#NephMadness 2021: Liquid Biopsy Region
Submit your picks! | NephMadness 2021 | #NephMadness | #LiquidBiopsy
Selection Committee Member: Florian Buchkremer @swissnephro
Florian Buchkremer is a Consultant Nephrologist at the Kantonsspital Aarau in Switzerland. He enjoys the incredible diversity of nephrology and its committed social media community. Special interests include electrolyte disorders, ultrasound, and the urinary sediment.
Writer: Caitlyn Vlasschaert @DrFlashHeart
Caitlyn Vlasschaert is an internal medicine resident and PhD student at Queen’s University in Canada. Her research is focused on somatic myeloid cell mutations in kidney disease. She is a graduate of the 2020 class of the Nephrology Social Media Collective (NSMC) Internship and now sits on its executive committee.
Competitors for the Liquid Biopsy Region
Microscopy Techniques vs Staining Techniques
Urine Microscopy for GN vs Urine Microscopy for ATN

Copyright: plenoy m / Shutterstock
A Brief History of the Pisse Prophet
Urinalysis is one of the oldest diagnostic tools in medicine – could good ol’ urine really be a serious contender in this year’s Big Dance? For centuries, health prophecies centered on this most-accessible bodily fluid. Careful study of its properties—from appearance to smell to taste—was called uroscopy and could yield clues to underlying disease processes. This methodology gave us the qualifiers “mellitus” (honey-sweet) and “insipidus” (flavorless), referring to the taste of urine in these polyuric (or “diabetic”) disorders. Let’s review this region’s sweet history before introducing this year’s teams.
The perceived value of uroscopy rose steadily as its scope broadened from its inception (predating Hippocrates) to the Middle Ages. Born from a need to organize the ever-expanding field, the Wheel of Urine (figure below) painted a multisensory profile of urine in its various flavors.

Urine wheel from Epiphanie medicorum by Ulrich Pinder, 1506. From Folger Shakespeare Library, released under CC BY-SA 4.0.
At the turn of the 13th century, Gilles de Corbeil wrote a lyrical poem detailing the many associations of urine called De Urinis, which medical students memorized for centuries to follow.

English translation of De Urinis, from the book Medieval Medicine.
De Corbeil also invented the matula, a special flask used to observe urine that rapidly became a symbol of professional prestige:
“Physicians would dress in long robes, and would make a ritual of holding the matula to the light and rotating it before confidently disclosing the patient’s fate. This impressed the patient and instilled assurance in the physician’s intelligence.”
Subsequently, uromancy, the popular art of using urine for fortune-telling, emerged. Physicians, as well as many with no medical training, posed as great divinators with their all-seeing matulas. In response, in 1637 Thomas Brian published his book Pisse Prophet, a brash critique of the ridiculous devolution of uroscopy.
Urinalysis: A New Age
The modern nephrologist and technician, like the pisse-monger of old, still cradle vials of the amber liquid in their hands as they divine its secrets. Only the microscope, a few chemicals, and the urinary dipstick have intruded to modify the art.
— K.D. Gardner Jr., 1971, NEJM
Though our predecessors got carried away with its implementation, the core tenet of uroscopy was correct; analyzing the urine is a critical part of the workup of several conditions. Nowadays, urinalysis generally entails urine dipstick and microscopy. But with the advent of automated technologies and centralized testing, and the associated decline in formal microscopy training, is urine microscopy on its deathbed?
This region opposes that sentiment and is poised to be this year’s dark horse. Its four teams are well-seasoned, with more experience than the other regions combined. It’s time for us to reclaim our status as urinalysis experts, masters of the liquid biopsy, bona fide modern-day pisse prophets. So grab a urine sample, drop it in the centrifuge, and let’s play ball!
Microscopy Techniques vs Staining Techniques
Microscopy Techniques
“The finest wits have their sediment.”
– Ralph Waldo Emerson

