#NephMadness 2022: Animal House 3
Submit your picks! | NephMadness 2022 | #NephMadness | #AnimalHouse
Selection Committee Member: Kelly Hyndman @DrKeeksPhD
Kelly Hyndman is an Assistant Professor of Medicine in the Section of Cardio-Renal Physiology and Medicine, Division of Nephrology, at the University of Alabama at Birmingham. She has trained both in comparative physiology and kidney physiology labs and is currently a principal investigator of a basic science lab with research interests in novel mechanisms of fluid-electrolyte balance.
Writer: Tiffany Truong @CRRTiff
Tiffany Truong is a transplant nephrology fellow at the University of California, San Francisco. Her academic interests include transplant nephrology, electrolyte disorders, medical education, and finding creative ways to better advocate for her patients.
Competitors for the Animal House Region
Giraffe vs Whale
Bear vs Lungfish

Finn by Susan Quaggin @SusanQuaggin (used with permission)
Welcome to the third evolution of Animal House! Over the years, we have progressed from fish to philosopher, foraying into the wild world of animals to explore the intricacies of water balance then the story of nephron evolution. If studying the diversity of life in all its forms has taught us anything, it is as French physiologist Claude Bernard stated best: “The constancy of the internal environment is the condition of a free and independent existence”. This, le Milieu Intérieur, our bodies and chemistries, is how we exist. In the world of nephrology, homeostasis is certainly no joke.
No matter how large or small the animal, how harsh or changing the environmental conditions, we exist because we control the ocean we carry within. As writer Jean-Baptiste Alphose Karr and singer Corinne Bailey Rae both concur, “The more things seem to change, the more they stay the same.” In a multitude of dynamic conditions, let us see how the living organism maintains balance in this changing world.
Giraffe vs Whale
Our first matchup is between two giants of land and sea. Nature’s skyscraper, the giraffe, must handle the unique hemodynamic stresses of the high arterial pressures needed to perfuse the brain. What secrets can the giraffe tell us about how to handle these high blood pressures? In the ocean, the massive whale has a whole lot of body mass for which it needs to maintain homeostasis, and a unique kidney structure with which it accomplishes this.
Giraffe

Copyright: Lukiyanova Natalia frenta / Shutterstock
Even at first glance, the giraffe intrigues. Given the species name of ‘camelopardalis’ by Carl Linnaeus himself for its camel-like shape and leopard-like coat, the giraffe is the tallest living terrestrial animal at ~20 feet tall with an extremely long neck and legs. With a head so high above the level of the heart, scientists have naturally questioned how the giraffe maintains cerebral perfusion and regulates its blood pressure.
With Dr. Robert Goetz’s expedition to South Africa in the 1950s, study into giraffe physiology was launched. During that expedition, they found that the mean arterial blood pressure in the carotid artery was 200 mmHg at the base of the brain, and only got higher moving down to the base of the neck where the blood pressure reached a whopping 300-360 mmHg. The giraffe tolerates twofold higher systemic blood pressure than other mammals of the same body mass. How does the ‘hypertensive’ giraffe protect its kidneys from hypertension-related injury?
Studies of the giraffe’s glomerular filtration rate (GFR) provide some clues. If the giraffe’s glomerular capillaries saw the high pressures present systemically, the GFR would be expected to be high since transmural pressure across the glomerular capillary drives glomerular filtration. However, the measured GFR was surprisingly low – only 342 mL/min on average, or 0.7 mL/min/kg when normalized to body weight. This GFR is 40% lower than the predicted value of 1.2 mL/min/kg based on a scaling relationship from Dr. Michael Singer which estimates GFR based on the body mass of different animals.
