NephMadness 2015: Obstetric Nephrology Region

Submit your picks! | For more on NephMadness 2015 | #NephMadness or #OBRegion on Twitter
The most intense and emotional moments one can have as a nephrologist are centered around obstetrics. Some examples include:
- Discussing fertility with young women about to start cyclophosphamide
- Deciding if it is safe for a patient with CKD from IgA nephropathy to try and have children
- Helping a patient with Gitelman syndrome through pregnancy
- Going from “I’m pregnant” to “It’s a girl!” with a patient on dialysis
These scenarios can be terrifying while simultaneously being intensely rewarding. These obstetric issues and clinical scenarios are thankfully rare but that means that few nephrologists deal with them regularly enough to be fully comfortable. That is the reason the Obstetric Nephrology region is in the tournament and the reason nephrologists need to buckle down and know their obstetrics. Any of these concepts could break out and make a run for the final four.
Selection Committee member for the Obstetric Nephrology Region:
Phyllis August, MD, MPH
Dr. August is the Ralph A. Baer, MD Professor of Research in Medicine, and a Professor of Medicine, Public Health, and Medicine in Obstetrics and Gynecology at Weill Cornell Medical College. She is Director of the NYP-Weill Cornell Hypertension Center, and the Program Director for the Nephrology Fellowship training program. Dr August is an expert in the field of hypertension and nephrology, Dr. August is committed to the prevention and treatment of high blood pressure. Her clinical practice at Weill Cornell is largely devoted to prevention and treatment of hypertension, kidney disease, and cardiovascular disease. She has served on numerous government advisory boards providing guidelines for the treatment of hypertension and hypertension in pregnancy. She is currently an Associate Editor for JASN.
Meet the Competitors for the Obstetric Nephrology Region
Podocyturia in Preeclampsia
sFlt1 in Preeclampsia
ACOG BP Goal < 160/110 in PE
Lower BP Goal in Preeclampsia
Preeclampsia Due to CKD
CKD Due to Preeclampsia
Pregnancy in ESRD
Pregnancy in Kidney Transplant
Podocyturia in Preeclampsia vs sFlt1 in Preeclampsia
 Preeclampsia represents the intersection between nephrology and obstetrics. Some have even suggested that it is the most common glomerular disease in the world. The last two decades have seen some significant advances to understanding both the treatment and pathophysiology of this disease. Nephrologists were at the center of many of these advances. Preeclampsia affects ~3-5% of all pregnancies worldwide and contributes to significant maternal and fetal morbidity and mortality. sFlt1 really jumped on the scene first in 2002 in a JCI paper (a nephrology fellow at the time, Dr. Maynard, was the first author!) and then in 2004 with this publication by Levine et al in the NEJM. sFlt1 is like the the UNLV of the mid-90s. The news of sFlt1 radiated throughout all of medicine not just nephrology and obstetrics. It was a major breakthrough. Team podocyte is starting to emerge as a serious contender as well. Let’s take a look at this intriguing matchup a little closer.
Preeclampsia represents the intersection between nephrology and obstetrics. Some have even suggested that it is the most common glomerular disease in the world. The last two decades have seen some significant advances to understanding both the treatment and pathophysiology of this disease. Nephrologists were at the center of many of these advances. Preeclampsia affects ~3-5% of all pregnancies worldwide and contributes to significant maternal and fetal morbidity and mortality. sFlt1 really jumped on the scene first in 2002 in a JCI paper (a nephrology fellow at the time, Dr. Maynard, was the first author!) and then in 2004 with this publication by Levine et al in the NEJM. sFlt1 is like the the UNLV of the mid-90s. The news of sFlt1 radiated throughout all of medicine not just nephrology and obstetrics. It was a major breakthrough. Team podocyte is starting to emerge as a serious contender as well. Let’s take a look at this intriguing matchup a little closer.
Podocyturia in Preeclampsia
Podocyturia is all the rage in almost every form of kidney disease. From diabetes, to FSGS, to lupus to glomerulonephritis. In fact, recent evidence is pointing to the presence of low level podocyturia even in normal individuals. A recent JASN paper discusses the possibility of using podocytes captured in the urine for genetic testing. Studies have demonstrated the presence of podocyte protein markers in the urine of women with preeclampsia. To further explore whether the presence of urinary podocyte shedding would predict the onset of preeclampsia a study published in the journal Hypertension in 2013 looked at 3 groups of women all of which were followed prospectively from their initial clinic visit. They matched in a 3:1 ratio normotensive controls (n=44) against patients with gestational hypertension (n=15) and the study group who eventually developed preeclampsia (n=15). What did they find at the end of the second trimester.
