialysis who had undergone bilateral nephrectomy to the health controls. It is for this reason that bilateral nephrectomy is considered a significant risk factor for hypotension in patients receiving hemodialysis owing to significant reduction in sympathetic nerve discharges.
Management
Although the management of intradialytic hypertension should be individualized according to patient needs, an initial approach must prioritize adequate volume control. However, the achievement of adequate volume control is commonly limited by the use of antihypertensive medications in the interdialytic period. Patients may develop significant hypotension with minimal UF, especially if the antihypertensive of choice is non-dialyzable. In some instances, it may be reasonable to down-titrate antihypertensive therapy to allow for better UF and subsequently more adequate volume control.
Increasing the frequency and duration of hemodialysis treatments may also afford a better opportunity to safely achieve better volume control. The findings from the Frequency Hemodialysis Network (FHN) trial demonstrated that short-daily hemodialysis sessions achieved better BP control compared to thrice-weekly hemodialysis sessions. It should be noted that this study was not designed to evaluate the effect of different modalities on intradialytic hypertension, but its findings have presented practitioners with an alternative strategy that can be used in this population.
One potential practice to manage intradialytic hypertension in dialysis units is to provide captopril at the beginning of therapy. This practice stems from the results of a very small non-randomized study that demonstrated a blunted rise in BP among patients who received captopril at the start of hemodialysis treatment. However, the majority of ACE inhibitors are dialyzable (a notable exception is fosinopril), with varying dialyzability that further complicates their selection in the management of intradialytic hypertension.
Additionally, a review of data from a large healthcare database failed to demonstrate an association of the dialyzability of ACE inhibitors with mortality or cardiovascular outcomes. As a result, the routine use of ACE inhibitors as well as angiotensin receptor blockades specifically in the management of intradialytic hypertension is plagued by inconsistent data that fail to demonstrate effectiveness or improved outcomes.
As mentioned earlier, UF and subsequent intravascular volume contraction may lead to activation of the sympathetic nervous system. This effect can be blunted with the use of long-acting agents with both 𝝰- and 𝞫-blocking properties. Additionally in 2009, a systematic review of randomized controlled trials concluded that 𝞫-blockers may significantly reduce cardiovascular morbidity and mortality among patients receiving hemodialysis.
Furthermore, carvedilol, a 𝞫-blocker with vasodilatory properties, has shown promising results in enhancing endothelial dependent flow which was associated with reduced episodes of intradialytic hypertension. Another possible agent is minoxidil, a potent vasodilator. Although minoxidil may be effective in treating elevated BP during dialysis, it should be used with extreme caution given potential side effects, including a high risk of pericardial effusion.
Reducing the intradialytic sodium load is another preventative strategy that can be used in dealing with intradialytic hypertension. A randomized study showed that patients treated with a sodium dialysate concentration of 5 mEq/L lower than their serum sodium had a significant reduction in intradialytic SBP compared to those with dialysate concentration of 5 mEq/L greater than serum sodium. However the use of lower sodium dialysate concentrations, particularly amongst those with predialysis hyponatremia (<130 mEq/L), may result in problems with conductivity on current commercially available dialysis machines.
In conclusion, intradialytic hypertension is not uncommon among patients receiving hemodialysis treatment and is associated with significant cardiovascular morbidity and mortality risk. Current practice should prioritize reducing volume overload and excessive sodium loads, which are often major contributing factors to hypertension during dialysis. Practitioners should avoid using a blanket approach with various antihypertensives and treatment must be individualized in a manner that carefully balances the benefits and risks for each patient.
Check out this podcast episode of Up My Nursing Game featuring Bourne Auguste @bourneauguste and Daniel Weiner @DanTheKidneyMan:
Blood Pressure and Dialysis
Small Molecules vs Middle Molecules
Solute clearance
Uremia, meaning “urine in the bloodstream”, describes the constellation of clinical symptoms and metabolic abnormalities that result from impaired kidney function. This impairment in kidney function leads to the “uremic syndrome” by virtue of retained uremic solutes. Dialysis treatments reduce the overall uremic solute concentration in a non-specific manner. Although there are numerous solutes that contribute to the uremic syndrome, hemodialysis adequacy is currently measured solely on the basis of one solute, urea nitrogen. Uremic toxins include small non-protein bound molecules such as urea, small and larger non-protein bound middle molecules such as ß2 microglobulin, and protein-bound solutes such as phenols. We will discuss the two main categories of uremic solutes, small and middle molecule solutes, along with current evidence and emerging strategies that can optimize their clearance with hemodialysis and hemodialysis-related modalities.