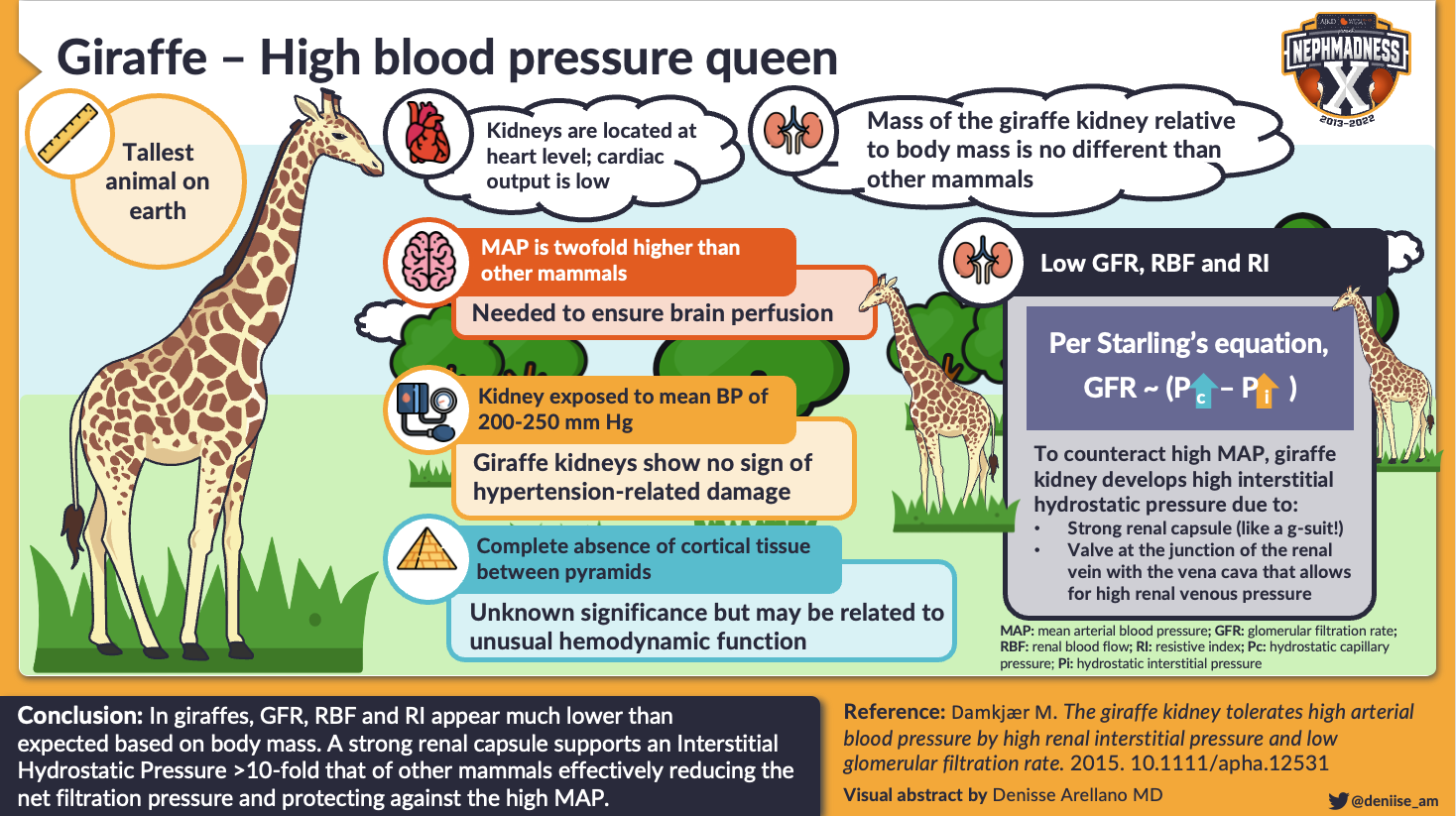
Visual abstract by @deniise_am on Damkjaer et al
Interestingly, the GFR of larger animals has been noted to not increase directly proportional to their body mass, in a similar manner to the metabolic rate of animals, suggesting that animals become more energy efficient with increasing size and also that GFR matches catabolic processes. The lower-than-expected GFR in the giraffe suggests that they do not obey the overall scaling relationship or that they have even lower than expected metabolism for their size. Though this potentially low basal metabolic rate of the giraffe could explain some of the lower-than-expected GFR seen in the giraffe, the GFR is still disproportionately low if the glomerular capillary is truly exposed to such high pressures as seen systemically.