- 100% of patients who eventually developed preeclampsia had podocyturia
- None of normotensive or gestational hypertensive patients did!
This equals 100% sensitivity and 100% specificity. Unbelievable! This paper also measured angiogenic factors (sFlt1, PIGF, and Endoglin). These produced much more heterogeneous data and it was difficult to predict which patients would eventually go on to develop preeclampsia. Since 2007, there have been ~11 studies (mostly small) that have demonstrated podocyturia in patients with preeclampsia according to a review published in Kidney International in 2014.
So why are patients with preeclampsia losing podocytes?
Since the presence of podocyturia predates the development of high blood pressure it stands to reason that this might be a very proximal event. This also suggests that the defect leading to preeclampsia also represents more than just endothelial injury and the podocyte might be involved as well. It has been postulated that the loss of podocytes could prompt glomerular destabilization, resulting in more podocyte loss and ultimately proteinuria. However, this is a small study that needs to be reproduced in a larger population. Advances in detection could help make the detection of podocytes in the urine a viable test.
sFlt1 in Preeclampsia
The identification of sFlt1 as a “biomarker” and a potential “pathogenic” factor was a huge advance in the field. What is sFlt1? It is soluble fms-like tyrosine kinase-1. It is actually a splice variant of the better known vascular endothelial growth factor receptor 1 (VEGF-R1). sFlt-1 freely circulates and reduces the level of both VEGF and placental growth factor (PIGF). A paper in JCI and another in NEJM solidified its role in preeclampsia. These publications demonstrate increasing levels of sFlt-1 and decreasing levels of PIGF in preeclampsia.
What is really happening here?
Is it the actual sFlt1 causing preeclampsia or is it preeclampsia itself leading to increased sFlt1 levels? Evidence has pointed to the production of excess sFlt-1 by the hypoxic/ischemic placenta. The sFlt-1 acts as a sort of sink for VEGF, not allowing it to bind to VEGF-R1 on the cell surface of the vasculature, further leading to generalized systemic endothelial dysfunction, and possibly worsening placental ischemia. In a real tour de force, Karumanchi and colleagues demonstrated that the administration of sFlt1 to pregnant rats induces the classic lesion of preeclampsia:
- Hypertension
- Proteinuria
- Glomerular endotheliosis
These results argue that sFlt-1 is “the” pathogenic entity responsible for causing the renal lesion of preeclampsia. This heralded great potential to actually offer novel treatment beyond blood pressure control, magnesium, and delivery. In 2010 another breakthrough came. A pilot study published in Circulation attempted to remove sFlt-1 with the use of apheresis treatment; however, this was not a randomized study. The authors examined the efficacy of using a negatively charged dextran sulfate cellulose column to adsorb sFlt-1 in 5 women with preterm preeclampsia and increased sFlt-1 levels. They showed that with one apheresis treatment, levels of sFlt-1 decreased. They also treated 3 women with very preterm preeclampsia and elevated circulating sFlt-1 levels with multiple rounds of apheresis. Again, they observed decreased sFlt-1 levels, reduced proteinuria, and stabilization of blood pressure without any evident adverse events. Just to note that there was no control group in this study. However, since this report nothing more has surfaced. A quick look at ClinicalTrials.gov shows a few trials that are currently in various stages of recruiting. Team sFlt-1 will be a difficult challenge for any team in this year’s NephMadness. A real UNLV from the Jerry Tarkanian era.
ACOG Blood Pressure Goal < 160/110 in Preeclampsia vs Lower Blood Pressure Goal in Preeclampsia
 ACOG Blood Pressure Goal < 160/110 in Preeclampsia
ACOG Blood Pressure Goal < 160/110 in Preeclampsia
Hypertension in pregnancy can be due to multiple etiologies:
- Pre-existing chronic hypertension
- Preeclampsia
- Gestational hypertension
- Preeclampsia superimposed on chronic hypertension
The prevalence of chronic hypertension among pregnant women has increased by 50% from 1995 to 2008 (0.9% to 1.5%). Gestational hypertension has likewise increased 184% from 1987 to 2004. Regardless of the etiology, the therapeutic goals in the treatment of hypertension are to prevent maternal morbidity (stroke, cardiac complications) while maintaining placental circulation and limiting medication toxicity to both the fetus and mother.