Small Molecules
While there has been an emphasis on urea as a surrogate marker for small solute clearance, there are other solutes that are cleared by the kidney but are not effectively cleared with conventional hemodialysis treatments that may also contribute to the uremic syndrome. How do we identify other small solutes that lead to uremia? Can the removal of other non-urea small solutes improve patient outcomes?

Copyright: vandycan / Shutterstock
The first commercially available dialyzers were all low-flux dialyzers, removing water-soluble non-protein bound small solutes, like urea. The effect of urea on patient outcomes was first examined by the National Cooperative Dialysis Study (NCDS) in 1981. The NCDS was a randomized controlled trial that examined 151 patients receiving dialysis treatment to determine if hemodialysis duration (short vs long) and target serum urea levels (high vs low) would be associated with time to first hospitalization. Although the study was small and not powered to detect mortality differences between high and low urea levels, higher time averaged urea nitrogen clearance was associated with a lower hospitalization risk, a finding that directly led to urea clearance targets (Kt/V urea) that are still used today in practice, as well as an emphasis on dialysis intensity over dialysis duration. Based in part on these data, the Kidney Disease Outcomes Quality Initiative (KDOQI) recommended a single pool Kt/Vurea target of 1.2 as a minimum standard for per session hemodialysis adequacy for patients treated thrice weekly. However, more recent trials, including the HEMO study, did not show any mortality benefit with increased urea clearance.
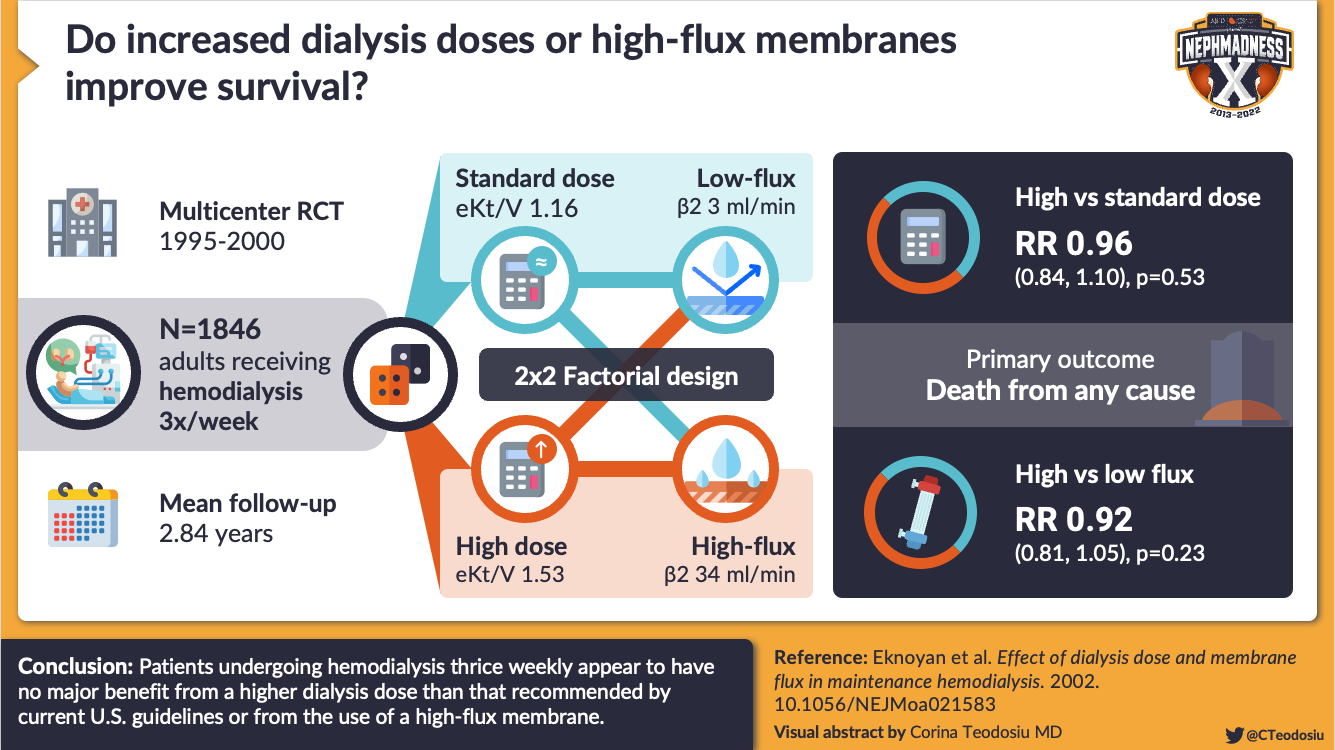
Visual Abstract by @CTeodosiu on Eknoyan et al.
