Transplant Practices During the COVID-19 Pandemic
In this author interview, AJKD Social Media Editor, Timothy Yau @Maximal_Change, talks to Krista Lentine @KristaLentine, Roslyn Mannon @mannonmom, and Michelle Josephson @MichelleJTx, co-authors of a recent Perspective article published in AJKD looking at the impact of the pandemic on kidney transplant practices across the world.
AJKDBlog: Can you give us a big picture overview of how transplant programs responded to the pandemic in March 2020?
Authors: In March 2020, most transplant programs grappled with how best to manage transplantation in the unfolding pandemic. Coupled with concerns over whether it was more or less risky for patients to receive transplants with the associated immunosuppression, the community also faced practical issues like shortages of Personal Protective Equipment (PPE), ICU beds, and staff, that made transplant procedures difficult. Furthermore, due to the ICU shortage, concerns about donors having COVID-19, and the ban on non-essential workers in the hospitals, there were fewer deceased donor organs available. Most programs proceeded with heart and liver transplants as they were felt to be lifesaving. Kidney transplantation fell into a gray zone. For deceased donor kidney transplantation, most programs proceeded with transplanting highly sensitized patients—as programs felt these offers may be the individual patient’s only chance to receive a compatible organ—while postponing deceased donor kidney transplantation in the non-sensitized. Living donor transplants initially largely paused out of concern for the kidney donor’s well-being, as well as the sentiment that recipients could be better managed once the pandemic was more under control. Living donor transplantation fell into the “elective” category and were deemed possible to safely delay, especially in locations with strained resources such as insufficient ventilators. Living donor transplantation is more complex, as it requires efforts to ensure the safety of two patients, the recipient and the living donor. Kidney paired donation, which incurs logistical complexities due to organ or patient travel, was particularity affected in a time when self-quarantine was advised to reduce the risk of SARS-CoV-2 transmission.
With time, better screening and reporting of deceased donors, and more available PPE, kidney transplant programs resumed activities. Additionally, the Organ Procurement and Transplantation Network (OPTN) implemented tools to collect and disseminate infection status, which helped support the re-opening of deceased donor transplantation. Nevertheless, many programs modified immunosuppression protocols, using less T cell depleting agents at the start. With time, deceased donor kidney transplantation volumes in 2020 surpassed those in 2019, even though there was a standstill from March through May of 2020. However, living donor transplantation volumes for 2020 were less than those seen in 2019.
AJKDBlog: How did COVID-19 affect potential living donors? Are there concerns about a reduced pool of donors as a result of the pandemic?
Authors: While surgery paused, expanded use of telehealth allowed some programs to continue donor candidate evaluations and develop new workflows for education, intake, and assessment. By June 2020, monthly living donor transplant rates returned to pre-pandemic levels, although the volume decrement from deferred procedures did not recover. Broader testing availability and the advent of effective vaccines now provide more assurance for safety, with the professional societies recommending complete vaccination of donors and recipients by 2 weeks before surgery.
As the prevalence of individuals with recovered COVID-19 increases in the population, determining if and when organs from these donors can be safely used is an important research priority. The pandemic-related economic recession may also have a long-lasting impact on opportunities for living donation, even after the end of the public health emergency. A recent survey identified economic impacts as a leading pandemic-related concern among living donors, including the possibility of losing employer-based health insurance.
AJKDBlog: You mention telehealth in the above question, and how it is being used to continue to screen living donors. Nephrology in particular is critically dependent on the need for frequent laboratory testing. What lessons did we learn in trying to deliver optimal care during this difficult time?
Authors: Telehealth has provided programs and patients with flexibility and convenience for some aspects of both recipient and living donor evaluation and follow-up care, but programs have not embraced the technology for all aspects of transplant evaluations—most remain unwilling to use telehealth for the surgical evaluation. As you mentioned, telehealth visits decrease the need for patients to visit facilities, but do not address the critical issues of laboratory test monitoring. During the pandemic, some programs adopted the use of home phlebotomy services while others have advised patients to reduce testing frequency and/or to time visits to minimize waiting times. Home blood pressure monitoring and other remote monitoring tools may increase the options for adjunctive testing and monitoring outside of the clinic setting.
AJKDBlog: One of the most common questions we encountered was how to manage immunosuppression in transplant recipients who contracted COVID-19. Obviously there is always a balancing act between the infection and the risk of rejection, but do you have general rules for your approach to this clinical decision?