One hypothesis to explain both the decreased GFR and the lack of hypertension-related injury is that the giraffe’s glomerular capillary basement membranes have different mechanical properties which decrease filtration across it, potentially protecting the vasculature from damage. However, this does not seem to be the case as the filtration fraction, or ratio of GFR to renal plasma flow, is not low at 0.3 in the giraffe. The microscopic structure of the giraffe’s glomeruli do not appear much different either. What may explain this lower GFR and a protective mechanism against high renal arterial pressures is that the giraffe has a high hydrostatic pressure in the renal interstitium. Recall Starling’s equation:
GFR = Kf (Pc – Pi) – σ (πc – πi)
Kf = filtration coefficient
Pc = hydrostatic capillary pressure
Pi = hydrostatic interstitial pressure
σ = reflection coefficient
πc = oncotic capillary pressure
πi = oncotic interstitial pressure
If the interstitial hydrostatic pressure is high, the pressure gradient across the glomerular capillary membrane (or the transmural pressure) could be reduced, accounting for decreased GFR despite high blood pressures. These high interstitial pressures are proposed to be created by two structural adaptations: a thick, strong renal capsule which can contain internal pressures 10x higher than that of other mammals and which functions as a kidney ‘g-suit’ to maintain internal pressure (or compression stockings, if you will, like the fascia in the giraffe legs which are thought to prevent edema), and a valve-like structure at the junction of the renal vein and vena cava.
High blood pressure is present systemically in the giraffe, and its ability to maintain homeostasis is unlikely to be explained simply by these two features in the kidney alone. The mechanisms underlying hypertension are complex, frequently multifactorial, and often thought to have genetic underpinnings such as noted with ApoE mutations. Could the giraffe’s ability to tolerate high blood pressure also be explained by the evolution of certain genes adapted to this as well?
One research group sequenced the genome of a giraffe and determined that compared to other mammals, giraffes have 101 genes with mutations that likely provide an adaptive advantage and an additional 359 genes that are considered to be rapidly evolving. These 460 genes are enriched in pathways involved in circadian rhythm, nervous system, and blood pressure regulation. Interestingly, the gene for fibroblast growth factor receptor-like protein 1 (FGFRL1) contains giraffe-specific mutations and has more nucleotide substitutions than any other gene in the giraffe genome. Mutations in this gene can cause cardiovascular and skeletal defects in humans and mice, and the variants in giraffes were hypothesized to play an adaptive role.
Researchers replaced the FGFRL1 genes of mice with those of giraffe and found that they could not induce hypertension with angiotensin II infusions for 28 days in those mice with the giraffe-type FGFRL1 genes, whereas mice with their regular genes developed hypertension with increased myocardial and renal fibrosis.

Giraffe-type FGFRL1 mice showed significantly lower systolic, diastolic, and mean arterial pressures (mmHg) than WT FGFRL1 mice after Ang II infusion for 28 days. *P < 0.05, **P < 0.01, ***P < 0.001, one-way ANOVA followed by Tukey’s post hoc test. Figure 2A from Liu et al, Science Advances (CC BY-NC).
Thus, there are clear genetic differences in the giraffe that likely provide an evolutionary advantage and physiologically allow these giants to sustain high blood pressures without damaging their organs.