Small solutes that are bound to plasma proteins such as phenols are poorly cleared with conventional hemodialysis. For example, p-cresol, a phenol, is poorly dialyzed despite having a molecular weight of only 108 Da since it is lipophilic and highly bound to proteins. Observational data has shown that p-cresol serum concentrations are strongly associated with uremic symptoms, hospitalizations, and overall mortality. Additionally, a meta-analysis examining another phenol, p-cresyl sulfate, demonstrated that elevated levels were associated with increased mortality in patients with advanced kidney disease.
However, limited studies have examined methods to enhance the plasma clearance of these protein-bound uremic solutes. There have been small observational studies that have demonstrated superior clearance with convective dialysis strategies compared to diffusive strategies. In a study that included 9 patients receiving thrice-weekly nocturnal dialysis, findings revealed that increasing the dialyzer size and dialysate flow rate had a significant reduction in protein-bound solutes without affecting the spKt/Vurea.
Another potential strategy to enhance clearance of these protein-bound solutes is to increase the free-fractional concentration that is filtered across the dialyzer membrane. A recent review summarized various approaches to increase free-fractional plasma concentrations by inducing conformational changes in protein structure. These include altering the plasma pH, increasing the plasma tonicity, and creating bioelectrical fields across the dialyzer. However, a large degree of uncertainty remains regarding safety and widespread adoption of these emerging strategies.
Finally, hemoperfusion may offer an alternative method to enhance clearance of protein-bound uremic solutes via adsorption. Hemoperfusion entails the direct passage of blood through cartridges that contain either an anion exchange resin or activated charcoal. It has been primarily used in the removal of poisons, but has major disadvantages in that cartridges have a short shelf life, are more expensive than conventional dialyzers, and may lead to bleeding in patients as a result of thrombocytopenia from cartridge induced platelet adsorption.
Despite the drawbacks of hemoperfusion and sorbent technology, current knowledge must be maximally leveraged to identify novel methods to bind specific solutes with limited complications at an affordable cost. Therefore, future studies are desperately needed to identify various solutes that contribute to the uremic syndrome. The identification of these solutes would allow for a more deliberate and targeted approach in adopting treatment strategies that enhance clearance of a broader range of culprit toxins. Unfortunately, a urea-centric approach has oversimplified the complexity of uremia down to once-a-month single pool Kt/Vurea clearance targets. The irony in all this is that our current approaches to target “adequacy” remain inadequate.
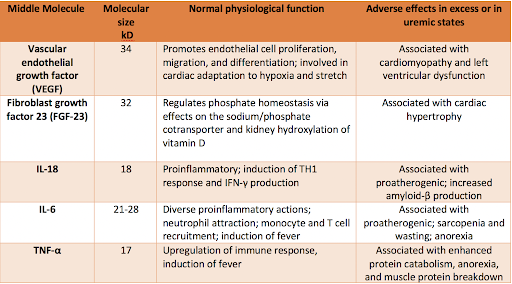
Various biological functions of larger middle molecules and related adverse effects in excess or in uremic states. Adapted from Table 2 in Wolley et al.
Middle Molecules
Beta 2 microglobulin is an important surrogate marker of middle molecular clearance. Should we specifically target this and other important middle molecules? Are there any clinical benefits with enhanced middle molecular clearance using high-flux dialyzers or higher cutoff dialyzers?

Copyright: vandycan / Shutterstock
During the early 1960s, hemodialysis was performed using low-efficiency dialysis membranes, providing suboptimal clearance of solutes with molecular weights in excess of 1,000 Da. Early experiences showed that patients receiving peritoneal dialysis had better improvement in uremic symptoms compared to hemodialysis. These observations by Babb and colleagues led to the development of the middle molecule hypothesis, which originally stated that molecules with a molecular weight between 500 to 2,000 Da played a significant role in the development of uremia and peripheral neuropathy.
Unfortunately, this hypothesis was never tested with randomized nor prospective trials. Middle molecules span a range of molecular weights and include proinflammatory cytokines, β2 microglobulin, complement components and free light chains. ꞵ2 microglobulin has a molecular weight of 12,000 Da and has been adopted as the surrogate marker of smaller middle molecule clearance, much like the role that urea has played as a marker of small solute clearance.