Authors: At this point, immunosuppression management in the setting of COVID-19 falls as much into the art of transplant medicine as the science. Data are lacking, as are useful biomarkers that could allow one to more scientifically titrate immunosuppression. The key is reducing immunosuppression enough to enable the immune system to fight SARS-CoV-2 infection, but not so much as to allow for a rejection or cytokine storm. The British Transplant Society (BTS) guidelines advise stopping anti-proliferative agents (MMF or azathioprine) in all patients with COVID-19 infection, whether or not they need hospitalization. They also advise considering reduction of CNIs after review of total immunosuppression burden for transplant recipients with COVID-19 who do not require hospitalization. For those who do require hospitalization, the BTS advises reducing or stopping CNIs. The American Society of Transplantation is less prescriptive and notes that the decision should be “based on severity of COVID-19 disease in comparison with rejection risk.”
Currently in our practice, we generally reduce or stop the anti-metabolite after COVID-19 diagnosis, and do not resume until recovery. For those who are severely ill we also stop the CNI. We continue steroids. If an individual is at high risk for rejection, we may continue but reduce the anti-metabolite dose. Our approach is to enable the immune system to fight the virus while trying to avoid risking rejection. As you can see, each decision requires an individualized approach.
AJKDBlog: Lastly, with vaccines rolling out and nearly 50% of the population in this country vaccinated, are there unique factors for kidney transplant donors and recipients? Is your general recommendation that anyone with a kidney transplant on immunosuppression should receive a vaccine? And given the lack of studies that included transplant recipients, should we assume the same efficacy in these patients as shown in clinical trials?
Authors: As the pandemic progressed and vaccine emergency use authorizations were in sight, the questions for the transplant community were two-fold: (1) would vaccination trigger an immune response against the allograft and (2) would vaccination be effective in the immunosuppressed transplant recipient, where both induction and maintenance therapies are targeted to limit T cell function and proliferation? Ultimately, both mRNA vaccines (Moderna, Pfizer) and the replication deficient-adenovirus-based vaccine (J&J/Janssen) were available to the public. The vaccine roll-out included those with chronic diseases and organ transplants in the second phase, but availability was limited initially. This was further complicated by state-by-state policies (in which transplant centers were not consistently provided vaccine supply for their patients) and some initial confusion about the adenoviral-vector based vaccine preparation.
There is no published estimate of the total number of vaccinated solid organ transplant recipients. Challenging vaccine hesitancy is a priority in the field. For otherwise healthy individuals that have been or aim to become living donors, a recent publicly available survey identified that the majority (77%) planned to or had received COVID-19 vaccination, with 23% either not planning to receive or unsure.
The immune response to vaccinations continues to be the most critical concern. Prior work has demonstrated suboptimal immune responses to annual influenza vaccination, although with a reduction in complications and the spread of influenza in those immunized. So, it should not be unexpected that recent reports have demonstrated limited responses to mRNA vaccination. In a voluntary participation study of 439 US kidney transplant recipients, only 17% of patients within a median of 20 days of their first vaccine dose expressed either S1 domains of the SARS-CoV2 spike protein or receptor binding. Similar findings were seen in recipients who received 2-dose vaccines, and are also seen across other solid organ recipients. Even more disconcerting has been the demonstration of rare but severe COVID-19 disease following vaccination.
Regardless of these findings, it is clear that current strategies are insufficient to assuredly protect our patients, who must continue their vigilance in the forms of mask wearing, hand hygiene, and social distancing. Further investigations and recommendations about additional vaccine doses are pending, and are much needed. These findings also support the importance of vaccination in the CKD and ESKD population, where seroconversion, although reduced compared to health individuals, still offers some protection.
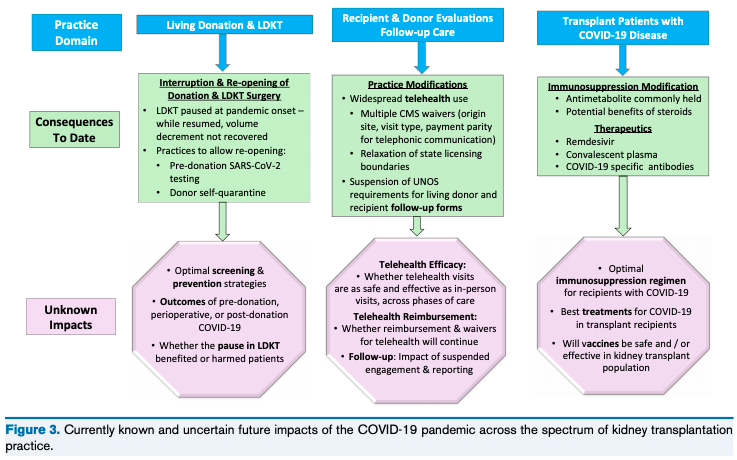
Figure 1 from Lentine et al, AJKD © National Kidney Foundation
To view Lentine et al [FREE], please visit AJKD.org.
Title: Practicing With Uncertainty: Kidney Transplantation During the COVID-19 Pandemic
Authors: Krista L. Lentine, Roslyn B. Mannon, Michelle A. Josephson
DOI: 10.1053/j.ajkd.2020.12.003

Leave a Reply