The definition of hypertension in pregnancy is the same as in non-pregnant patients (see last year’s NephMadness winner, JNC8), however the American College of Obstetrics and Gynecology (ACOG) doesn’t recommend drug intervention until the blood pressure reaches or exceeds 160/110 and then recommends physicians to target SBP between 140 and 160 and a DBP between 90 and 100. They point to a case series by Martin et al in which 28 patients with either eclampsia or preeclampsia who sustained strokes were scrutinized in terms of blood pressure control. Martin et al showed that all strokes occurred at SBP > 155 mm Hg and all but one exceeded 160 mm Hg. Diastolic blood pressures and mean arterial pressures were not nearly as reliable at predicting stroke. Thus, the argument is that treatment is warranted only if the systolic is sustained over 160 mm Hg.
Doctors urging more aggressive blood pressure control, more in line with the rest of medicine, have been stymied repeatedly by the Cochrane review which has not been able to find any benefit to treating mild to moderate hypertension during pregnancy. This review includes the most recent RCT published this past January which randomized ~1000 pregnant women to either a target diastolic BP of 100 mm Hg (less tight control) or 85 mm Hg (tight control) and could not detect a difference in pregnancy loss or high-level neonatal care. There was also no difference in serious maternal complications or preeclampsia.
For now the weight of data rests on the side of decreased medical interventions and a “let it ride” mentality when it comes to mild to moderate hypertension in pregnancy. This will be a tough battle for first round supremacy for sure.
Lower Blood Pressure Goal in Preeclampsia
Obstetricians are more tolerant of hypertension than other fields in medicine. The reason behind this is three-fold:
- The outcome of interest is delivery of a healthy baby, and after delivery most of the hypertension and all of the controversy melts away. Given the limited time exposure there are fewer maternal events to worry about.
- There is legitimate concern regarding fetal exposure to antihypertensives: renin-angiotensin-aldosterone inhibitors are known teratogens and diuretics in the third trimester can induce premature delivery.
- Lowering blood pressure could adversely affect uterine hemodynamics, leading to decreased fetal growth and poor fetal outcomes.
Let’s take a look at the fetal growth story first. In the absence of well done, adequately powered clinical trials, the alarmists point to this meta-analysis from 2000 which found a decrease in fetal weight of 145 grams for every 10-mm Hg decrement in blood pressure. However the R2 was only 0.15, meaning that other factors were much more important than blood pressure at determining fetal weight. In the most recent RCT published in NEJM, there was no difference in the proportion of babies born at less than the tenth or third percentile for weight.
In regard to exposure to teratogens, there are certainly drugs that should be avoided but there is a cohort of drugs including methyldopa, labetalol, nifedipine, hydralazine and thiazides that are commonly utilized in pregnancy and have a long safety record.
And the last reason doctors are tolerant of hypertension in pregnancy is the belief that since pregnancy is a time-limited medical condition, there is little maternal morbidity from mild to moderate hypertension. This is probably false as the Martins et al case series of 28 women who had hypertensive strokes shows hypertensive morbidity is real and likely avoidable with judicious treatment of hypertension. Additionally, more aggressive use of antihypertensives in moderate maternal hypertension was shown to reduce the progression to severe hypertension while helping to avoid thrombocytopenia and elevated liver enzymes.
People are endlessly worrying about the over medicalization of natural human processes, but hypertension in pregnancy is pathologic and we have the means to treat it and should to prevent serious maternal complications.
Preeclampsia Due to CKD vs CKD Due to Preeclampsia
 This is a chicken-and-egg–like paradox. We know that CKD is a risk factor for preeclampsia and we know that having preeclampsia predicts future kidney disease. The association of adverse renal and cardiovascular outcomes after preeclampsia is certain and conclusive. What remains to be determined is the etiology of this association. Does the preeclampsia cause renal and vascular damage that subsequently manifests as renal and cardiovascular disease? Or is preeclampsia merely the first symptom in a patient with underlying kidney disease that would have ultimately presented later even if the patient never became pregnant or developed preeclampsia?