Copyright: Konstantin Kolosov / Shutterstock
Who says nephrology doesn’t have procedures? Urine microscopy technique can be the nephrologist’s “Force” when it comes to looking for etiologies of kidney diseases (non-invasively, of course). To wield this power, knowledge of the basics is key, including effective application of light microscopy, phase contrast, and the polarizer. Without these techniques, you might be left on the Dark Side.
Urine sediment may contain a wealth of information. Cells, casts, lipids, crystals, and microorganisms in the urine can serve as real-time biomarkers of underlying pathology. So how does one take it all in? Though time- and labor-intensive, manual microscopic examination of the sediment by nephrologists often outperforms hospital laboratory and automated analyses, especially when clinical context is applied as an additional lens with which to interpret live findings within a systematic framework. A far cry from the multisensory experience of uroscopy, urine microscopy nonetheless uses a variety of “tasty” techniques. In the nephrologist’s battle against kidney disease, the urine sediment might just provide the fastest and simplest scouting report on the opponent. Will back-to-the-basics squad Microscopy Techniques survive in this year’s tournament against the stunning, glamorous images from team Staining Technique?
Light microscopy
The superpower of this team? Manipulation of light. The original and simplest microscope design, light microscopes illuminate specimens from below to highlight their magnified properties. Bright-field, phase-contrast, and polarized light microscopy are variants of light microscopy used in urine microscopy.

Outline of how to perform urine sediment analysis using light microscopy, adapted from Cavanaugh and Perazella, AJKD © National Kidney Foundation.
Bright-field is typically the default microscope setting that provides a useful first look at the composition of the urine sediment. The steps for performing this technique are simple and include examining a centrifuged urine sample at low and high power (see figure above). Additional microscope techniques can then help confirm suspected findings.
- Muddy brown granular cast
- Red cell rouleaux
- Mixed cellular cast containing calcium oxalate stone
An assortment of unstained bright-field images: muddy brown granular cast (Juan Carlos Velez), red cell rouleaux (José Poloni), mixed cellular cast containing calcium oxalate stone (Jay Seltzer). Used with permission.
Developed by Frits Zernicke in the 1930s (and culminating in a Nobel Prize in 1953) the phase-contrast microscope is a powerful tool for urine microscopy. Whereas bright-field microscopy relies on differences in transparency to visualize a specimen, phase contrast transforms the path of light traveling through an object to enhance its contours.

Crisp definition of acanthocyte edges, blebs and all, by phase contrast microscopy. Image by Florian Buchkremer. Used with permission.
This can be particularly helpful for identifying glomerular hematuria (see figure below), for example, or for seeing sediment elements that may be missed by other microscopy techniques, such as the lowly refractive hyaline casts. In fact, phase-contrast microscopy is recommended by European guidelines, and is thought to make staining completely dispensable.
Polarized light microscopy makes use of a polarized filter to view sediment components with birefringence. Turning on the polarizer is like putting polarized sunglasses on your microscope. Instead of helping to reduce the amount of light reflecting in your eyes on a sunny day, these sunglasses allow you to pick out objects that are normally hard to see, like crystals and lipids. For example, cholesterol esters appear as maltese crosses under the polarizer (see figure below).

Lipid cast in a patient with nephrotic proteinuria by phase contrast (left panel), bright field (upper right) and polarized light (lower right) microscopy. Image by Florian Buchkremer. Used with permission.
Urine microscopy technique is critical to accurate urine sediment interpretation. A study of nephrologists found significant interobserver variability in the interpretation of urine sediment findings, even among expert nephrologists! Luckily there is interest, even among internal medicine residents, to learn urine microscopy techniques and achieve #PisseProphet status.

Visual abstract by Edgar Lerma on Palsson et al.
Staining Techniques
“Examination of the urine of all patients has been routine for centuries. Since now, however, the microscope is so universally used, staining urinary deposits should become routine in clinical medicine.”
— Edward Sandwith Johnson, 1914, Dublin Journal of Medical Science