Check out this podcast episode of Freely Filtered featuring Kelly Hyndman @DrKeeksPhD:
#43 NephMadness and Animal House
Whale

Copyright: Christian Vinces / Shutterstock
It is easy to believe that we are familiar with the whale, the enormous marine mammal seen during a whale-watching tour or at an aquatic park, whose capture is an industry that dates back to 3000 BCE, and which occasionally make headlines after being stranded or beached. But what we know of the whale is akin to what we know of the sea, which comprises more than 90% of the planet’s living space and yet remains largely unexplored, dubbed Earth’s final frontier. What we know is that the whale is intelligent – they are social and playful, can hunt in a group, and have brain structures that support this complexity with more convolutions and more intricate limbic systems than even seen in humans. They have few offspring and live long lives, with one species, the bowhead whale, having a lifespan of over 200 years.
From an evolutionary perspective, whales are cetaceans, a group of animals that include dolphins and porpoises, with the unusual lineage of having returned to the sea from land, losing their hindlimbs and morphing their forelimbs into flippers which to this day retain the vertical orientation of locomotive limbs in contrast to the horizontal fins of fish. In fact, the ancestors of cetaceans were 4-limbed hoofed animals closely related to the hippo. One of these ancestors, the Indohyus (translates into ‘India’s pig’), lived on the Indian subcontinent and was mostly terrestrial but could dive into the water to hide from predators. It is remarkable to imagine this small, hoofed pig-deer-like animal is an ancestor of the whale! The other hoofed animals that diverged in evolution became deer, llamas, sheep, cows, and… giraffes. That’s right – our head-to-head competitors share a common evolutionary path, and the whale and giraffe are more closely related than the whale is to salmon or any other fish that may be swimming in your aquarium!
Evolutionary intrigue aside, what makes whales so spectacular is also what makes them both fascinating and difficult to study. They are foremost enormous animals – the largest animals on the planet with the blue whale weighing in at 199 tons or 199,000 kilograms. They can also dive to great depths of 2,000 meters, enduring great external pressure. These great depths are associated with all sorts of physical and biochemical challenges. For example, with every 10 meters of depth, the diving mammal will experience twice the hydrostatic pressure it would at the surface. This pressure would also result in the air in the lungs occupying half of its volume. Although the whale’s cousin, the giraffe, has to handle very high internal pressure, whales have to handle great changes in external pressure. How has the whale adapted such that while diving all of its body fluids aren’t forced into the interstitium, the lungs don’t collapse, and the kidneys aren’t severely injured?

Infographic by @brian_rifkin.
To handle their enormous size, the whale does have quite large kidneys. For example, the killer whale (Orca) weighs roughly 2,000 kg and the kidney weighs in at 3.4 kilogram per kidney, or the weight of a newborn human baby. Relative to its body weight, the total kidney mass is proportionally right in line with other mammals at 0.33% of total body weight (A spectacular constant of nature, which applies to humans as well! Do the math!). Where the whale distinguishes itself is how this kidney is structured. The whale kidney is made up of hundreds of grape-like clusters of small kidney units called renules, which each have a cortex, medulla, calyx, and duct that joins with others to form the ureter. It is speculated that the structure of these renules helps whales adapt to diving. How is this the case?
Firstly, the cortex and medulla in each renule is separated by a layer of collagen, elastin, and sometimes smooth muscle. This is called the sporta perimedullaris musculosa. Although the function of this part of the kidney is unclear, it is hypothesized that it acts to quickly contract and push urine out of the medulla right before a dive as concentrated urea would be cytotoxic during prolonged submersion.
Second, the proximal tubules in each renule have significant glycogen reservoirs. Given the high metabolic demand of the kidney, during diving, renal blood flow, GFR, and urine flow are significantly reduced and blood diverted to the muscles and brain. You can imagine how hard this would be to study renal hemodynamics in a diving whale, but in diving seals this has been documented. Given this hypoxic-anoxic kidney state during diving, these glycogen stores are likely key to promote anaerobic glycolysis and hopefully limit any ischemic injury.

Infographic by @cteodosiu.
