#NephMadness 2023: It’s Always Rosy in the World of Complement
Submit your picks! | NephMadness 2023 | #NephMadness | #TMAregion
John Sperati @JohnSperati
Dr. John Sperati is an Associate Professor of Medicine and an adult nephrologist at The Johns Hopkins University School of Medicine. He serves as the director of the Nephrology Fellowship Training Program, and his clinical and research interests focus on complement inhibitors and thrombotic microangiopathy.
Competitors for the Thrombotic Microangiopathy (TMA) Region
Primary TMA vs Secondary TMA

Copyright: ShaiKu/Shutterstock
What’s in a name? That which we call a rose
By any other name would smell as sweet
-William Shakespeare, Romeo and Juliet
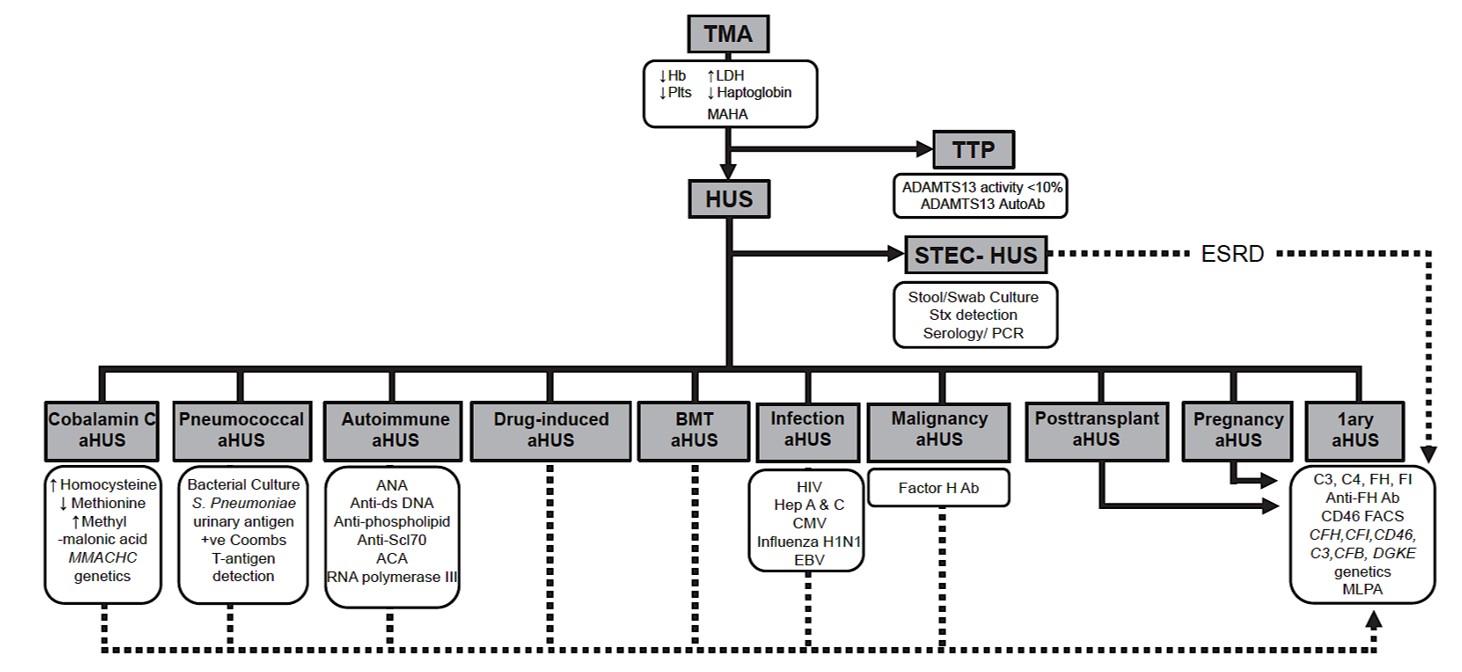
Hemolytic uremic syndrome. Atypical hemolytic uremic syndrome. Primary. Complement mediated TMA. Malignant hypertension induced TMA. Secondary. We-don’t-think-this-is-complement-and-it’s-not-a-TMA-but-it-looks-like-a-TMA. Maybe-it’s-complement-mediated-maybe-it’s-not-complement-mediated-TMA. Oh my! What is more maddening– other than those squeaking sneakers on the courts throughout the month of March- than our current, arbitrary classification of thrombotic microangiopathies?