Copyright: HAKKI ARSLAN / Shutterstock
Sometimes you just need a pop of color. Both innate and exogenous stains can allow the urine microscopist to better identify structures and improve resolution. And, of course, these stains can turn otherwise colorless cells and casts into dazzling works of art for your home, office, or bathroom.
While the microscope as a standalone is a powerful tool, even better visualization may be achieved with staining. Stains can help classify cells in the sediment and determine the composition of casts, especially when enhanced modalities like phase contrast are not available.
The Classic: Sternheimer-Malbin (SM) Stain
The Sternheimer-Malbin (SM) stain is most widely used for urine sediment analysis. Developed in 1948 by Richard Sternheimer and Barney Malbin, it is composed of crystal violet and safranin, similar to the Gram stain. While common staining techniques like the Wright method and its derivatives (commonly used for blood films) require serial stain-and-wash steps to be performed on air-dried samples, the SM stain is supravital, meaning it is applied to live sediment, fresh out of centrifuge. The steps for using the Sternheimer-Malbin stain are simple: First, split the urine sediment during sample preparation. Next, apply the SM stain to one slide (use 1 drop, mix gently and allow 1-2 minutes for uptake). Finally, assess under bright-field illumination.
- Unstained mixed cellular casts
- Stained mixed cellular casts
Unstained and stained mixed cellular casts by Jay Seltzer. Used with permission.
The SM stain helps to distinguish urinary red blood cells (RBCs), white blood cells (WBCs), tubular epithelial cells, and acellular casts components by improving resolution and thus identification of structures. For the hematologic cells, SM stain uptake can tell us about their age. Younger or more viable urine granulocytes with visibly active cytoplasmic granules tend to be more pale-staining than their senescent counterparts; they are actually referred to as “Sternheimer-Malbin cells” or glitter cells. The age of RBC casts can also be inferred. The acellular cast material—whether it is hyaline, granular or waxy in nature—can also be better visualized with the SM stain.
- Nature’s powerful pigments. The left panel shows bilirubin coloring renal tubular epithelial cells (image from a RFN post by José Poloni). The right panel shows free hemoglobin coloring the supernatant after centrifugation (image courtesy of Samira Farouk). Used with permission.
- Nature’s powerful pigments. The left panel shows bilirubin coloring renal tubular epithelial cells (image from a RFN post by José Poloni). The right panel shows free hemoglobin coloring the supernatant after centrifugation (image courtesy of Samira Farouk). Used with permission.
SM-stained mixed cell cast under bright-field illumination. Polarized light identifies an oval fat body within a WBC in the right panel. Images by Jay Seltzer. Used with permission.
The Outdated: Staining for eosinophils in urine
Certain stains can be helpful in identifying specific cell types in urine. The eosin-containing Wright stain was first used to identify urine eosinophils, which were considered an accurate biomarker of acute interstitial nephritis at the time. The Wright stain was soon replaced by the more sensitive Hansel stain, which colors eosinophilic granules bright orange instead of ‘eosin pink’.
However, the utility of eosinophiluria in acute interstitial nephritis has since come into question due to its lack of specificity in this context, and searching for eosinophils is not currently recommended. But every team is only as strong as at its weakest link, right?
The Innate: Pigmented casts
Some sediment components are “born” pre-stained. Endogenous pigments, including hemoglobin, myoglobin, and bilirubin, can color urinary casts and yield clues to underlying systemic pathology. These natural stains can, however, be overpowering. Bilirubin will stain the entire sediment yellow (see figure below), which sometimes makes it difficult to distinguish between cast types.
- Bilirubin coloring renal tubular epithelial cells
- Free hemoglobin coloring the supernatant after centrifugation
Nature’s powerful pigments. The left panel shows bilirubin coloring renal tubular epithelial cells (image from a RFN post by José Poloni). The right panel shows free hemoglobin coloring the supernatant after centrifugation (image courtesy of Samira Farouk). Used with permission.
The Detective Partner: Sudan stain for lipiduria
Finally, the Sudan stain is used to detect fat globules in urine and other bodily by-products (ie feces). Whereas the polarizer can only reliably detect cholesterol esters (see lipid cast figure above in the Microscopy Techniques section), Sudan stains are thought to identify a broader variety of lipids, including triglycerides and lipoproteins. When on the hunt for lipid clues, the polarizer and Sudan stains are like Sherlock Holmes and Dr. Watson. But who is the lead detective: stains or ‘scopes? That’s for you to decide as you cast your vote for the winner of this region.
The game is afoot.
— Arthur Conan Doyle
Urine Microscopy for GN vs Urine Microscopy for ATN
Urine Microscopy for Glomerulonephritis (GN)