This is a chicken-and-egg–like paradox. We know that CKD is a risk factor for preeclampsia and we know that having preeclampsia predicts future kidney disease. The association of adverse renal and cardiovascular outcomes after preeclampsia is certain and conclusive. What remains to be determined is the etiology of this association. Does the preeclampsia cause renal and vascular damage that subsequently manifests as renal and cardiovascular disease? Or is preeclampsia merely the first symptom in a patient with underlying kidney disease that would have ultimately presented later even if the patient never became pregnant or developed preeclampsia?
Preeclampsia Due to CKD
Pregnancy in patients with CKD is surprisingly rare. The HUNT II study was an epidemiologic study performed in Nord-Trøndelag, Norway that recorded MDRD eGFRs for 66,149 people.
- In the subsequent 11 years there were 5,655 singleton pregnancies in women in HUNT II.
- – Only 6 were to women with an eGFR < 60 mL/min
- – None were to women with eGFR < 30 mL/min.
In the HUNT II study, no association was found between decreased eGFR and preeclampsia. However, very few patients had significant CKD. In contrast, women with hypertension and an eGFR < 90 had an increased odds ratio for preeclampsia:
- OR of 1.82 in women with hypertension and GFR > 90 vs normotensive women
- OR of 4.24 in women with hypertension and GFR < 90
Most other reports on preeclampsia in CKD have been case series. Cunningham reported 64% of women with severe CKD developed preeclampsia. In a case-control study women with CKD had an OR of 7.2 for preeclampsia.
While the data for increased risk of ESRD and kidney biopsy are compelling, what is surprising is that the results of the biopsies and the etiologies of ESRD are no different than found in the surrounding background population. One would think that if preeclampsia caused kidney disease it would cause one particular type of kidney disease that would be identified by providers at dialysis or at least by pathologists at biopsy. This may mean that the association of preeclampsia and future kidney disease is not due to kidney damage from preeclampsia but already existing subtle CKD increasing the risk for both the preeclampsia and subsequent kidney disease.
CKD Due to Preeclampsia
The more one looks at patients with preeclampsia the more it looks like the disease has significant health effects that last for years after the pregnancy. Hypertension, ischemic heart disease, cardiovascular death (8 fold higher in women with preterm preeclampsia compared to women with either term, or no preeclampsia!), albuminuria, and future kidney biopsy have all been shown to occur at increased rates after preeclampsia.
The most feared renal end point is dialysis dependence. Vikse et al did a comprehensive study of the association between preeclampsia and ESRD in Norway. Of the 570,433 women who had a baby between 1967 and 1991, 20,918 developed preeclampsia with the first pregnancy and 8,531 developed preeclampsia with a second pregnancy.
Preeclampsia during the first pregnancy was associated with a RR of ESRD of 4.7. The authors demonstrated a clear dose-dependent relationship with increased risk with more episodes of preeclampsia. In women with multiple pregnancies, having preeclampsia in a later pregnancy was worse than having preeclampsia in an earlier pregnancy.