Interestingly, despite the whale having so many renules, the total mass of glomeruli relative to renal parenchymal mass is similar to that of other mammals. Instead, the glomerular mass is composed of many more, but smaller glomeruli. That is, they have 8 times more nephrons than would be predicted by their weight, and 100 times more nephrons than humans. Having more but smaller glomeruli allows the tubules to not become longer and longer as the total renal parenchymal size grows with the size of the animal. Not all large animals adopt this method; the elephant has larger glomeruli with accompanying longer tubules to accommodate its increasing kidney mass. In mammals, the resistance to flow in these longer tubules may be overcome by higher arterial pressures, but in non-mammalians with longer tubules, cilia are required to drive the filtrate through the tubules. These varying approaches, their advantages and disadvantages, are interesting aspects to consider when applied to the mechanics of building a synthetic kidney and the desired structure of organoids.
COMMENTARY BY KELLY HYNDMAN:
Animal House 3: Life at the Extremes
Bear vs Lungfish
For our next pair of animals, we explore how two animals use states of dormancy to maintain homeostasis in harsh conditions. The hibernating bear in winter is a ‘metabolic magician’, but can it hold a flame to the lungfish – an actual fish out of water?
Bear

Copyright: Photo Spirit / Shutterstock
“This paradigm of fuel efficiency, absence of toxic wastes, budget balancing, and productivity is one from which individuals, governments, and businesses have much to learn.” – Ralph A. Nelson in Black Bears and Polar Bears—Still Metabolic Marvels (Mayo Clinic Proceedings)
The American black bear hibernates for up to 7 months each year, when they neither eat, drink, move, urinate or defecate. Suffice it to say that hibernation is no regular old nap, but a state of altered and decreased metabolism coordinated by multiple organ systems to conserve energy when food is scarce in the winter. It is characterized by hypothermia, slower breathing and a slower heart rate, and entry into this state must also allow for recovery from it without injury.
So bears can get away without eating by slowing their metabolic processes, but how can they get away without clearing waste products? Even with minimal extra effort on the bear’s part, the everyday cellular processes of living produce enough waste that must be excreted, right? During hibernation, bears decrease their GFR by ~70% and become anuric as the filtrate is reabsorbed in the kidney and the bladder, which reabsorbs ~100 mL/day. The human bladder does not reabsorb urine. Despite their decreased GFR, hibernating bears are able to maintain their blood urea nitrogen at a low concentration of only ~5-10 mg/dL through two mechanisms.
Firstly, bears generate less urea during hibernation, as they and other hibernating animals switch their metabolic programs to primarily catabolize fat rather than protein. Using fat for energy not only avoids the generation of nitrogenous waste products, but also has the added benefit of generating metabolic water to maintain hydration. Shifting to fat catabolism is such a crucial part of hibernation that bears behaviorally prepare for it with a period of hyperphagia in the late summer, storing lipid reserves before they enter hibernation.
The second way that bears maintain nitrogen balance is perhaps what earned them the moniker ‘metabolic magicians’: the urea that bears do generate is recycled – back into amino acids and protein! This was demonstrated when injection of urea radiolabeled with radioactive carbon resulted in radiolabeled carbon in amino acids and plasma proteins. Transforming urea into the building blocks of protein may sound more like metabolic alchemy than recycling, and indeed how urea is recycled still is not well understood though a few theories exist. One proposed mechanism is that urea passes through the intestine, is hydrolyzed by bacteria into ammonia which is then reabsorbed and reacts with glycerol derived from lipolysis to make amino acids.

Visual abstract by @JMTeakell on Stenvinkel et al.
Another theory of urea recycling posits that there is a central role for a protein called sirtuin. Sirtuins are a class of NAD-dependent proteins important in cellular metabolism. The mammalian subtype SIRT5 specifically upregulates the activity of carbamoyl phosphate synthetase 1 (CPS1) which catalyzes the initial and rate-limiting step of the urea cycle converting ammonia to urea in mitochondria. During fasting, NAD in mitochondria increases, activating SIRT5 and subsequently CPS1-catalyzed ammonia detoxification to urea. SIRT5 can thus link states of starvation to increased detoxification of nitrogenous waste products from amino acid catabolism. Sirtuins have yet to be studied in hibernating mammals, but their importance in transcriptional control of metabolism has been inferred to be important in the hibernating state. Furthermore, they provide a target to modulate metabolism through dietary changes.