Copyright: Piboon Suwankosai / Shutterstock
Regularly discarded urine might hold more answers than you think, even when you’re not expecting it. When urine microscopy reveals elements like acanthocytes or red blood cell casts, you could be on your way to clinching a diagnosis of glomerulonephritis. If you don’t look, how do you know what’s there?
RBCs on urinalysis signal that there may be a lesion in the urinary tract, anywhere from the glomerulus to the urethra. The strength of this team? Its game plan to help identify the source. Depending on whether a nephrogenic or urologic source of bleeding is suspected, different arms of investigation may be initiated. Often, invasive procedures are involved (ie a kidney biopsy or a cystoscopy). Erythrocyte morphology can inform our pretest probability regarding the source of bleeding, as some bear battle scars chronicling their journey. Team Urine Microscopy for Glomerulonephritis (GN) is all about wise, careful diagnostic strategy planning. Why perform an invasive urologic procedure that might be avoided? Why aggressively charge to the hoop and try to draw a foul, when you can step back and sink a three-pointer? The three-pointer in this case is… uncovering acanthocytes, of course.
Acanthocytes, a subset of dysmorphic RBCs, are the hallmark of glomerular bleeding. Recall that phase-contrast microscopy is especially helpful in detecting acanthocytes—RBCs that develop arm-like projections (or “blebs”) after passing through the glomerular basement membrane (GBM) and then being exposed to the osmotic challenges of the tubular system (see figure below). Glomerular bleeding with acanthocyturia commonly occurs in various glomerulonephritides, but can also be seen with inherited disorders of the GBM like Alport syndrome and in non-GN syndromes like membranous nephropathy and diabetic nephropathy.
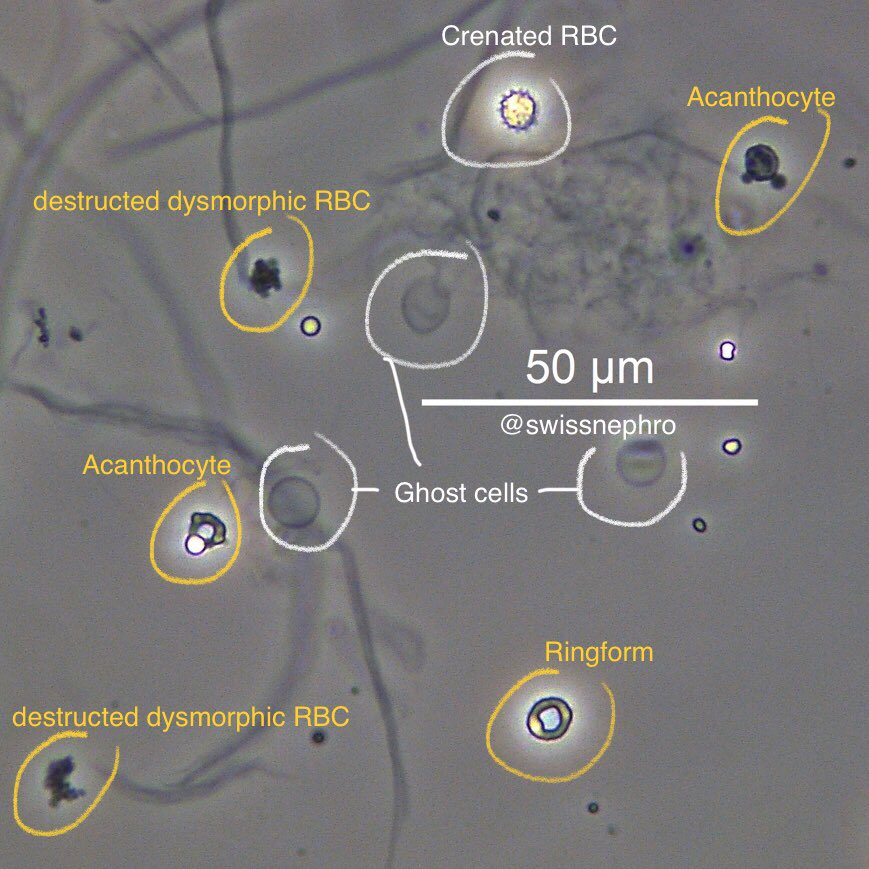
Different RBC morphologies, dysmorphic (yellow) and not (white), in a patient with glomerular hematuria by phase contrast microscopy. Crenated RBCs (or echinocytes) are a common technical artifact and are not hallmarks of glomerular bleeding. Image by Florian Buchkremer. Used with permission.
Red cell casts, formed in the distal nephron, indicate intraluminal bleeding that has occurred somewhere upstream—at the level of the glomerulus or in the tubules (see figure below). Though commonly considered a hallmark of GN, red cell casts can occasionally be found in acute interstitial nephritis as well. In theory, all RBCs (or any cell, for that matter) passing through the distal nephron can become glued together by precipitated Tamm-Horsfall Protein (THP, aka uromodulin) and form a cast. Any blood entering the urine beyond the collecting ducts typically does not change shape; RBCs from a urologic source are characteristically isomorphic and homogenous.
Left panel: SM-stained RBC cast under bright-field microscopy. Image by Jay Seltzer. Used with permission. Right panel: schematic showing that all urinary casts are formed in the distal nephron. Created with BioRender.com.
How accurately does RBC shape predict GN? In 1982, Fairley and Birch reported that the “indisputable” presence of dysmorphic RBCs by phase contrast microscopy was 100% specific and 95% sensitive for a diagnosis of glomerulonephritis (GN) in their 88-person cohort. Acanthocyturia, defined as acanthocytes constituting ≥ 5% of urinary RBCs, has since been confirmed to be about 95% specific for a glomerular disorder. However, finding an abundance of dysmorphic RBCs in GN isn’t as common as initially touted; the sensitivity of this benchmark is as low as 15–20%. GN can yield isomorphic urine RBCs or even present without hematuria. So, as dysmorphic RBCs (especially acanthocytes) are fairly specific but with poor sensitivity, we should not use them as the sole screening tool for GN. Interestingly, detecting persistent hematuria (via light microscopy) in established GN is linked to worse outcomes, including increased risk of relapse in ANCA vasculitis and higher risk of progression to ESKD in IgA nephropathy.
Urine Microscopy for Acute Tubular Necrosis (ATN)
“One damaged kidney’s trash is the shrewd clinician’s treasure.”
— Anonymous (2021)