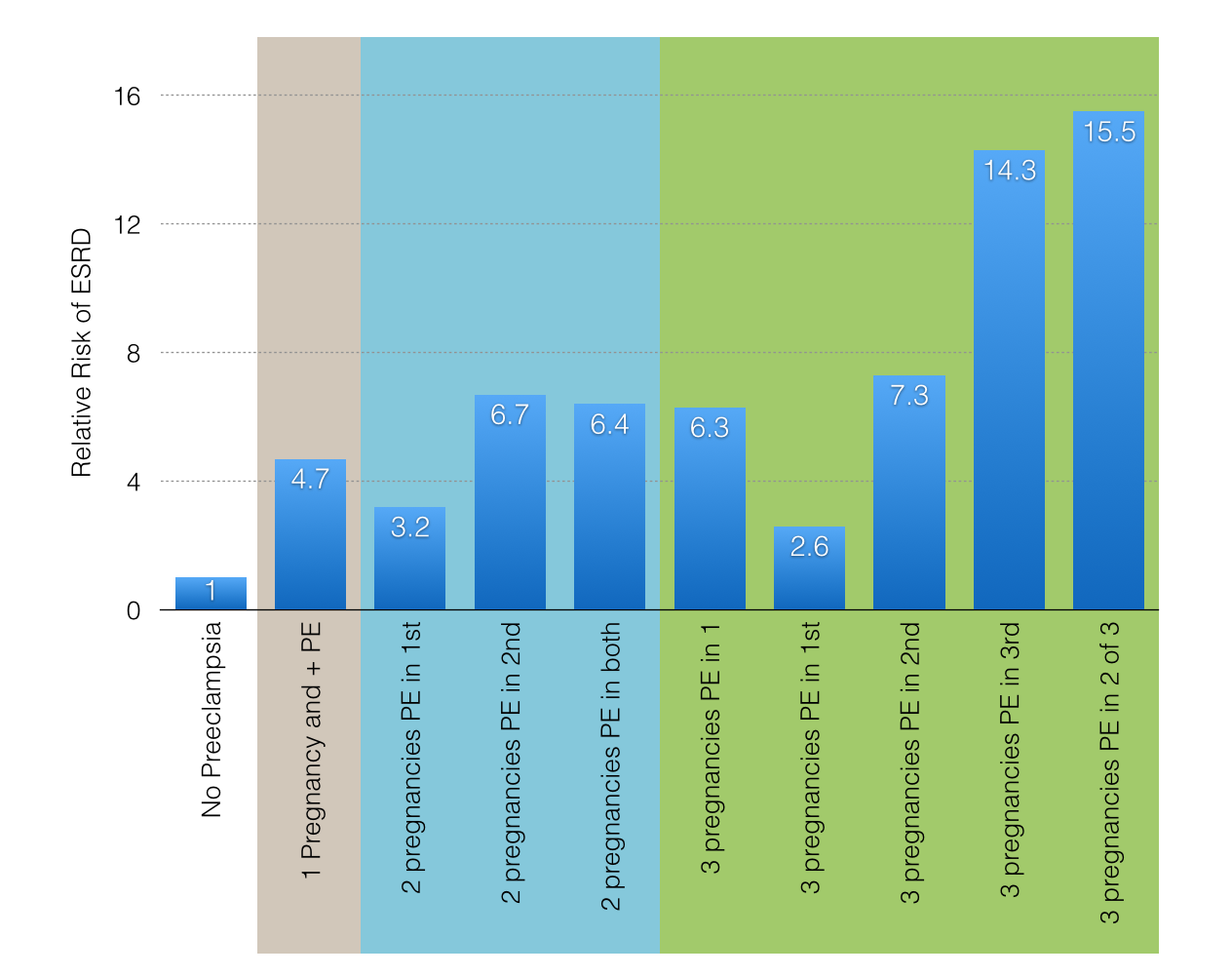
Data from Vikse et al.
To strengthen the findings, the authors examined the data after excluding patients with pre-pregnancy hypertension, diabetes or renal or rheumatic disease. Even with these patients censored the risk of ESRD remained, thus strengthening the assertion that the preeclampsia caused the kidney disease rather than the other way around.
The association of pregnancy complications and ESRD is not limited to preeclampsia, having a low birth weight baby or premature delivery has also been associated with increased cardiovascular disease and ESRD.
Could preeclampsia and ESRD have a common cause that puts patients at risk for both diseases? Obesity, hypertension, insulin resistance, and endothelial function are all risk factors for both outcomes. Antiangiogenic factors are another possibility leading to preeclampsia and for CKD.
Despite the accumulating evidence of harm that follows preeclampsia it is important to keep in mind that the vast majority (more than 99%) of women with preeclampsia never develop dialysis-dependent renal failure.
Pregnancy in ESRD vs Pregnancy in Kidney Transplant
 Pregnancy in ESRD
Pregnancy in ESRD
Pregnancy in dialysis-dependent kidney failure leads to significant maternal and fetal morbidity and mortality. As a result, most experts advise women who want children to wait until a successful transplant, but when the biologic clock is ticking and the PRA is high and the wait for a transplant can be years. As such, pregnancy while on dialysis may actually be the best option. And sometimes it just happens.