The term “atypical hemolytic uremic syndrome” may have been first used in 1965 to differentiate diarrheal-associated HUS from non-diarrheal. By 1978, there was a call for an etiologic based classification of HUS while acknowledging that specific molecular mechanisms likely remain unknown. Nevertheless, aHUS evolved into the preferred term for all TMAs that were not Shiga toxin HUS or TTP. This was eventually further ensconced in our collective lexicon by the product labeling for eculizumab that restricted its use to aHUS. But what is aHUS? The term does not convey mechanism or etiology. It lacks a precise definition. There are kidney limited forms that lack the “H,” and kidney injury is at times sufficiently mild such that the “U” is not present either.
Recognizing the limitations of aHUS as a classifying term, and to better orient the focus on endothelial injury as the foundational event in these disease processes, TMA has emerged as a better but still imperfect rubric. Yet, this field is complicated, and old habits die hard. Over the past several years, for many experts in the field, aHUS has become synonymous with complement. The KDIGO controversies conference included primary aHUS as a complement-mediated etiology of HUS, distinct from other causes (yet with many interconnected arrows).
Another group maintained the primary aHUS label while subcategorizing others (including some associated with complement) under a secondary header. Still others move secondary causes to the primary category and then further subdivide by hereditary and acquired. Now, in 2023, we find ourselves watching Primary (complement-mediated) vs Secondary (maybe complement mediated) TMA in the Sweet 16. Unlike Chris Webber, I’m calling a real timeout.
As nephrologists, we are abandoning primary membranous, primary FSGS, primary MPGN…this is an opportunity to think mechanistically about TMA as well. TMA due to pathogenic genetic variants in complement is estimated to occur in approximately 1 per million individuals, with a familial penetrance well below 50% in many studies (Sansbury et al, Arjona et al, Ardissino et al). Variants are identified in only 50-60% of affected individuals, and many of these variants are of uncertain significance and potentially non-contributory to disease pathogenesis. Thus, pathogenic complement variants are like a well-positioned defender– they need to be poked for a foul to occur. If a multi-hit mechanism (i.e., infection, medication, pregnancy, etc.) is likely required, then how do we draw a dividing line between primary and secondary etiologies of TMA?
When carefully phenotyped, “secondary” TMA are more common than “primary.” While subject to biases of small cohorts, patients with “secondary” TMA are oftentimes not enriched for rare complement variants. Yet despite the absence of identifiable genetic changes, dysregulated complement activity may nevertheless underlie many “secondary” TMAs, such as gemcitabine, malignant hypertension, and antiphospholipid antibody syndrome, to name a few. On the other hand, “secondary” TMAs such as hematopoietic stem cell transplant associated TMA and pregnancy associated TMA do have a high prevalence of germline complement variants. Should these be “primary?” It is also important to remember that not all TMAs are associated with complement activation. Given this, focusing on mechanisms, triggers, and response to therapy is a more meaningful approach than primary versus secondary.
It is estimated that over 300 compounds contribute to the lovely scent we know as rose. Some are even detectable by the human nose at the parts per billion level. Is the rare molecule any less important than the most common to that scent? The amplification loop of the alternative pathway of complement can generate 10,000,000,000 C3b molecules in 15 minutes. If it smells like complement, why call it by any other name? At 500 million years old, complement/TMA is clearly the most senior and wisest competitor in this year’s lineup. Primary (complement mediated)– if I must use that term– for the win!
– Guest Post written by John Sperati @JohnSperati
As with all content on the AJKD Blog, the opinions expressed are those of the author of each post, and are not necessarily shared or endorsed by the AJKD Blog, AJKD, the National Kidney Foundation, Elsevier, or any other entity unless explicitly stated.
Click to read the Thrombotic Microangiopathy (TMA) Region
Submit your picks! | #NephMadness | @NephMadness | #TMAregion


Leave a Reply