Copyright: Vladimir Konstantinov / Shutterstock
To give fluid or not to give fluid, that is the question. Urine microscopy findings—specifically those dark, pigmented casts—might shed some light on this existential nephrologist’s question. If a peek into the microscope reveals a sea of muddy brown waters, look no further for the diagnosis.
Acute tubular necrosis (ATN) is the most common cause of acute kidney injury in hospitalized patients. Kidney ischemia or toxic insults precipitate tubular injury in ATN, and when severe, these lead to hypoxic parenchymal cell death (necroptosis). In such cases, the kidney neatly discards sloughed renal tubular epithelial cells (RTECs) and various casts in the urine. RTECs form the lumen of all nephron segments, and therefore can take on many shapes in the urine. They can be difficult to distinguish from other cell types, including urothelial cells. They generally have large, unsegmented cell nuclei and may contain yellowish-brown lipofuscin granules.
- RTECs by phase contrast microscopy
- RTECs by bright field microscopy
RTECs by phase contrast (left) and brightfield (right) microscopy, by Florian Buchkremer. Used with permission.
Cellular and acellular casts are identifiable in ATN. Similar to red cell casts, granular casts are the product of cellular debris and plasma proteins trapped in a THP matrix within the distal nephron. In general, the darker and coarser the cast, the more likely that it represents ATN. The “muddy brown cast” is a type of granular cast with dark pigmentation (possibly from denatured RTEC lipofuscin) and is usually associated with ATN. RTECs can become trapped in these casts, sometimes with other cell types such as WBCs.
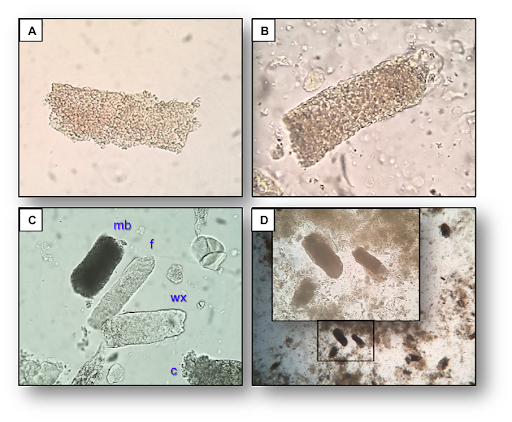
A. Finely granular cast B. Slightly coarser granular cast C. “Urine poker quads” showing a fine (f), coarse (c), and muddy brown (mb) granular cast accompanied by a waxy (wx) cast D. “Muddiness” of mb granular casts is better appreciated at low power field compared to high power field (inset). From the RFN post on granular casts by Juan Carlos Velez. Used with permission.
If you find this aspect of urine microscopy, you’re not alone; correct identification of casts is consistently difficult for automated urine analyzers, too. It is worth the effort however. A urinary sediment scoring system for AKI was developed in 2008 by Perazella et al. that very accurately predicted the likelihood of a final diagnosis of ATN using the pretest probability and the number of RTECs and granular casts on initial microscopy (see Visual Abstract below). When ATN was the leading diagnosis based on clinical information pre-microscopy, the detection of even a single granular cast or RTEC in the urine unequivocally confirmed the diagnosis.