The first hurdle with pregnancy in dialysis is fertility. Menstrual irregularities typically begin with a GFR of 15 mL/min and amenorrhea occurs at a GFR of 5 mL/min. Even when menstruation is maintained, ovulation can be absent due to loss of the LH surge. In a registry of female dialysis patients of childbearing age from the 90s the fertility rate was 2.4% for HD and 1.1% for PD over 4 years (0.5% per year for the entire registry). There is data that shows that fertility improves with a higher doses of dialysis. Also, registries and case series include women who get pregnant before starting dialysis and then required dialysis later in the pregnancy.
The earliest outcomes reported for pregnancies while on dialysis tended to be poor with only ~23% resulting in a live baby, with a mean gestational age of 32 weeks. But in 1998 Okundaye at al reported a trend toward better fetal survival in women who received more than 20 hours of dialysis a week. Three series have been published with all of the pregnancies occurring after 2000, and they have reported live delivery rates from 86% to 100% and dialysis hours from 20-48 hours per week (Eroğlu et al, Haase et al, Barua et al). In the two series that reported 100% live births, both employed intensive dialysis, either hemodiafiltration for 28 hours a week or nocturnal hemodialysis 48 hours a week. More impressive is that, in neither of those series did the women have significant residual renal function.
Anemia is a difficult problem in pregnancy given reports of increased EPO and iron requirements in pregnancy. Additionally estimating dry weight can be a challenge. Usually there is little weight gain in the first trimester followed by up to a pound per week during the second and third trimester. Physicians should target weight gain of 25 to 35 pounds. Frequent reevaluation of dry weight is needed and extra care should be taken to avoid hypotension.
Another issue that can complicate pregnancy is fetal polyhydramnios (or excessive amniotic fluid). Polyhydramnios is postulated to be the result of fetal solute diuresis secondary to a high urea concentration. This was corrected by increased dialysis time and has not been reported in contemporary series with intensive hemodialysis.
Pregnancy in dialysis is rare, but increasingly possible with good outcomes. The most generalizable lesson from pregnancy and dialysis is that a formerly hopeless situation has been completely transformed by a radical rethinking of what the appropriate dose of dialysis should be. Might pregnancy be a lantern shining a the way to think about dialysis dose in general?
Pregnancy in Kidney Transplant
Fertility usually returns to ESRD patients within ~3-4 months after a kidney transplant. However, menopause typically occurs 4-5 years earlier in patients with ESRD than in the general population, so this should be considered if there is a delay in the return of fertility. Pregnancy is fairly common after kidney transplantation. Despite a number of registries to track pregnancy after kidney transplant a minority of pregnancies are actually tracked. This means there is, almost certainly, a significant reporting bias in these registries that needs to be kept in mind when looking at the data.
Guidelines suggest delaying pregnancy until after the peritransplantation period as this is the time patients are exposed to the most fetotoxic and teratogenic anti-rejection medications. Previously, guidelines have suggested delaying pregnancy at least 2 years. However, given the increasing age of transplant patients these guidelines are being replaced with more realistic guidelines. Look for a stable creatinine less than 1.5 mg/dL with less than 500 mg/24 hours of proteinuria and of course no fetotoxic infections (CMV, etc) or fetotoxic/teratogenic medications prior to pregnancy.
Current recommendations advise against mycophenolate mofetil (MMF) and rapamycin for 6 weeks before pregnancy. Though some recommend using higher doses of calcineurin inhibitors, most recommend doctors maintain pre-transplant drug levels. Frequent monitoring may be required due to changes in eGFR and plasma volume with pregnancy. Transplant rejection can be difficult to detect clinically but kidney biopsy is generally considered safe during pregnancy, as is methylprednisolone to treat rejection. The gravid liver may be more prone to azathioprine toxicity, so regular assessment of liver enzymes is recommended. Tacrolimus pharmacokinetics can be altered during pregnancy and dose adjustments could be required.
The National Transplant Pregnancy Registry reports that about a third of post-transplant pregnancies are complicated by preeclampsia, possibly related to calcineurin inhibitor effects. Most registries report a high risk of preterm birth and low birth weight, on the order of 50-60%, and usually it is due to maternal or fetal compromise, rather than spontaneous labor.
Patients with functional kidney transplants have been conceiving and delivering babies for ~50 years. They represent a high-risk population and care must be taken for the fetus, mother, and graft but good outcomes are still likely and it is probably the best road through ESRD to motherhood.

– Post written and edited by Drs. Joel Topf, Matthew Sparks, and Phyllis August.


Leave a Reply