While bears may be the first animals that come to mind when we think of hibernation, they differ from ‘true’ or ‘deep’ hibernators such as squirrels and hedgehogs which drop their body temperatures nearly to ambient temperatures of 0-5°C and decrease their metabolic rate by >90%. Bears only drop their body temperature to 30-35°C and decrease their basal metabolic rate by 40%, so it was questioned for some time whether they hibernate or not. Yet, this spectrum of metabolic shifting is perhaps what makes bears so fascinating. Bears can revert to a low waste-producing, protein-conserving state when food is not available without as drastic a change in overall metabolism as ‘true’ hibernators, and this state is dynamic and regulated by processes which may be triggered by external stimuli. In the summer, black bears that did not have access to food and water did not have the hibernation responses that they did in the winter, and subsequently became azotemic and dehydrated.
With this in mind, studies into hibernation have recently shifted into the molecular and genetic basis which regulate multiple processes involved in sustaining a hibernation state. Specifically, epigenetic mechanisms including DNA methylation and histone modification as well as transcriptional control of genes through covalent attachment to proteins of the small ubiquitin-related modifier (SUMOylation) may be global controls which suppress the expression of certain genes while enhancing others. In a quest to understand how the kidneys of bears are able to withstand months of decreased function and metabolism, a genome-level study of the American black bear identified 68 genes that were upregulated in the kidney during hibernation and 101 genes that were downregulated. The upregulated genes including cytokine suppression genes which are anti-inflammatory, and genes that cluster with a developmental stage of glomerulogenesis, tubulogenesis and maturation, while genes that were downregulated were clustered with a developmental stage of FGF and Wnt signaling known to be pro-fibrotic in the kidney.
Elucidating the mechanisms of the processes of hibernation can have wide-ranging clinical applications from managing chronic kidney disease, preventing muscle wasting and osteoporosis, addressing ischemia resistance, hypothermic organ preservation, and even increasing the duration of space travel.
Lungfish
“Methuselah leaped outside, spending up to an hour on the floor before she was discovered.” – Peter Hartlaub in The oldest aquarium fish in the world lives in San Francisco. She likes belly rubs. (San Francisco Chronicle)
You can still meet Methuselah today, a 90-year-old lungfish and the oldest fish in captivity, at the California Academy of Sciences in San Francisco, where about a decade ago she is rumored to have escaped her tank. For this fish out of water – she probably could have survived just fine.

Photo of Methuselah by Gayle Laird / California Academy of Sciences (used with permission)
Found in the waters of Africa, South America, and Australia, the lungfish is a peculiarity – a fish that can walk and that would drown in water if unable to breathe air. Despite being obligate air breathers, the lungfish cannot remain completely out of water or it would desiccate and die. Yet the freshwater rivers and lakes it inhabits frequently do dry out. To avoid desiccation, the lungfish burrows itself into the mud and cocoons itself in a coat of mucus secreted from its skin with a small hole to breathe. This is a state called estivation, and can last anywhere from 6 months to a whopping 4 years when wet conditions return. Similar to a bear in hibernation, the lungfish in estivation is also in a state of dormancy during which it decreases its metabolism and becomes anuric.
During estivation, the lungfish switches from a state of high-water intake and excretion to a state of water scarcity and conservation. It will use its muscle proteins to create an emergency supply of water. From the same proteins it will produce urea, an endogenous osmole, to hold on to this water. How does a kidney itself adapt to such profound cyclical changes? One study of the lungfish kidney by microscopy found dynamic changes in the structure of the renal corpuscle (the glomeruli and Bowman’s capsule) during estivation. In essence, the renal corpuscle shrinks. The typically flat parietal cells of Bowman’s capsule become stratified, the glomerular capillaries collapse, the glomerular basement membrane thickens up to 4 times its regular size, and the podocytes are arranged closer. These changes thicken the filtration barrier and reduce the filtration coefficient, minimizing glomerular filtration and helping the lungfish to conserve water.