Despite the establishment of spKt/Vurea clearance targets, patients receiving hemodialysis treatment developed complications including dialysis-related arthropathy such as carpal tunnel syndrome secondary to ꞵ2 microglobulin accumulation. The accumulation of ꞵ2 microglobulin was also been associated with systemic amyloidosis in patients receiving dialysis, usually after many years of therapy. High-flux dialyzers are dialysis membranes with larger pores than low-flux dialyzers, and have replaced low-flux dialyzers worldwide. Despite enhanced clearance of ꞵ2 microglobulin, high-flux dialyzers failed to demonstrate better survival outcomes when compared to lower-flux dialyzers in the seminal HEMO trial.
Whatever the impact of improved dialysate, more frequent and longer treatments coupled with higher flux dialyzers on the incidence of dialysis-related amyloidosis should not be overlooked. Prospective post-mortem data have shown that some patients may develop systemic amyloidosis within 2 years of beginning dialysis treatment. Over the period of a decade, data from a large European center revealed an 80% decline in the incidence of amyloidosis. The authors highlighted that this decrease was likely attributable to improvements in dialysate composition and possibly the development of more biocompatible hemodialyzers. Therefore, the overall reduced rates of amyloidosis seen amongst patients receiving hemodialysis treatment is more likely representative of broader advancements in the quality of dialysis treatment, and not necessarily due to dialyzer flux capacity.

Differences between various dialysis membranes relative to pore size and number of pores, where the pink bar represents the area of large middle molecules just before albumin. Adapted from Wolley et al.
The removal of other, larger middle molecules such as free light chains (FLCs), both as proof of concept markers of the feasibility of enhanced larger middle molecule clearance as well as in exploring treatments for myeloma, has also been explored. Current high-flux dialyzers do not have pores large enough to allow for the significant clearance of Kappa (𝞳) (~ 22.5 kilodaltons) and lambda (𝛌) (45 kilodaltons) FLCs. Recent studies have explored the safety and efficacy of using high cut-off (HCO) membranes and high retention onset membranes for clearing larger middle molecules.
There is a modest amount of data exploring the role of HCO membranes in treating light chain associated diseases. High cut-off hemodialysis membranes have much larger pores than high-flux dialyzers, allowing middle molecules up to 50 kilodaltons in size to be removed. Given that nearly half of all patients with multiple myeloma develop kidney disease as a result of cast nephropathy, effective strategies to reduce the burden of serum FLCs have been studied.
The MYRE trial randomized 98 patients in France with biopsy proven myeloma cast nephropathy to receive intensive hemodialysis with either a high-cutoff dialyzer or a high-flux dialyzer. In patients who received bortezomib-based chemotherapy, there was no difference in hemodialysis independence between high-cutoff and high-flux dialysis treatment at 3 months (41% vs 33%, P = 0.42), although at 6 months the rate of hemodialysis independence was greater in patients treated with high-cutoff dialysis compared to conventional hemodialysis (57% vs. 35%, P = 0.04). Most importantly, there was no observed mortality difference between the two groups at 12 months.
Similar findings were seen in the more recently published EuLITE phase 2 trial, with no clinical benefit of high-cutoff hemodialysis over conventional hemodialysis for patients with myeloma cast nephropathy. These findings, along with high associated cost, limited commercial availability, and increased risk of hypoalbuminemia continue to favor the use of high-flux dialyzers in myeloma.
Alternatively, the removal of middle molecules can be enhanced by generating greater hydrostatic pressure differences across the dialyzer through convective transport, resulting in hemofiltration. When combining hemofiltration with the diffusive transport of hemodialysis, hemodiafiltration (HDF) is achieved. HDF provides better clearance of small and middle uremic solutes when compared to hemodialysis or hemofiltration alone, and a Cochrane review of 35 studies with 4,039 patients comparing convective therapies to hemodialysis showed that the former may reduce cardiovascular events but not all-cause mortality. This cardiovascular benefit may be explained by reduced risk of intradialytic hypotension with convective therapies. However, strong conclusions about this benefit should be taken with caution given the limitations associated with study designs and methodologies.
More contemporary data have also not shown any significant benefit with hemofiltration in reducing neuropathy. A recent study involving 124 patients randomized to HDF and hemodialysis showed no significant difference in mean modified total neuropathy scores (95% CI, -0.7 to 1.7, P = 0.37). Critically, HDF, including online HDF, is more expensive than hemodialysis. Widely used in Europe, HDF has not been done widely in North America for maintenance dialysis, likely in part due to higher costs and equipment demands.