Visual abstract by Priti Meena on Perazella et al.
Sediment findings not only help to accurately distinguish ATN from prerenal AKI, but can also provide an early prediction of the severity of kidney injury to help guide supportive treatment, including fluid management and planning for acute renal replacement therapy.

Cellular and molecular biomarkers by site of kidney injury. Figure 2 from Perazella, AJKD © National Kidney Foundation.
Best Urine Microscopy Image contest: Calling all medical students, residents, and fellows! Do you love to #SpinUrine? Share your original urine microscopy photo(s) on Twitter or Instagram with the hashtag #LiquidBiopsy AND #NephMadness by April 9, 2021. The #PisseProphets comprising Florian Buchkremer, Jay Seltzer, Jose Tesser Poloni, and Juan Carlos Velez will judge and determine the winner based on image quality and an accompanying post identifying the element in the image. No protected health information (PHI) should be shared.
Now comes the time to cast your vote! Which will reign supreme: muddy brown, or RBC?
Additional Resources on Microscopy
There are a growing number of published and #FOAMed resources available for clinicians seeking to refine their urine microscopy skills:
- Renal Fellow Network: Urine Sediment of the Month
- Twitter: #urinemicroscopy, #urinesediment, #urinalysis, #spinurine
- Textbooks
- The urinary sediment: an integrated view by G. Fogazzi
- A color atlas of urine microscopy by D. Birch, K. Fairley, P. Kincaid-Smith, G. Becker
- Fundamentals of Urine and Body Fluid Analysis (e-book) by N. Brunzel
– Executive Team Member for this region: Samira Farouk, AJKD Editorial Board Member. Follow her @ssfarouk.
How to Claim CME and MOC
US-based physicians can earn 1.0 CME credit and 1.0 MOC point for reading this region.
- Register/log in to the NKF’s Professional Education Resource Center (PERC). If you select “Physician” in the drop-down menu during registration, the ABIM ID will pop up – make sure to complete this during registration to receive MOC points after course completion.
- Review the activity, disclosure, and accreditation information.
- Click “Continue” and review Course Instructions.
- Complete Post-Test. Please note: By selecting “Yes” to the participation questions for each region, the corresponding Post-Test questions will appear. Click “Save Draft” to save your responses and finish later. When you are ready to submit your answers, click “Preview” to review all responses, then click “Submit.”
- Click “Next” to complete the Evaluation form, then click“Submit.”
- Claim 1.0 CME credit and 1.0 MOC point per region (up to 8.0 total for 8 regions of NephMadness).
- Save/print your certificate.
The CME and MOC activity will expire on June 14th, 2021.















Leave a Reply