Meanwhile, all the tubular segments of the lungfish dilate, though it is unclear what the significance of this change is. These tiny, dense glomeruli with thickened basement membranes shut down their functioning as filtering units when filtration is not advantageous, and then, remarkably, return to their prior state and restore filtration after a few days of recovery from estivation. From Ojeda et al, the lungfish glomeruli “… is not a fixed composite.” Rather, it appears to be a highly dynamic structure capable of modifying its architecture in response to environmental changes.”

Visual Abstract by @SRameshMD on Fishman et al
While all the components of the renal corpuscle change during estivation, the podocyte holds particular interest. In estivation, the podocyte bodies come closer together, and the podocyte processes flatten and can be lost. These morphologic changes also appear in freshwater fish adapting to seawater, hibernating animals, dehydrated rabbits, and early ischemic acute kidney injury in humans. In these conditions where conserving water is key, the loss of podocyte processes can be physiologic as is the case with estivating lungfish or pathologic as is the case with ischemic kidney injury in humans. As a physiologic process, the loss of podocyte processes allows podocytes to become cuboidal in shape and rest directly on the glomerular basement membrane, reducing the area of filtration.
How this process is controlled and how podocytes return to their prior state is of great interest but still unknown. In humans, the loss of podocytes is not associated with regeneration and recovery, so the ability of the lungfish to do this is quite a marvel and a mystery. What has been observed during this recovery period after estivation is only that the podocytes of lungfish resemble undifferentiated cells and exhibit cell processes which resemble developing axons.

Photo of Methuselah by Gayle Laird / California Academy of Sciences (used with permission)
If having dormant glomeruli which perk up in water weren’t impressive enough, the lungfish distinguishes itself for its special place in evolutionary biology. Dubbed a ‘living fossil’, the lungfish has not undergone significant evolutionary change since it first showed up in the fossil record about 400 million years ago in the Devonian period. This period, also known as the Age of Fishes, was when many diverse fish species evolved, including the tetrapods who were the first creatures to move from sea to land and are the nearest common ancestor to all living amphibians, reptiles, birds and mammals. As lungfish and tetrapods share a common ancestor, the lungfish may be the closest living animal today to tetrapods and the link to our origins as terrestrial beings.
Investigating the characteristics present in lungfish reveals how long these features have been around. For example, the development of lungs and 4 similarly sized fins which resemble a tetrapod’s limbs tells us that animals first learned to breathe and walk in water. Similarly, functional studies have found that lungfish have vasotocin(non-mammalian vasopressin)-aquaporin and aldosterone-ENaC pathways, indicating that these pathways preceded tetrapods and are ancient mechanisms to conserve water that predate even our own terrestriality! With this sort of lineage, it is no wonder that the lungfish is a favorite pet among paleontologists, or that vasopressin has been attributed as a part of our identity as humans:
“What is the role of vasopressin?”
A nephrologist will talk about osmolality and aquaporins.
An intensivist might talk about its use as a pressor.
But, ask an evolutionary or social scientist and you might be surprised by the answer…#MedTwitter #NephTwitter #NSMC
— Caitlyn Vlasschaert (@DrFlashHeart) August 13, 2020
– Executive Team Members for this region: Anna Vinnikova @KidneyWars and Matthew Sparks @Nephro_Sparks
How to Claim CME and MOC
US-based physicians can earn 1.0 CME credit and 1.0 MOC per region through NKF PERC (detailed instructions here). The CME and MOC activity will expire on June 1, 2022.
Click to read more NephMadness 2022 Regions
Submit your picks! | #NephMadness | @NephMadness | #AnimalHouse

Leave a Reply