Thrombotic Microangiopathies and Stem Cell Transplant
A recent narrative review published in AJKD discusses the association between thrombotic microangiopathy (TMA) and hematopoietic stem cell transplantation (HSCT). AJKD Social Media Editor, Timothy Yau (AJKDBlog), interviewed two of the authors, Rimda Wanchoo (RW) and Kenar Jhaveri (KDJ), to discuss this topic.
AJKDBlog: The association between TMA and HSCT been well-established. Can you tell us how the pathophysiology between HSCT-associated TMA (HSCT-TMA) differs from other forms such as atypical HUS (aHUS) and thrombotic thrombocytopenic purpura (TTP)?
KDJ: TMA is a syndromic presentations of an underlying pathophysiology that might be different in various disease states. In TTP, the defect is in an inhibitor or deficiency in ADAMTS-13. In HUS, it is a disease associated with bacterial antigen activating a complement-mediated process. In aHUS, this is also a complement-mediated process (either acquired or genetic). Drugs, over-the-counter medications (e.g. quinine in tonic water), targeted therapies, chemotherapies, SLE, and other autoimmune diseases can also cause endothelial injury leading to a systemic or renal-limited TMA.
Similarly, HSCT-TMA is also a form of endothelial injury that has a multitude of factors that might trigger it such as chemotherapy induction, radiation, viruses, and/or calcineurin inhibitors (CNIs). The pathophysiology is either a complement-mediated process or perhaps a T cell process that is an “endothelial” variant of graft versus host disease (GVHD) as we discuss in the review.
As clinicians, we should consider moving away from using terms such as TTP and aHUS and use more of a pathophysiologic reason for the TMA (either systemic and/or renal-limited). A concept map we created summarizes all the causes of TMA.
AJKDBlog: Are there any histologic findings unique to HSCT-TMA that set it apart from other forms of TMA?
RW: In general, the pathology shows the classic features of TMA. C4d staining is a marker used in the assessment of antibody-mediated rejection in kidney transplant biopsies and is a marker of complement activation by tissue-specific antibodies. As we discuss in our review, the presence of C4D in one study of children was noted in focal renal arteriolar C4d staining more commonly in patients with (75%) versus those without (8%) HSCT-TMA. Glomerular C4d staining was found to be similar in both groups. Thus, arteriolar C4d staining in kidney biopsy specimens of patients with TMA may be a pathologic marker implicating localized complement fixation.
AJKDBlog: TMA may be an “endothelial variant” of GVHD as you discuss in your review. Can you elaborate on that concept?
KDJ: Some of the literature proposes that TMA and GVHD are not related but both affect the complement cascade. As clinicians we have seen several cases of TMA and concurrent GVHD and a recent reported case series confirms this. It is intriguing and possible that renal-limited TMA might be a variant of GVHD.
GVHD is usually an epithelial cell disease but having an “endothelial” target might be possible in the kidney. In most cases, when TMA is diagnosed in a patient with HSCT, the knee jerk response is to discontinue CNIs. Whether this is of potential benefit or harm is not clear.
If this is CNI-induced TMA, the TMA should get better. If this is a variant of GVHD, we might be causing harm by stopping CNIs. We propose a novel way to consider the link between the two. We hope that this can spark further basic and clinical research to allow for better treatment options for HSCT-TMA.
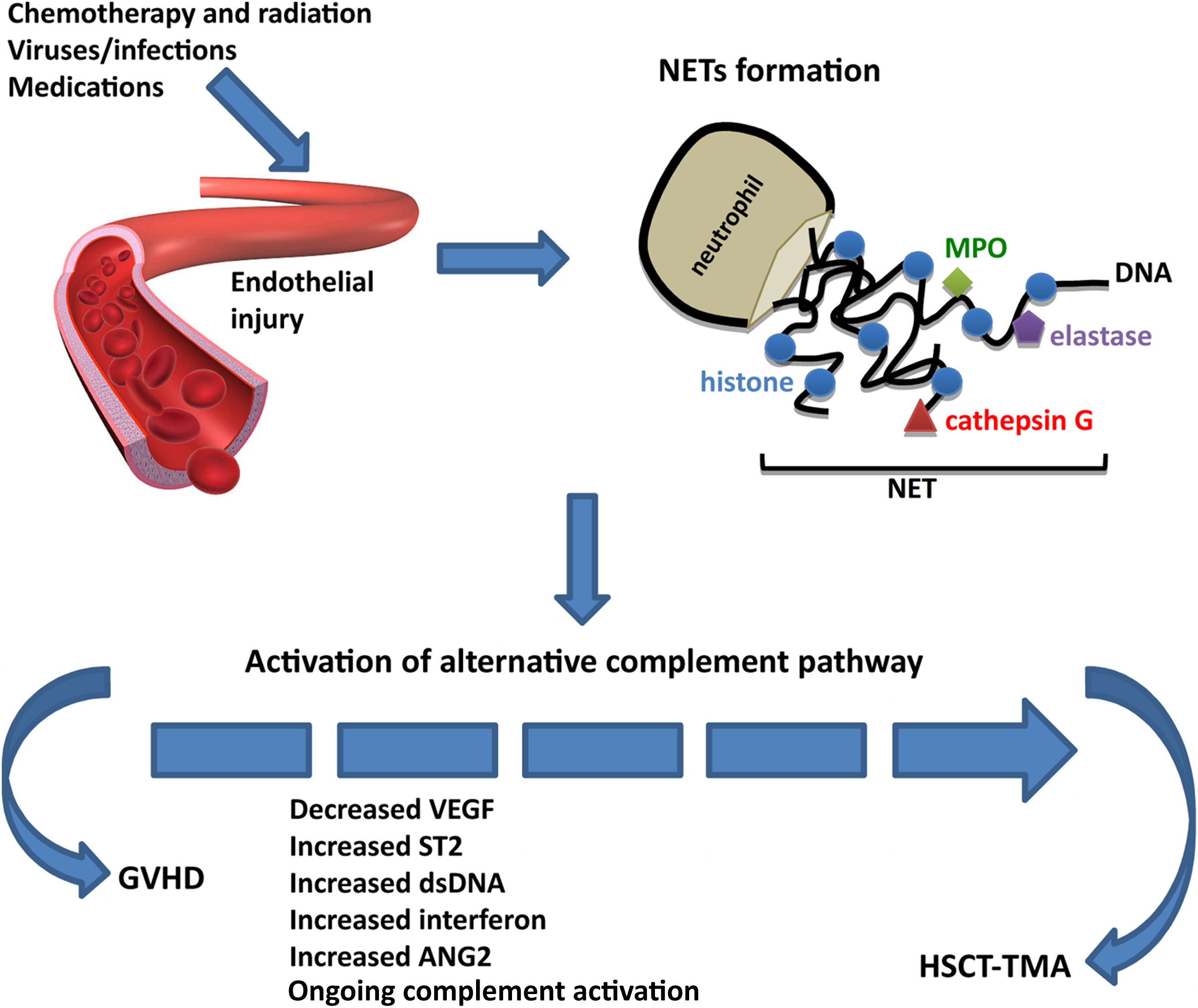