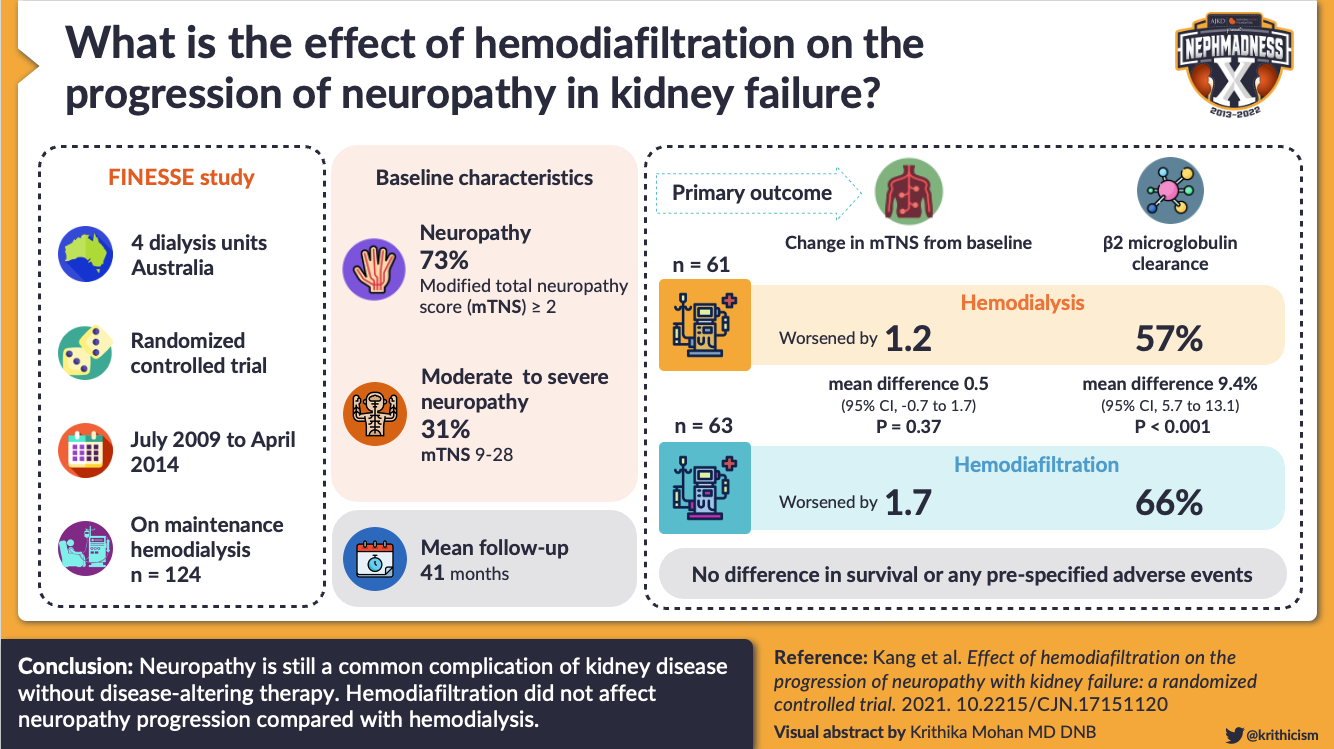
Visual Abstract by @krithicism on Kang et al.
Newer dialysis membranes incorporate enhanced convective transport within an HCO membrane, resulting in greater middle molecule clearance without significant changes in serum albumin levels. Whether this newer technology, which costs somewhat more than current high-flux membranes, will be associated with improved clinical outcomes remains unknown, and, in the United States at least, no additional money has been added to the dialysis payment bundle to fund the use of these membranes.
In summary, the accumulation of middle molecules plays an important role in contributing to the overall uremic syndrome. Future research must consider other middle molecules beyond ꞵ2 microglobulin to determine if their targeted clearance may improve overall outcomes for patients receiving dialysis treatment.
– Executive Team Members for this region: Pascale Khairallah @Khairallah_P and Joel Topf @kidney_boy
How to Claim CME and MOC
US-based physicians can earn 1.0 CME credit and 1.0 MOC per region through NKF PERC (detailed instructions here). The CME and MOC activity will expire on June 1, 2022.
Click to read more NephMadness 2022 Regions


Wanted to clarify the middle molecule size in the post. Otherwise excellent learning.