Proposed mechanism of how chronic graft-versus-host disease (GVHD) could be linked with hematopoietic stem cell transplantation–associated thrombotic microangiopathy (HSCT-TMA). Neutrophils release nucleosomes, a complex of DNA and histones, in response to infection or inflammatory stimuli such as chemotherapy, radiation, or infections. Nucleosomes decorated with granular components including myeloperoxidase (MPO), neutrophil elastase, and cathepsin G are known as neutrophil extracellular traps (NETs). In addition to binding and killing microbes, NETs immobilize platelets and erythrocytes. Pre-existing risk factors for HSCT-TMA (infection, medications, radiation) can induce nucleosome release and, in conjunction with granular proteins being released, acute TMA could be triggered. The NETs lead to complement activation, resulting in ongoing HSCT-TMA and GVHD. In certain cases, GVHD then leads to decreased vascular endothelial growth factor (VEGF), increased ST2, ongoing elevation in double-stranded DNA (dsDNA), interferon, and angiopoietin 2 (ANG2), worsening the HSCT-TMA. This creates a potential link (dotted line) between GVHD and HSCT-TMA. Figure 3 from Wanchoo et al, AJKD © National Kidney Foundation.
TY: As you mention above, the etiology of TMA is often unclear – is it from the underlying malignancy, CNIs, concomitant infections, a manifestation of GVHD, or the SCT itself? What is your clinical approach to these complicated patients in determining the etiology?
RW: The etiology of the TMA is important. Just like in renal transplant patients, TMA can be seen with rejection, recurrence of disease, BK virus or CMV, or with CNI use. Similarly, in HSCT-TMA, the clinician should rule out secondary causes that can be treated for the cause of TMA.
Allogenic HSCT patients require a CNI and/or sirolimus for GVHD prophylaxis. Given the association of both CNIs and sirolimus with TMA, replacing them with other appropriate GVHD treatment/prophylaxis such as corticosteroids, mycophenolate mofetil, IL-2 inhibitors, or anti-CD20 agents might be helpful, but this approach has not been studied sufficiently to guide clinical decision making. Supportive measures include platelet and red blood cell transfusion, hypertension management, antimicrobial treatment/prophylaxis, and dialysis if clinically indicated initially. Once the clinician has ruled out infectious causes of TMA and or recurrence or de novo hematologic disease, one can consider using agents listed in the table below:

Summary of Treatment Strategies Used and Being Studied for HSCT-TMA. Table 1 from Wanchoo et al, AJKD, © National Kidney Foundation.
AJKDBlog: These treatments are where we usually see clinicians reaching for different options. How do you decide whether further steps such as plasma exchange, rituximab, or even eculizumab should be considered?
RW: This is a tough decision. The data on most of these drugs are based on observational trials or case reports. In addition, the majority of the data are from pediatric literature. Because most of the available evidence is derived from observational studies and case series, very few proven treatment options are available for nephrologists who encounter patients with HSCT-TMA in clinical practice.
Rituximab can be considered as a treatment option given its utility in the treatment of GVHD involving other organs. Eculizumab as a terminal complement inhibitor may have a therapeutic role with the caveat that reports of its use in this setting have been mostly in the pediatric literature. We hope this review induces other clinical researchers to consider looking at larger trials in using novel B cell and complement inhibitors in the treatment of HSCT-TMA.
AJKDBlog: Many thanks to Drs. Jhaveri and Wanchoo for taking the time to answer these important questions about a difficult disease!
Follow Dr. Jhaveri (@kdjhaveri) and Dr Wanchoo (@renalmyeloma) on Twitter.
To view Wanchoo et al (subscription required), please visit AJKD.org.
Title: Emerging Concepts in Hematopoietic Stem Cell Transplantation–Associated Renal Thrombotic Microangiopathy and Prospects for New Treatments
Authors: Rimda Wanchoo, Ruthee L. Bayer, Claude Bassil, and Kenar D. Jhaveri
DOI: 10.1053/j.ajkd.2018.06.013

Leave a Reply